ASTM E1197-12(2021)
(Guide)Standard Guide for Conducting a Terrestrial Soil-Core Microcosm Test
Standard Guide for Conducting a Terrestrial Soil-Core Microcosm Test
SIGNIFICANCE AND USE
4.1 This guide provides a test procedure for evaluating the potential ecological impacts and environmental transport of a chemical in an agricultural (tilled, low-till, or no-till) or natural field soil ecosystem that may be released or spilled into the environment. The suggested test procedures are designed to supply site-specific information for a chemical without having to perform field testing. (See EPA 560/6-82-002 and EPA 560/6-82-003.)
4.2 This guide is not specifically designed to address fate of chemicals in soils of forested ecosystems. However, with some modifications, it may be adapted for that purpose by the individual investigator.
4.3 Specifically, this guide is used to determine the effect of a chemical on (1) growth and reproduction of either natural grassland vegetation or crops, and (2) nutrient uptake and cycling within the soil/plant system. Additionally, the soil-core microcosm will provide information on (1) potential for bioaccumulation (enrichment) of the chemical into plant tissues, and (2) the potential for and rate of transport of the chemical through soil to groundwater.
4.4 The results of this test should be used in conjunction with information on the chemical and biological activity of the test substance to assess the relative environmental hazard and the potential for environmental movement once released.
4.5 The test methods described in this guide are designed specifically for liquid or solid materials. Significant modifications of the exposure system would be necessary to accommodate chemicals that are volatile or that may be released in a gaseous or aerosolized form. For methods that could be adapted for use with volatile or gaseous test substances see Refs (3, 4, 5, 6).
4.6 Results of a multi-year soil-core microcosm test have been correlated with data derived from a series of multi-year field plot tests for a limited number of materials. Information on the correlation between microcosm and field results can be fo...
SCOPE
1.1 This guide defines the requirements and procedures for using soil-core microcosms to test the environmental fate, ecological effects, and environmental transport of chemicals that may enter terrestrial ecosystems. The approach and the materials suggested for use in the microcosm test are also described.
1.2 This guide details a procedure designed to supply site-specific or possibly regional information on the probable chemical fate and ecological effects in a soil system resulting from the release or spillage of chemicals into the environment in either liquid or solid form.
1.3 Experience has shown that microcosms are most helpful in the assessment process after preliminary knowledge about the chemical properties and biological activity have been obtained. Data generated from the test can then be used to compare the potential terrestrial environmental hazards of a chemical.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2020
- Technical Committee
- E50 - Environmental Assessment, Risk Management and Corrective Action
- Drafting Committee
- E50.47 - Biological Effects and Environmental Fate
Relations
- Effective Date
- 01-Mar-2019
- Effective Date
- 15-Jul-2017
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jul-2010
- Effective Date
- 15-Jun-2009
- Effective Date
- 15-May-2009
- Effective Date
- 15-May-2009
- Effective Date
- 01-May-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Apr-2008
- Effective Date
- 15-Oct-2007
- Refers
ASTM D422-63(2007)e2 - Standard Test Method for Particle-Size Analysis of Soils (Withdrawn 2016) - Effective Date
- 15-Oct-2007
- Effective Date
- 01-Nov-2006
- Effective Date
- 01-Mar-2005
- Effective Date
- 01-Jul-2004
Overview
ASTM E1197-12(2021), "Standard Guide for Conducting a Terrestrial Soil-Core Microcosm Test," provides a comprehensive methodology for assessing the ecological and environmental impact of chemicals introduced into terrestrial soils. Developed by ASTM International, this guide outlines procedures for evaluating the environmental fate, ecological effects, and potential movement of chemicals in agricultural and natural field soil ecosystems. The soil-core microcosm approach allows for the generation of site-specific data without the need for full-scale field trials, making it a valuable tool in environmental risk assessment and regulatory decision-making.
Key Topics
- Purpose and Scope
- Defines requirements for using soil-core microcosms to simulate and study the behavior and effects of chemicals in terrestrial soils.
- Emphasizes applications in agricultural soils (tilled, low-till, no-till) and natural field conditions.
- Notes that, with modification, the methodology can extend to forested ecosystem soils.
- Test System Description
- Soil-core microcosms are intact samples extracted from the local ecosystem, maintaining the natural assemblage of soil organisms and vegetation or crops.
- The system simulates environmental conditions, including light, water, and relevant forcing functions, to mirror field scenarios closely.
- Chemical Fate and Ecological Effects
- Assesses chemical impacts on plant growth, reproduction, and soil nutrient cycling.
- Measures bioaccumulation potential and evaluates chemical transport through soil to groundwater.
- Provides data for comparing relative environmental hazards among chemicals.
- Limits and Adaptability
- Designed for testing chemicals introduced as liquids or solids.
- Requires adaptation for volatile or gaseous substances.
- Not a substitute for field tests but intended as a supporting tool to provide robust, site-specific information.
Applications
- Regulatory Environmental Risk Assessment
- Supports compliance with environmental protection and risk management guidelines by providing reproducible, scalable testing procedures.
- Complements data from laboratory and field tests, aiding in comprehensive chemical hazard evaluation.
- Agricultural and Land Management Practices
- Assists in evaluating potential ecological impacts of pesticides, fertilizers, and accidental chemical spills in soil environments.
- Useful for agricultural researchers analyzing effects on crop growth, productivity, and soil health.
- Chemical Product Development
- Enables manufacturers and developers to anticipate ecological risks associated with new agricultural chemicals.
- Provides meaningful data prior to large-scale field application.
- Site-Specific Environmental Monitoring
- Delivers localized assessment of chemical behavior in soils, assisting in informed decision-making for contaminated site remediation and environmental restoration projects.
Related Standards
- ASTM D422 (Particle-Size Analysis of Soils)
- ASTM D2216 (Laboratory Determination of Water Content of Soil and Rock)
- ASTM D2167 (Density and Unit Weight of Soil in Place)
- ASTM D2488 (Description and Identification of Soils)
- EPA 560/6-82-002 & EPA 560/6-82-003 (Environmental Effects and Chemical Fate Test Guidelines)
Practical Value
Implementing ASTM E1197-12(2021) helps organizations reliably predict and monitor how chemicals will behave once introduced into terrestrial environments. By simulating key soil and ecosystem parameters, researchers obtain actionable data on chemical fate, movement, and impact without the complexity or variability of field studies. This standard is instrumental for regulatory compliance, sustainable agricultural management, and the responsible development and deployment of chemicals impacting soil ecosystems.
