ASTM D513-16
(Test Method)Standard Test Methods for Total and Dissolved Carbon Dioxide in Water
Standard Test Methods for Total and Dissolved Carbon Dioxide in Water
SIGNIFICANCE AND USE
4.1 Carbon dioxide is a respiration product of plants and animals and a decomposition product of organic matter and certain minerals. The atmosphere averages about 0.04 vol % of CO2. Surface waters generally contain less than 10 mg/L, except at local points of abnormal organic or mineral decomposition; however, underground water, particularly deep waters, may contain several hundred mg/L.
4.2 When dissolved in water, CO2 contributes significantly to corrosion of water-handling systems. This is particularly troublesome in steam condensate systems. Loss of CO2 from an aqueous system can disturb the carbonate equilibrium and result in calcite encrustation of confining surfaces. Scaling of water heaters is a good example. Because of the delicate balance between corrosion and encrustation tendencies, much care must be given to control of CO2 and related species in water systems. Recarbonation of municipal supplies during final stages of softening and amine neutralization of steam condensate are applied for these purposes.
SCOPE
1.1 These test methods cover the measurement of total or dissolved carbon dioxide present as carbon dioxide (CO2), carbonic acid, bicarbonate ion, and carbonate ion in water:
Range
Sections
Test Method A (Gas Sensing Electrode)
2 to 800 mg/L
8 to 15
Test Method B (CO2 Evolution, Coulometric Titration)
5 to 800 mg/L
16 to 24
1.2 Carbon dioxide may also be detected from carbonates present in particulates in samples.
1.3 Test Method A is applicable to various natural waters and brines.
1.4 Test Method B is applicable to natural waters, brines, and various industrial waters as delineated in 16.4.
1.5 It is the user's responsibility to ensure the validity of these test methods on waters of untested matrices.
1.6 Several test methods were discontinued from this standard in 1988. Refer to Appendix X1 for historical information.
1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 14-Jun-2016
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.05 - Inorganic Constituents in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-Jan-2012
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 15-Dec-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D513-16 is a recognized international standard that outlines the methodologies for the measurement of total and dissolved carbon dioxide (CO₂) in water. Published by ASTM International, this standard is essential for understanding carbon dioxide content in various water matrices, such as natural waters, brines, and industrial waters. Accurate determination of CO₂ levels is vital for water quality management, corrosion control, and maintaining the carbonate equilibrium in water systems.
CO₂, present as free gas, carbonic acid, bicarbonate, and carbonate ions, is both a natural byproduct of biological activity and a result of decomposition of organic matter and minerals. The standard provides detailed test methods to measure CO₂ concentrations, ensuring consistent and reliable results for industries and public utilities concerned with water quality and system integrity.
Key Topics
- Importance of CO₂ Measurement: Monitoring CO₂ is crucial as it significantly impacts corrosion rates in water-handling systems and influences scaling and encrustation, especially in steam condensate systems and water heaters.
- Applicable Methods: The standard describes two primary analytical approaches:
- Test Method A: Gas Sensing Electrode (applicable from 2 to 800 mg/L CO₂) for natural waters and brines.
- Test Method B: CO₂ Evolution, Coulometric Titration (applicable from 5 to 800 mg/L CO₂) for a wider range including industrial waters.
- Parameters and Definitions: The standard encompasses total or dissolved CO₂, covering all relevant associated species (CO₂, carbonic acid, bicarbonate ion, and carbonate ion).
- Precision and Bias: It details guidelines for calibration, quality control, and assessment of precision and bias based on interlaboratory studies.
- Sample Collection and Handling: Emphasizes best practices for sampling, filtration, storage, and prevention of CO₂ loss during handling.
- Interferences: Describes potential analytical interferences and strategies for their mitigation to ensure the accuracy of CO₂ measurement.
Applications
The test methods detailed in ASTM D513-16 are applied across a wide range of sectors where water quality and system performance are critical:
- Municipal Water Treatment: For maintaining carbonate balance and preventing calcite scaling during water softening and distribution.
- Industrial Water and Boiler Systems: To control CO₂-induced corrosion and scaling in pipelines, storage tanks, and steam condensate systems.