Keywords: soil-core microcosm test, chemical fate, ecological effects, environmental transport, ASTM E1197, terrestrial soil ecosystems, environmental risk assessment, agricultural soil testing, bioaccumulation, groundwater protection, site-specific chemical impact
Buy Documents
ASTM E1197-12(2021) - Standard Guide for Conducting a Terrestrial Soil-Core Microcosm Test
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM E1197-12(2021) is a guide published by ASTM International. Its full title is "Standard Guide for Conducting a Terrestrial Soil-Core Microcosm Test". This standard covers: SIGNIFICANCE AND USE 4.1 This guide provides a test procedure for evaluating the potential ecological impacts and environmental transport of a chemical in an agricultural (tilled, low-till, or no-till) or natural field soil ecosystem that may be released or spilled into the environment. The suggested test procedures are designed to supply site-specific information for a chemical without having to perform field testing. (See EPA 560/6-82-002 and EPA 560/6-82-003.) 4.2 This guide is not specifically designed to address fate of chemicals in soils of forested ecosystems. However, with some modifications, it may be adapted for that purpose by the individual investigator. 4.3 Specifically, this guide is used to determine the effect of a chemical on (1) growth and reproduction of either natural grassland vegetation or crops, and (2) nutrient uptake and cycling within the soil/plant system. Additionally, the soil-core microcosm will provide information on (1) potential for bioaccumulation (enrichment) of the chemical into plant tissues, and (2) the potential for and rate of transport of the chemical through soil to groundwater. 4.4 The results of this test should be used in conjunction with information on the chemical and biological activity of the test substance to assess the relative environmental hazard and the potential for environmental movement once released. 4.5 The test methods described in this guide are designed specifically for liquid or solid materials. Significant modifications of the exposure system would be necessary to accommodate chemicals that are volatile or that may be released in a gaseous or aerosolized form. For methods that could be adapted for use with volatile or gaseous test substances see Refs (3, 4, 5, 6). 4.6 Results of a multi-year soil-core microcosm test have been correlated with data derived from a series of multi-year field plot tests for a limited number of materials. Information on the correlation between microcosm and field results can be fo... SCOPE 1.1 This guide defines the requirements and procedures for using soil-core microcosms to test the environmental fate, ecological effects, and environmental transport of chemicals that may enter terrestrial ecosystems. The approach and the materials suggested for use in the microcosm test are also described. 1.2 This guide details a procedure designed to supply site-specific or possibly regional information on the probable chemical fate and ecological effects in a soil system resulting from the release or spillage of chemicals into the environment in either liquid or solid form. 1.3 Experience has shown that microcosms are most helpful in the assessment process after preliminary knowledge about the chemical properties and biological activity have been obtained. Data generated from the test can then be used to compare the potential terrestrial environmental hazards of a chemical. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This guide provides a test procedure for evaluating the potential ecological impacts and environmental transport of a chemical in an agricultural (tilled, low-till, or no-till) or natural field soil ecosystem that may be released or spilled into the environment. The suggested test procedures are designed to supply site-specific information for a chemical without having to perform field testing. (See EPA 560/6-82-002 and EPA 560/6-82-003.) 4.2 This guide is not specifically designed to address fate of chemicals in soils of forested ecosystems. However, with some modifications, it may be adapted for that purpose by the individual investigator. 4.3 Specifically, this guide is used to determine the effect of a chemical on (1) growth and reproduction of either natural grassland vegetation or crops, and (2) nutrient uptake and cycling within the soil/plant system. Additionally, the soil-core microcosm will provide information on (1) potential for bioaccumulation (enrichment) of the chemical into plant tissues, and (2) the potential for and rate of transport of the chemical through soil to groundwater. 4.4 The results of this test should be used in conjunction with information on the chemical and biological activity of the test substance to assess the relative environmental hazard and the potential for environmental movement once released. 4.5 The test methods described in this guide are designed specifically for liquid or solid materials. Significant modifications of the exposure system would be necessary to accommodate chemicals that are volatile or that may be released in a gaseous or aerosolized form. For methods that could be adapted for use with volatile or gaseous test substances see Refs (3, 4, 5, 6). 4.6 Results of a multi-year soil-core microcosm test have been correlated with data derived from a series of multi-year field plot tests for a limited number of materials. Information on the correlation between microcosm and field results can be fo... SCOPE 1.1 This guide defines the requirements and procedures for using soil-core microcosms to test the environmental fate, ecological effects, and environmental transport of chemicals that may enter terrestrial ecosystems. The approach and the materials suggested for use in the microcosm test are also described. 1.2 This guide details a procedure designed to supply site-specific or possibly regional information on the probable chemical fate and ecological effects in a soil system resulting from the release or spillage of chemicals into the environment in either liquid or solid form. 1.3 Experience has shown that microcosms are most helpful in the assessment process after preliminary knowledge about the chemical properties and biological activity have been obtained. Data generated from the test can then be used to compare the potential terrestrial environmental hazards of a chemical. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1197-12(2021) is classified under the following ICS (International Classification for Standards) categories: 13.080.10 - Chemical characteristics of soils. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1197-12(2021) has the following relationships with other standards: It is inter standard links to ASTM D2216-19, ASTM D2488-17, ASTM D2167-15, ASTM D2216-10, ASTM D2488-09a, ASTM D3867-09, ASTM D2488-09, ASTM D511-09, ASTM D511-08, ASTM D2167-08, ASTM D422-63(2007), ASTM D422-63(2007)e2, ASTM D2488-06, ASTM D2216-05, ASTM D3867-04. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1197-12(2021) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1197 − 12 (Reapproved 2021)
Standard Guide for
Conducting a Terrestrial Soil-Core Microcosm Test
This standard is issued under the fixed designation E1197; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D422 Test Method for Particle-SizeAnalysis of Soils (With-
drawn 2016)
1.1 This guide defines the requirements and procedures for
D511 Test Methods for Calcium and Magnesium In Water
using soil-core microcosms to test the environmental fate,
D515 Test Method for Phosphorus In Water (Withdrawn
ecological effects, and environmental transport of chemicals
1997)
that may enter terrestrial ecosystems. The approach and the
D1426 Test Methods for Ammonia Nitrogen In Water
materials suggested for use in the microcosm test are also
D2167 Test Method for Density and Unit Weight of Soil in
described.
Place by the Rubber Balloon Method
1.2 This guide details a procedure designed to supply
D2216 Test Methods for Laboratory Determination of Water
site-specific or possibly regional information on the probable
(Moisture) Content of Soil and Rock by Mass
chemical fate and ecological effects in a soil system resulting
D2488 Practice for Description and Identification of Soils
from the release or spillage of chemicals into the environment
(Visual-Manual Procedures)
in either liquid or solid form.
D3867 Test Methods for Nitrite-Nitrate in Water
2.2 U.S. Environmental Protection Agency:
1.3 Experience has shown that microcosms are most helpful
in the assessment process after preliminary knowledge about Environmental Effects Test Guidelines, EPA 560⁄6-82-002,
the chemical properties and biological activity have been
obtained. Data generated from the test can then be used to Chemical Fate Test Guideline, EPA 560⁄6-82-003, 1982
compare the potential terrestrial environmental hazards of a
3. Terminology
chemical.
3.1 Definitions:
1.4 This standard does not purport to address all of the
3.1.1 soil-core terrestrial microcosm—an intact soil-core
safety concerns, if any, associated with its use. It is the
containing the natural assemblages of biota surrounded by the
responsibility of the user of this standard to establish appro-
boundary material. The system includes all equipment,
priate safety, health, and environmental practices and deter-
facilities, and instrumentation necessary to maintain, monitor,
mine the applicability of regulatory limitations prior to use.
and control the environment.
1.5 This international standard was developed in accor-
dance with internationally recognized principles on standard-
3.2 Definitions of Terms Specific to This Standard:
ization established in the Decision on Principles for the
3.2.1 terrestrial microcosm or micro-ecosystem—aphysical
Development of International Standards, Guides and Recom-
model of an interacting community of autotrophs, omnivores,
mendations issued by the World Trade Organization Technical
herbivores, carnivores and decomposers within an intact soil
Barriers to Trade (TBT) Committee.
profile. The forcing functions, for example, light intensity and
duration, water quality and watering regime, temperature, and
2. Referenced Documents
toxicant dose for the test system, are under the investigator’s
control. This test system is distinguished from test tube and
2.1 ASTM Standards:
single-species toxicity tests by the presence of a natural
1 3
ThisguideisunderthejurisdictionofASTMCommitteeE50onEnvironmental The last approved version of this historical standard is referenced on
Assessment, Risk Management and CorrectiveAction and is the direct responsibil- www.astm.org.
ity of Subcommittee E50.47 on Biological Effects and Environmental Fate. Available from the Office of Pesticides andToxic Substances,Washington, DC.
Current edition approved Jan. 1, 2021. Published February 2021. Originally Also available as PB82 – 23992 from National Technical Information Service
approved in 1987. Last previous edition approved in 2012 as E1197–12. DOI: (NTIS), United States Department of Commerce, 5285 Port Royal Rd., Spring-
10.1520/E1197-12R21. field, VA 22161.
2 5
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Available from Office of Pesticides and Toxic Substances, Washington, DC.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Also available as PB82 – 233008 from National Technical Information Service
Standards volume information, refer to the standard’s Document Summary page on (NTIS), United States Department of Commerce, 5285 Port Royal Rd., Spring-
the ASTM website. field, VA 22161.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1197 − 12 (2021)
assemblage of organisms. This assemblage creates a higher 3.2.7 biota—the biota of the microcosm are characterized
order of ecological complexity and, thus, provides the capacity by the organisms in the soil at the time of extraction (1, 2) and
to evaluate chemical effects on component interactions and bythenaturalvegetationorcropsintroducedastheautotrophic
ecological processes. Certain features of this test system, component. The biota may include all heterotrophic and
however, set limits on the types of questions that can be carnivorousinvertebratestypicallyfoundinthesoilandallsoil
addressed. Those limitations are related to scale and sampling, and plant microbes.
which in turn constrain both (a) the type of ecosystems and
4. Significance and Use
species assemblages on which one can gain information, and
(b) the longevity of the test system.
4.1 This guide provides a test procedure for evaluating the
potential ecological impacts and environmental transport of a
3.2.2 physical, chemical, and biological conditions of test
chemical in an agricultural (tilled, low-till, or no-till) or natural
system—determined by the type of ecosystem from which the
field soil ecosystem that may be released or spilled into the
test system was extracted and by either the natural vegetation
environment. The suggested test procedures are designed to
in the ecosystem or the crops selected for planting. Vegetation
supply site-specific information for a chemical without having
and crop selection are constrained and determined by the size
to perform field testing. (See EPA 560⁄6-82-002 and
(width and depth) of the soil core extracted.
EPA 560⁄6-82-003.)
3.2.3 boundaries—the boundaries of the test system are
4.2 This guide is not specifically designed to address fate of
determinedbythesizeofthesoil-coreandthespaceneededfor
chemicals in soils of forested ecosystems. However, with some
vegetative growth.
modifications, it may be adapted for that purpose by the
3.2.4 light—light for the test system can be supplied by
individual investigator.
artificial means in either a growth chamber or a greenhouse, or
4.3 Specifically, this guide is used to determine the effect of
it can be the natural photoperiod occurring in a greenhouse. If
a chemical on (1) growth and reproduction of either natural
the test is performed in a growth chamber, the daily photope-
grassland vegetation or crops, and (2) nutrient uptake and
riod should be equal to or greater than the average monthly
cycling within the soil/plant system.Additionally, the soil-core
incident radiation (quantity and duration) for the month in
microcosm will provide information on (1) potential for
which the test is being simulated. During extremely short
bioaccumulation (enrichment) of the chemical into plant
natural photoperiods, which might not allow for flowering or
tissues, and (2) the potential for and rate of transport of the
seed-set, photoperiod should be artificially lengthened to in-
chemical through soil to groundwater.
duce those responses. The spectral quality of visible light
4.4 The results of this test should be used in conjunction
supplied during testing should simulate that of sunlight (for
with information on the chemical and biological activity of the
example, include commercially available visible full-spectrum
test substance to assess the relative environmental hazard and
lamps).
the potential for environmental movement once released.