- Environmental Monitoring: Assessing the impact of organic decomposition and mineral dissolution on water bodies.
- Brine and Groundwater Analysis: Providing reliable methods for analyzing elevated CO₂ in underground or high-salinity waters.
- Research and Regulatory Compliance: Supplying laboratories with standard procedures aligned with regulatory quality requirements.
Implementing ASTM D513-16 ensures that water quality data is scientifically reliable and suitable for decision-making in maintenance, compliance, and environmental management.
Related Standards
Several ASTM standards complement ASTM D513-16 in the analysis of water quality and system assessment:
- ASTM D1066 - Practice for Sampling Steam
- ASTM D1129 - Terminology Relating to Water
- ASTM D1193 - Specification for Reagent Water
- ASTM D1293 - Test Methods for pH of Water
- ASTM D2777 - Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
- ASTM D3370 - Practices for Sampling Water from Closed Conduits
- ASTM D5847 - Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
- ASTM E200 - Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis
Utilizing ASTM D513-16 in conjunction with these related standards helps ensure comprehensive, accurate, and quality-assured results for water analysis.
Keywords: dissolved carbon dioxide, total carbon dioxide, water analysis, gas sensing electrode, coulometric titration, ASTM D513, water quality, corrosion, scaling, carbonate equilibrium.
Buy Documents
ASTM D513-16 - Standard Test Methods for Total and Dissolved Carbon Dioxide in Water
REDLINE ASTM D513-16 - Standard Test Methods for Total and Dissolved Carbon Dioxide in Water
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D513-16 is a standard published by ASTM International. Its full title is "Standard Test Methods for Total and Dissolved Carbon Dioxide in Water". This standard covers: SIGNIFICANCE AND USE 4.1 Carbon dioxide is a respiration product of plants and animals and a decomposition product of organic matter and certain minerals. The atmosphere averages about 0.04 vol % of CO2. Surface waters generally contain less than 10 mg/L, except at local points of abnormal organic or mineral decomposition; however, underground water, particularly deep waters, may contain several hundred mg/L. 4.2 When dissolved in water, CO2 contributes significantly to corrosion of water-handling systems. This is particularly troublesome in steam condensate systems. Loss of CO2 from an aqueous system can disturb the carbonate equilibrium and result in calcite encrustation of confining surfaces. Scaling of water heaters is a good example. Because of the delicate balance between corrosion and encrustation tendencies, much care must be given to control of CO2 and related species in water systems. Recarbonation of municipal supplies during final stages of softening and amine neutralization of steam condensate are applied for these purposes. SCOPE 1.1 These test methods cover the measurement of total or dissolved carbon dioxide present as carbon dioxide (CO2), carbonic acid, bicarbonate ion, and carbonate ion in water: Range Sections Test Method A (Gas Sensing Electrode) 2 to 800 mg/L 8 to 15 Test Method B (CO2 Evolution, Coulometric Titration) 5 to 800 mg/L 16 to 24 1.2 Carbon dioxide may also be detected from carbonates present in particulates in samples. 1.3 Test Method A is applicable to various natural waters and brines. 1.4 Test Method B is applicable to natural waters, brines, and various industrial waters as delineated in 16.4. 1.5 It is the user's responsibility to ensure the validity of these test methods on waters of untested matrices. 1.6 Several test methods were discontinued from this standard in 1988. Refer to Appendix X1 for historical information. 1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 4.1 Carbon dioxide is a respiration product of plants and animals and a decomposition product of organic matter and certain minerals. The atmosphere averages about 0.04 vol % of CO2. Surface waters generally contain less than 10 mg/L, except at local points of abnormal organic or mineral decomposition; however, underground water, particularly deep waters, may contain several hundred mg/L. 4.2 When dissolved in water, CO2 contributes significantly to corrosion of water-handling systems. This is particularly troublesome in steam condensate systems. Loss of CO2 from an aqueous system can disturb the carbonate equilibrium and result in calcite encrustation of confining surfaces. Scaling of water heaters is a good example. Because of the delicate balance between corrosion and encrustation tendencies, much care must be given to control of CO2 and related species in water systems. Recarbonation of municipal supplies during final stages of softening and amine neutralization of steam condensate are applied for these purposes. SCOPE 1.1 These test methods cover the measurement of total or dissolved carbon dioxide present as carbon dioxide (CO2), carbonic acid, bicarbonate ion, and carbonate ion in water: Range Sections Test Method A (Gas Sensing Electrode) 2 to 800 mg/L 8 to 15 Test Method B (CO2 Evolution, Coulometric Titration) 5 to 800 mg/L 16 to 24 1.2 Carbon dioxide may also be detected from carbonates present in particulates in samples. 