3.2.5 water—water for the test system should either be
4.5 The test methods described in this guide are designed
purified, untreated laboratory water, should be precollected,
specifically for liquid or solid materials. Significant modifica-
filtered rainwater from the site or region being evaluated, or
tions of the exposure system would be necessary to accommo-
formulated rainwater (for example, based on rainfall of the
date chemicals that are volatile or that may be released in a
region). Chemical characterization of the water, either labora-
gaseous or aerosolized form. For methods that could be
tory or rainwater, is required and must be performed usingTest
adapted for use with volatile or gaseous test substances see
Methods D511, D515, D1426, and D3867.
Refs (3, 4, 5, 6).
3.2.6 soil—the soil-core used for the microcosm test should
4.6 Results of a multi-year soil-core microcosm test have
be an intact, undisturbed (nonhomogenized) core extracted
been correlated with data derived from a series of multi-year
from a soil type typical of the region or site of interest. The
field plot tests for a limited number of materials. Information
core should be of sufficient depth to allow a full growing
on the correlation between microcosm and field results can be
season for the natural vegetation or the crops selected, without
found in Refs (7, 8, 9, 10).
causing the plants to become significantly rootbound. Distur-
bances during extraction and preparation should be kept to a
5. Chemical Characterization of Test Substance and Soil
minimum. It should be noted that soil characteristics play an
5.1 Information Required on Test Substance:
important role in how the microcosm responds to a test
5.1.1 Minimum information required to properly design and
substance. In addition, within-site soil heterogeneity also
conductanexperimentonatestchemicalincludesthechemical
influences the microcosm response and contributes to a loss of
source, composition, degree of purity, nature and quantity of
sensitivity of the test. The approach used in this test system,
however, is based on a comparison of responses among and
between treatments rather than on the absolute values mea- 6
The boldface numbers in parentheses refer to a list of references at the end of
sured. this guide.
E1197 − 12 (2021)
anyimpuritiespresent,andcertainphysiochemicalinformation this guide. According to Refs (6, 18), modification of the test
such as water solubility and vapor pressure at 25 °C (11, 12). system should be useful for handling gaseous or aerosolized
chemicals.
Ideally,thestructureofthetestchemicalshouldalsobeknown,
including functional groups, nature and position of substituting
5.2 Information Required on Soil:
groups, and degree of saturation. The octanol-water-partition
5.2.1 Soil sorption of an organic molecule depends on
coefficient,thedissociationconstant,thedegreeofpolarity,and
several properties of the chemical (molecular size, ionic
the pH of both pure and serial dilutions should also be known.
speciation, acid-base properties, polarity, and nature of func-
Where mixtures are involved or where a significant impurity
tional groups) and of the soil (for example, organic matter
(>1 %) occurs, data must be available on as many components
content, clay content, clay mineralogy and nature, pH, water
as practical. However, the octanol-water-partition coefficient
content, bulk density, cation exchange capacity, and percent
(K ) stands out as a key value for lipophilic compounds. Soil
base saturation). Highly sorbed chemicals may displace inor-
ow
partition coefficient (K ) can be determined or estimated, and
ganicnutrientionsfromexchangesitesinthesoilandalsomay
d
organiccarbonpartitioncoefficient(K )canbeestimatedfrom be effectively immobilized, depending on soil pH. Thus,
oc
log K using the organic matter content. Water solubility can
chemicals attracted more strongly to soil surfaces than to water
ow
be predicted with some degree of accuracy from log K if this may be very immobile in soil. In some cases, this may render
ow
value is less than seven. In combination with other chemical the compound relatively resistant to biodegradation. In other
cases, however, immobilization of the compound on soil
characteristics, log K can also be used to estimate Henry’s
ow
Law Constant and thus provide a rough estimate of the particles may render it susceptible to extracellular enzymatic
degradation. Specific information on descriptive data required
potential volatility of the test substance from soil solutions.
for soil can be found in 6.2.2.
5.1.2 Several tests may be needed to supply information on
environmental mobility and stability. Support information on
6. Terrestrial Microcosm Extraction and Maintenance
phytotoxicity, the physicochemical nature of the chemical, its
mammalian toxicity, or its ecological effects (for example,
6.1 Microcosm and Chamber Design:
species-specific LC , invertebrate toxicity, biodegradability)
6.1.1 A ≥ 60-cm deep by ≥ 10-cm diameter terrestrial
not only assist in proper design of the microcosm experiment, soil-core microcosm is designed to yield pertinent information
but also are useful in assessing the fate and effects of the
about a chemical for either a natural grassland ecosystem or an
chemical in a terrestrial microcosm. If the chemical is radio- agricultural ecosystem planted with a multiple-species crop
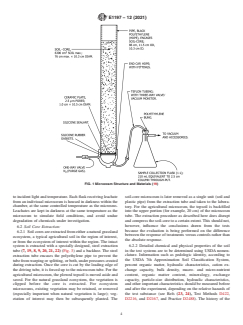
actively labeled, the position and specific element to be labeled (Fig.1) (7, 19, 8, 9, 20, 21).Theagriculturalmicrocosmisa10
should be specified. to 17-cm diameter tube of plastic pipe that is made of
ultra-high molecular weight, high-density, and nonplasticized
5.1.3 It is imperative to have an estimate of the test
polyethylene and contains an intact soil core (≥ 40 cm)
substancetoxicitytomammalsasaprecautionforoccupational
including topsoil.Amicrocosm for large plants may require an
safety. In addition, hydrolysis or photolysis rate constants
intact totally undisturbed 17-cm diameter by ≥60-cm deep test
should be known in order to determine necessary handling
system. The plastic pipe should be impermeable to water,
precautions. When a radiolabeled material is used, normal
light-weight, tough, rigid, and highly resistant to acids, bases,
laboratory techniques for radiation safety provide an ample
andbiologicaldegradation.Additionally,oneshoulduseplastic
margin of safety (13), except for chemicals in the “very highly
pipe that does not release plasticizers or other compounds that
toxic” category (rat oral LD <1 mg/kg). In this case a
may interfere with test results. At the bottom of each pipe
combinationofradiationsafetyandchemicalsafetyprocedures
containing a soil-core, a controlled-pore ceramic plate should
should be followed. For additional information on individual
be installed in direct contact with the intact soil-core; this
compounds, see Refs (14, 15, 16, and 17).
controlled-pore ceramic plate should be installed air-tight, and
5.1.4 Water solubility, soil sorption and octanol-water
contained within an appropriate end-cap (19) where leachate
partitioning, and vapor pressure largely will control the physi-
may flow by gravity for collection into a receiving flask, or
cal transport and bioavailability of a test chemical in soil.
transfer into flask accomplished by transfer at intervals using
Water-solublechemicalsarelikelytomovewithsoilwaterinto
an inert gas (19) (Fig. 1 and Fig. 2). The controlled-pore
the water films surrounding soil particles and root surfaces.
ceramic is included so that a partial-tension (30-35 kPa) may
Mostmicrobially-mediatedbiodegradationoccursinthewater-
be applied at the bottom of each microcosm to mimic field
containing microsites of soil particles. Plant uptake and bioac-
conditions, thus preventing undue buildup of water within the
cumulation is largely a function of water transfer to roots,
microcosms that otherwise would change chemical, physical,
active or passive uptake, internal partitioning (hydrophilic and
and biological properties of the microcosm for all except very
inorganic compounds) and solubility in fatty tissues. In
light-textured soils (for example, sands and loamy sands).
addition, water-soluble chemicals and their transformation
6.1.2 Six to twelve microcosms and receiving flasks are
productsmaybeleachedtogroundwater.Watersolubilityofan
typically contained within a temperature controlled chamber
organic chemical is a function of the dissociation of ionic
packed with insulation beads, to reduce drastic changes in
compounds and the polarity of nonionic compounds.
temperature profile (19, 20) (Fig. 2). Chamber dimensions are
5.1.5 Compounds with very high vapor pressures (boiling determined by the size required and space availability within
point <80 °C or vapor pressure >25 mm Hg) are not suitable
the greenhouse. Tops of chambers have aperatures to accom-
for testing in the terrestrial soil-core microcosm described in modate each microsm, so that tops of microcosms are exposed
E1197 − 12 (2021)
FIG. 1 Microcosm Structure and Materials (19)
to incident light and temperature. Each flask receiving leachate soil-core microcosm is later removed as a single unit (soil and
from an indivisual microcosm is housed in darkness within the plastic pipe) from the extraction tube and taken to the labora-
chamber, at the same controlled temperature as the microsms.
tory. For the agricultural microcosm, the topsoil is backfilled
Leachates are kept in darkness at the same temperature as the into the upper portion (for example, 20 cm) of the microcosm
microcosm to simulate field conditions, and avoid undue
tube. The extraction procedure as described here does disrupt
degradation of chemicals under investigation. and compress the soil-core to a certain extent. This should not,
however, influence the conclusions drawn from the tests
6.2 Soil Core Extraction:
because the evaluation is being performed on the difference
6.2.1 Soilcoresareextractedfromeitheranaturalgrassland
between the response of treatments versus controls rather than
ecosystem, a typical agricultural soil in the region of interest,
the absolute response.
or from the ecosystem of interest within the region. The intact
6.2.2 Detailed chemical and physical properties of the soil
system is extracted with a specially designed, steel extraction
in the test systems are to be determined using USDA nomen-
tube (7, 19, 8, 9, 20, 21, 22) (Fig. 3) and a backhoe. The steel
clature. Information such as pedologic identity, according to
extraction tube encases the polyethylene pipe to prevent the
the USDA 7th Approximation Soil Classification System,
tubefromwarpingorsplitting,orboth,underpressurescreated
percent organic matter, hydraulic characteristics, cation ex-
during extraction. Once the core is cut by the leading edge of
change capacity, bulk density, macro- and micro-nutrient
the driving tube, it is forced up to the microcosm tube. For the
agricultural microcosm, the plowed topsoil is moved aside and content, organic matter content, mineralogy, exchange
capacity, particle-size distribution, hydraulic characteristics,
saved. For the natural grassland ecosystem, the vegetation is
clipped before the core is extracted. For ecosystem and other important characteristics should be measured before
microcosms, existing vegetation may be retained, or removed and after the experiment, depending on the relative hazards of
(especially important when natural vegetation is large); veg- the test substance (see Refs (23, 24), Test Methods D422,
etation of interest may then be subsequently planted. The D2216, and D2167, and Practice D2488). The history of the
E1197 − 12 (2021)
FIG. 2 Arrangement of Microcosm and Support Apparatus within Temperature Controlled Chamber
soil, including previous crops grown, pest control, and other an appropriate depth of soil. Similarly, the method used to
management practices used, should be documented to assist in apply the test substance should approximate the way in which
the interpretation of the results. the test substance might arrive at the site in question. For
example, solid test substances might be mixed with the topsoil
6.3 Microcosm Vegetation and Harvesting:
before planting, thus mimicking the plowing of an agricultural
6.3.1 For the natural ecosystem (undisturbed grassland) test
fieldbeforeseedissown.Alternatively,itmaybedustedonthe
system, natural plant cover should be sufficiently diverse to be
surface to simulate dry deposition.
representative of plant species in the ecosystem of interest.
When the agricultural microcosm is used, a mixture of grasses
6.3.3 For an agricultural system, harvesting of plant tissues
and broad leaves (for example, legumes) should be included.
should be consistent with those practices used in a given
Two or three species of grasses or legumes that are typically
region.Plantsfromunitsareharvestedfromeachmicrocosmat
grown together as an agricultural crop in the region of interest
the end of the test period (20, 25). They are then air dried and
should be chosen. The species chosen must have compatible
then oven dried. In the range-finding test (see 7.3.1) the crop is
growth habits and be able to grow to maturity in the small
harvested four weeks after first exposure to the test substance.
surface area (for example, 83.3 cm for 10.3–cm diameter to
In the definitive test (see 7.4.1) plants may be harvested one or
227 cm for 17– cm diameter) of the microcosm. In some
two times during the 12-week growing period or at the end of
cases, it may be appropriate to select a grain crop normally
thetest.Thedefinitivetestmayneedtobeextendedbeyondthe
grown for human consumption to evaluate the uptake of the
12-week test period to accommodate plant species that take
radiolabeled test substances and their degradation products (7).
longer to reach the desired maturity (for example, seed
6.3.2 The seed application rate should duplicate standard
production).