1.3 Test Method A is applicable to various natural waters and brines. 1.4 Test Method B is applicable to natural waters, brines, and various industrial waters as delineated in 16.4. 1.5 It is the user's responsibility to ensure the validity of these test methods on waters of untested matrices. 1.6 Several test methods were discontinued from this standard in 1988. Refer to Appendix X1 for historical information. 1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM D513-16 is classified under the following ICS (International Classification for Standards) categories: 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D513-16 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D1066-18, ASTM D1066-18e1, ASTM D2777-12, ASTM D1293-12, ASTM D1066-11, ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1066-06, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D513-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D513 − 16
Standard Test Methods for
Total and Dissolved Carbon Dioxide in Water
This standard is issued under the fixed designation D513; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
1.1 These test methods cover the measurement of total or
D1293 Test Methods for pH of Water
dissolved carbon dioxide present as carbon dioxide (CO ),
D2777 Practice for Determination of Precision and Bias of
carbonic acid, bicarbonate ion, and carbonate ion in water:
Applicable Test Methods of Committee D19 on Water
Range Sections
D3370 Practices for Sampling Water from Closed Conduits
Test Method A (Gas Sensing Electrode) 2 to 800 mg/L 8 to 15
Test Method B (CO Evolution, Coulometric 5 to 800 mg/L 16 to 24 D5847 Practice for Writing Quality Control Specifications
Titration)
for Standard Test Methods for Water Analysis
1.2 Carbon dioxide may also be detected from carbonates
E200 Practice for Preparation, Standardization, and Storage
present in particulates in samples. of Standard and Reagent Solutions for ChemicalAnalysis
1.3 Test Method A is applicable to various natural waters
3. Terminology
and brines.
3.1 Definitions:
1.4 Test Method B is applicable to natural waters, brines,
3.1.1 For definitions of terms used in this standard, refer to
and various industrial waters as delineated in 16.4.
Terminology D1129.
1.5 It is the user’s responsibility to ensure the validity of
these test methods on waters of untested matrices. 4. Significance and Use
1.6 Several test methods were discontinued from this stan- 4.1 Carbon dioxide is a respiration product of plants and
dard in 1988. Refer to Appendix X1 for historical information.
animals and a decomposition product of organic matter and
certain minerals.The atmosphere averages about 0.04 vol % of
1.7 The values stated in SI units are to be regarded as
CO . Surface waters generally contain less than 10 mg/L,
standard. No other units of measurement are included in this
except at local points of abnormal organic or mineral decom-
standard.
position; however, underground water, particularly deep
1.8 This standard does not purport to address all of the
waters, may contain several hundred mg/L.
safety concerns, if any, associated with its use. It is the
4.2 When dissolved in water, CO contributes significantly
responsibility of the user of this standard to establish appro- 2
to corrosion of water-handling systems. This is particularly
priate safety and health practices and determine the applica-
troublesome in steam condensate systems. Loss of CO from
bility of regulatory limitations prior to use. 2
an aqueous system can disturb the carbonate equilibrium and
2. Referenced Documents
result in calcite encrustation of confining surfaces. Scaling of
water heaters is a good example. Because of the delicate
2.1 ASTM Standards:
balance between corrosion and encrustation tendencies, much
D1066 Practice for Sampling Steam
care must be given to control of CO and related species in
water systems. Recarbonation of municipal supplies during
These test methods are under the jurisdiction of ASTM Committee D19 on
Water and are the direct responsibility of Subcommittee D19.05 on Inorganic
final stages of softening and amine neutralization of steam
Constituents in Water.
condensate are applied for these purposes.