farming practice for the region of interest in agricultural
microcosms. Seeds should be planted evenly and covered with 6.4 Microcosm Watering and Leachate Collection:
E1197 − 12 (2021)
FIG. 3 Diagram of Microcosm Extraction Tube (8)
6.4.1 Microcosms are watered as dictated by a predeter- 2). Fifteen percent more soil cores are extracted than are
mined water regime, usually established on the basis of site required for a combination of both the range finding and
history, with either purified laboratory water (for example, definitive tests. When the microcosms are leached before
distilled,reverseosmosis),orrainwaterthathasbeencollected, planting,thosewhichdonotleach,orleachtooquickly,ortake
filtered, and stored in a cooler at 4 °C ; or formulated rainwater longer than two days to produce 100 mL of leachate after the
(6, 20, 22). If comparisons are being made between micro- soil has been brought to field capacity are discarded.
cosms and field plots, then parallel watering in both units
6.5 Greenhouse and Growth Chamber Environments—
should be used. Care needs to be taken to deliver sufficient
Microcosmsinchambersarekeptinagreenhouse,orwithinan
water while preventing overwatering, which can induce fungal
environmental chamber, where temperature and light can be
disease and stress.
controlled. Temperatures in environmental chambers and
6.4.2 Microcosms are continuously leached by the partial
greenhousesaredesignedtoapproximateoutdoortemperatures
pressure exerted at the controlled-pore ceramic. Natural rain-
that occur during a typical growing season in the region of
fall amounts should be used to guide selection of the watering
interest. If the experiment is not conducted in the greenhouse
regime. Caution should be exercised to prevent overwatering,
during the normal agricultural growing season, then lights
which may drastically alter the rate of degradation,
suitable for plant growth, controlled by timing devices, should
transformation, translocation and transport of chemicals within
beusedtosimulatethephotoperiod,intensity,andspectrumfor
the microcosm.
a typical growing season in the area of interest. If the
6.4.3 Leachate is collected at regular intervals (for example,
experiment is conducted in the greenhouse during periods
everytwodays)intoflasks(previouslywashedwith0.1NHCl,
when the photoperiod of the natural light is not long enough to
rinsed with purified water, and dried). The 500-mL (alterna-
induce flowering and seed set, then supplemental lighting will
tively 1-L) collection flasks are attached to receiving end-caps
be required.
(ultra-high molecular weight, high-density, nonplasticized
polyethylene) using vinyl tubing or other tubing that is 6.6 Soil Sampling for Environmental Fate During the Test—
compatible,suchaspolyvinylchlorideorvinyltubing(seeFig. Thesoilinthemicrocosmsystemisnotdesignedtobesampled
E1197 − 12 (2021)
during the test.This would alter the leaching and movement of 7.3.1 The range-finding test should last a minimum of four
test substance in the system and make that particular micro- weeks from first exposure of the test substance to final harvest.
cosm useless for other test results. If it is necessary to take soil At the start of the test, the microcosms are dosed with a
samples during the test to determine the rate of movement of minimum of five concentrations of the test substance. Three
the test substance at intermediate time scales, then the number replicate microcosms are used for each of the four or five
of replicates will have to be increased to account for this treatmentlevelsandthecontrols,resultinginatotalof15or18
sacrificial sample. microcosms. Concentrations typically used are 0.1, 1.0, 10,
100, and even 1000 µg/g within the upper 20 cm of topsoil of
the microcosm if a realistic scenario is not known. The
7. Test Procedures
logarithmic scale for concentration in a range-finding test is
7.1 Test Purpose and Assumptions—The purpose of the
suggested by Rand (27). The bulk density (g/cm ) of the dry
terrestrial soil-core microcosm test is to determine the fate and
topsoil is used to calculate the concentrations. Depending on
ecological effects of a test substance, including its transforma-
mode of release of the test chemical, select either a single, or
tion products, within a particular natural grassland,
a multiple application, based on a reasonable exposure sce-
agricultural, or other natural ecosystem. The relationship of
nario.
fate and ecological effects data from treated versus control
7.3.2 When possible, randomly move each chamber, hold-
microcosms is assumed to be very similar to that from treated
ing one replicate of each of the four or five test concentrations
versus control field plots (7, 8, 20, 22). This assumption is
and a control, in the greenhouse each week to avoid location-
supported by the comparisons of microcosms and field results
induced effects. When such rotation is not possible, chambers
according to Refs (7, 20, 25, 26). The fate and effects from the
should include a complete random set(s) of treatments and
microcosm test should then be related to either the natural or
block effects investigated.
agricultural ecosystems that have the same combination of soil
7.3.3 The range-finding tests yield two necessary types of
type, vegetation, crop species, and environmental variables
information. These are (1) estimates of the bounds of toxicity
used during the microcosm test.
within which the 50 % response (for example, LC ) lies, and
7.2 Evaluation of Test Substance:
(2) initial estimates of variance in response. Given the identi-
7.2.1 Physicochemical information supplied for the test
fication of bounds of toxicity for the range-finding tests, the
substance (see 5.1.1) is used to tailor the general range-finding
concentrations for the definitive tests may be refined. Use the
test procedures to the specific substance. Phytotoxicity, or
variance estimates to determine sample sizes needed in the
bacteriostatic, action, or both, if known, should be taken into
definitive tests to achieve statistical tests able to detect speci-
account when designing the exposure concentrations of the
fied differences (∆) among concentrations with a specified
range-finding experiment. If the information is available, only
power (1-β).
one concentration above that known to cause at least 50 %
7.4 Definitive Test Experimental Design:
change in plant growth or 50 % change in bacterial growth/
7.4.1 Thedefinitivetestlastsfor12ormoreweeksfromfirst
respiration will need to be tested. In any case, the lowest
exposure of the test chemical to final harvest. Test results may
treatmentlevelshouldnotbelessthan10timesgreaterthanthe
be influenced by extraneous environmental sources of
analytical limits of detectability of the parent compound at the
variation, such as temperature or light gradients within a
start of the experiment.
greenhouse. These sources of variation may be accounted for
7.2.2 The water solubility and soil sorption capacity can be
by randomly repositioning the chambers, or by using random-
used to determine the appropriate frequency of leachate analy-
ized block, latin-square, or other more complex experimental
ses for the radiolabeled test substance and its transformation
designs. If such extraneous sources of variability in test results
products.This same information will also determine the design
are not taken into account, results may be biased, thus
of the soil sampling procedures for the range-finding test.
jeopardizing the outcome of the experiment. The types of
Chemical structure and any degradation information is used to
statistical analyses to be performed are decided at this point
determine which transformation products for the soil, leachate,
and are dictated largely by the experimental and treatment
and plant tissue will be analyzed.
designs. The experimental design determines the method of
7.2.3 As stated in 6.3.2, exposure should approximate a
randomization of the treatments to account for extraneous
reasonable scenario. Additionally, one must account for the
sources of variability in the experiment environments. The
water solubility, dissociation constant(s), and soil pH when
treatment design determines the number of treatments and the
determining the concentration and when selecting the specific
arrangement of treatments with respect to one another.
formulation of the chemical to apply. Solubility, however, may
7.4.2 At the start of the test, the microcosms are dosed with
be markedly altered by ionization in soil. If the soil pH is such
three concentrations of the test substance. Determine the
that a more soluble form is likely, adjust accordingly the test
number of microcosms to be dosed by the desired power of the
substance pH with either sodium hydroxide or hydrochloric
statistical tests. Power is influenced by the variance of the
acid before adding to the soil in the microcosm. If the pH
response (estimated from range-finding tests), the size of the
adjustment to increase solubility is extreme (4 < pH > 9),
difference to be detected among the treatments, and the alpha
chemical and photolytic degradation may be enhanced when
(α) level. The desired power, alpha level, and detectable
preparing the chemical solutions.
difference are specified by the researcher, and the variance
7.3 Range-finding Test: estimates are obtained from the range-finding tests. Based on
E1197 − 12 (2021)
these four values, determine the sample size, or number of and wet the plants to the point where they begin to drip. Use a
replicates for each treatment level. See Refs (28, 29) for chromatography sprayer or nebulizer used to spray plants that
discussion of power of a test. The three treatments chosen are arepasttheseedlingstage.Followtherecommendationsbythe
estimated from the range-finding test data to produce a 20 % to test substance manufacturer for field spraying as closely as
25 % change in productivity for each subsequent concentration possible, but terminate the test (last harvest) at least eight
of the test chemical. Reduce analytical costs associated with weeks after the plants are sprayed.
the fate studies by using the replicate microcosms in each
7.6 Waste Disposal:
treatment as replicate pairs. Thus, leachate and plant tissue
7.6.1 Retain all liquid (leachate) and solid (soils and plant
analyses are conducted on the pooled specimens from paired
tissues) specimens for proper disposal. Clean (acid wash) all
microcosms. However, pooling of specimens will reduce the
specimen collection bottles, collection apparatus, microcosm
power of the test and reduces the effective number of repli-
tubes, and sampling tools thoroughly and analyze for radioac-
cates. Productivity data, on the other hand, are analyzed for
tive contamination before they are stored or used on another
each individual microcosm. Each cart holds six to twelve
test system. Dispose of all samples and the remaining, undis-
microcosms (see Fig. 2). Place the microcosms paired for
turbed portion of the test system in accordance with United
analyses in different carts to ensure that all microcosms are
StatesEnvironmentalProtectionAgency(USEPA)andNuclear
housed under similar conditions.
Regulatory Commission (NRC) regulations, if radiolabeled
7.4.3 Depending on the type of natural vegetation or crop
compounds were used. Treat soil leachate and all other
planted, it may be possible to harvest more than once, such as
aqueous-sample wastes prior to disposal using one or more of
during the middle and at the end of the test. If growth is
the following techniques: (a) filtration, (b) activated charcoal
vigorous,harvestgrassesatapre-arrangedheight,forexample,
filtration, or (c) ion exchange.
2 to 6 cm above soil surface during the middle of the 12-week
7.6.2 Soils contaminated with organic residues or radiola-
test period. Multiple harvests permit evaluation of both gross
beled compounds, or both, as well as the plastic pipe, sample
plant yield and plant uptake of the test substance with respect
bottles, glassware, gloves, masks, filters, activated charcoal
to time (7, 8, 30).
from aqueous cleanup, and any other potentially contaminated
7.5 Exposure Techniques:
equipment must be either certified as uncontaminated or
7.5.1 If the primary mode of exposure of the test chemical
packaged and disposed of in accordance with existing USEPA
isanticipatedtobebyadditionofpH-adjustedlaboratorywater
and NRC guidelines and regulations.
or rainwater containing appropriate concentrations of the test
substance, then use the following exposure techniques. In no
8. Fate and Effects Sampling Procedures
case shall the total aqueous volume of a single exposure be
8.1 Sampling procedures have been divided into two basic
sufficient to cause leaching any of the microcosms. Test
categories: ecological effects sampling and test-chemical fate
substancesthatarelikelytobereleasedintotheenvironmentas
sampling. Ecological effects sampling may include productiv-
a liquid or solid, and which can be mixed with water, are
ity measurements, physical appearance of plants, and nutrient
applied as a single exposure sufficient in volume to bring the
loss or uptake measurements.Test-chemical fate sampling may
microcosmtofieldcapacity.Thevolumeoflaboratorywateror
include leachate, soil, and plant analyses.
rainwater required for exposure can be determined using an
8.1.1 Ecological Effects Sampling—Productivity Measure-
unplanted microcosm of the same soil type. The volume
ments:
selected should be the same for all microcosms. Carriers other
8.1.1.1 Primary productivity is a commonly measured pa-
than water are not recommended unless they are likely to be
released into the environment in conjunction with the test rameter in terrestrial effects testing. Depending on the plant
substance in an effluent stream. If a carrier is necessary, then species, it may be desirable to report total yield or yield by
consider acetone or ethanol. However, avoid the use of carriers plant part. For example, in the case of grain crops, such as
unless they are essential to produce a realistic exposure. Also, soybeans, oats, and wheat, the total biomass yield can be
tests for carrier effects should be required with inclusions of reported in addition to the grain yield. This will allow total
additional microcosms in the experimental design for this biomasstobecomparedwithgrainyieldstypicallyreportedfor
local agriculture. In addition, separate grain samples may be
purpose.
useful for later tissue analyses to determine whether the test
7.5.2 Several typical exposure modes are suggested for
chemical was enriched in potentially edible plant parts. For
particular types of test substances if either a hypothetical or
other systems, such as natural grassland microcosms, segrega-
real (actual) exposure scenario is not available. If the test
tion into plant parts may be unnecessary.
substance is likely to be a contaminant of irrigation water,
apply the test substance daily or weekly in proportionate 8.1.1.2 Productivity should be reported as oven-dry weight.
concentrations, such that the total amount applied equals the According to Jones and Steyn (31) 65°C for 24 h are adequate
desired concentration. If the test substance does not mix with conditions for drying without unnecessary thermal decompo-
water, apply it as evenly as possible to the top of the unplanted sition of plant material. Evaluate information on the chemical
microcosm and mixed into the topsoil prior to planting. If the volatility when selecting a drying temperature. It may be
test substance is normally sprayed on growing plants (for desirableinsomecircumstancestoreportair-driedproductivity
example, pesticide), then mix the desired amount with the ortobeabletocalculateair-driedyieldsbasedonmoistureloss
volume of solvent or water necessary to wet the soil surface after oven-drying. These data could be useful if agricultural
E1197 − 12 (2021)
crops are the plants used in the microcosm and if it is desirable sensitivity factors adequate to verify exposure and distinguish
to compare productivity with yields reported in local agricul- between parent material, transformation products, and natu-
ture. rallyoccurringmaterialspresentinthetestsystem.Usuallythis
test will involve use of a radiolabeled parent compound and
8.1.1.3 The number of harvests will depend on the types of
plants grown. An agricultural crop, alfalfa/timothy for subsequent analysis of microcosm components for radioactiv-
ityandchemicalidentity.Methodsappropriatetothelattermay
example, may require two or more harvests over the course of
the testing period (7, 32). be adequate for quantification of fate, but usually cannot reveal
bound residues in soil or plants and frequently are inadequate
8.1.2 Physical Appearance of Plants— Throughout the test
for cost-effectively tracing movement and transformation. To
period, it is desirable to record the physical appearance of
the extent that the fate in soil and plants is well enough
plants in the terrestrial microcosm. Monitor symptoms of
understood from other experiments and depending on the
nutrient deficiency or toxicity, pathogenicity, water stress, or
degree to which the microcosm test is being used to verify fate
test-chemical-induced toxicity. These observations may be
and exposure hypotheses, analytical requirements may be
useful in interpreting the specific ecological effects of a test
reduced (43, 44, 45, 46, 47). If sampling of soils is planned
chemical relative to responses in plants elicited by known
during the experiment, then increase the number of replicates
environmental toxicants or stresses (33). Careful observation
accordingly.
on physical appearance in controls versus treated microcosms
may also aid in determining whether abnormal physical ap-
8.1.4.2 Radiolabeling the Parent Compound— Label the
pearance is a result of the test chemical or is a manifestation of
parent compound with C either in an appropriate aromatic,
microcosm management.
cyclic carbon group, or in a linear chain (13, 48). Other labels,
8.1.3 Nutrient Loss Measurements: including stable isotopes such as N, may be more useful and
8.1.3.1 An important ecological effects sampling procedure informative. In order for the microcosm test to permit an
analysis of the fate of the parent compound, or its metabolites,
is to monitor nutrient losses in leachates (6, 26, 34, 35). The
rationale for such monitoring is explained in detail in Refs (8, or both, consider the known or hypothesized metabolic path-
9). One of the desirable attributes of the terrestrial microcosm ways for test substances. Hence, the location and form of label
approach to testing chemicals is the relative ease with which is an integral part of the total test design. The laboratory
soil leachates can be collected. This approach offers the
conducting the test is not required to have the capability for
potential to construct nutrient budgets for the model ecosystem radiolabeling, since this is routinely handled by specialty
(36, 37).
chemical firms. Sufficient radioactivity must be present in
order to detect at least 1 % of the initial parent compound in a
8.1.3.2 The final suite of nutrients monitored in leachates
probablywilldependonthenatureofthetestchemical (38, 35, typical sample of leachate, soil, or plant tissue.
39, 40). Consider initially those nutrients during the range-
8.1.4.3 Compartment Analysis for Labeled Compounds—
finding test that include calcium, potassium, nitrate-nitrogen,
Analyze several compartments of the terrestrial microcosm for
ortho-phosphate, ammonium-nitrogen, and dissolved organic
radioactivity.Thecomponentsincludesamplesofsoilleachate,
carbon (DOC). Depending on the results of nutrient losses
plant tissue, including roots and shoots, and soil from different
measuredduringtherange-findingtest,asetofnutrientscanbe
depths. Select different soil depths used for radiochemical
selected for monitoring during the definitive test.
analyses based on information on soil sorption of the com-
8.1.3.3 Various methods exist to analyze for nutrients.
poundofinterest.Experienceindicatesthatthesedepthsshould
Standard techniques proven useful include atomic absorption
be relatively close to the soil surface (1 to 2 cm) for
spectrophotometry for Ca and K and analysis using a Techni-
radiolabeled chemicals that are strongly sorbed to soils. If any
con Autoanalyzer II for nitrate-nitrogen, ortho-phosphate,
isotope appears in the leachate, the depth selection should be
DOC, and ammonium-nitrogen. See Method D511, Test Meth-
lower in the soil profile. Homogenize and extract specimens
ods D515, D1426, and D3867 and Refs (41, 42) for more
with solvents appropriate for the parent compound.Additional
information. For less rigorous determinations, such as during
extraction steps may be necessary. These include acidification
the range-finding test, ion-specific electrodes may be useful for
and extraction with nonpolar solvents, soxhlet extractions with
nitrate- and ammonium-nitrogen detection.
polar or non-polar solvents, or both, alkaline or acid hydrolysis
8.1.3.4 A standard procedure, described below, has proven with or without heat, detergent extractions, and protease
to be useful in handling leachates. As soon as soil water (that digestion. Oxidize and analyze as CO , according to Ref
is, leachate) samples are collected, the sample volume is (44), the C in the soil or plant samples that cannot be
recorded and the pH determined using a glass electrode. extracted or dissolved as described by Cole (46). The extracts
Samples are centrifuged at low speed (5000 r/min) to remove and the oxidized or dissolved samples should be counted by
large particles and the remaining liquid is passed through a C liquid scintillation (46, 47).
0.45-µm filter. Divide the specimen into two aliquots prior to
8.1.4.4 At the termination of the range-finding test, collect
storage in the dark at 4 °C. Prepare and s
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...