Current edition approved June 15, 2016. Published June 2016. Originally
ɛ1
approved in 1938. Last previous edition approved in 2011 as D513 – 11 . DOI:
5. Purity of Reagents
10.1520/D0513-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
5.1 Reagent grade chemicals shall be used in all tests.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Unless otherwise indicated, it is intended that all reagents shall
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. conform to the specifications of the Committee on Analytical
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D513 − 16
Reagents of the American Chemical Society. Other grades 8.2 Samples containing 2 to 800 mg/L total CO can be
may be used, provided it is first ascertained that the reagent is analyzed by this test method. The concentration range may be
of sufficiently high purity to permit its use without lessening extended by dilution of an appropriate aliquot.
the accuracy of the determination.
8.3 Samples should be analyzed immediately. If this is not
5.2 Unless otherwise indicated, references to water shall be possible, preserve by making them slightly alkaline (pH
understood to mean water conforming to Type I of Specifica-
between 8 and 9) using carbonate-free NaOH solution and
tion D1193. Other reagent water types may be used provided it store them in a tightly capped vessel. The latter step prevents
isfirstascertainedthatthewaterisofsufficientlyhighpurityto
absorption of CO from the air.
permititsusewithoutadverselyaffectingthebiasandprecision
8.4 The precision and bias were obtained on reagent water
of the test method. Additionally, for those test methods
and a water matrix of choice that included natural waters and
requiring water free of CO , refer to 8.2 of Practice E200.
brines. It is the responsibility of the analyst to determine the
acceptability of this test method for the water being analyzed.
6. Precautions
6.1 Warning—Carbon dioxide is easily lost from solution
9. Summary of Test Method
during transit and storage of samples. It is also possible for
9.1 Carbon dioxide is liberated by acidification of the
total CO concentration to increase after sampling due to
sample to pH 5.0. The carbon dioxide electrode uses a
solution of finely divided calcium carbonate as a result of
gas-permeable membrane to separate the sample solution from
temperature or pressure changes.
the electrode internal solution. Dissolved carbon dioxide in the
sample solution diffuses through the membrane until an equi-
7. Sampling
librium is reached between the partial pressure of CO in the
7.1 Collect the sample in accordance with Practice D1066
sample solution and the CO in the internal filling solution. In
and Practices D3370, as applicable.
any given sample, the partial pressure of CO will be propor-
tional to the concentration of CO .The diffusion of CO across
2 2
7.2 Filter samples when they are collected if particulates are
the membrane affects the concentration of hydrogen ions in the
present that may contain carbonates if dissolved species only
internal filling solution:
are to be determined. When aliquots of sample are taken from
sample bottles containing particulates, the bottle must be
→
1 2
CO 1H O H 1HCO
2 2 3
shaken or otherwise homogenized to ensure a representative
←
sample is taken. When particulates form in samples due to
9.2 The hydrogen ion concentration of the internal solution
changes in temperature, pH, etc., after the sample has been
is measured by the pH electrode located behind the membrane.
collected, these particulates must be included in tests of the
Since the hydrogen ion concentration is directly related to CO
sample. Care must be used to avoid loss of CO during any
concentration, the electrode response is Nernstian.
homogenization of filtration of samples. Do not filter samples
unless it is required to remove potentially interfering particu-
9.3 Samples are treated prior to measurement with a buffer
lates. solution that sets the pH between 4.8 and 5.2. At this pH,
interferences are minimized and the various ionic forms are
7.3 Use a hard, glass, chemically resistant bottle for collect-
converted to CO (see Section 10).
ing the sample.
7.4 Fill the sample bottle completely, with no air space
10. Interferences
remaining beneath the cap, and store the sample at a tempera-
10.1 Volatile weak acids are potential positive electrode
turebelowthatatwhichitwascollecteduntilthedetermination
interferences. Concentrations of these interfering species that
is made.
cause a 10 % error at 44 mg/L CO or 100 mg/L (as CaCO )
2 3
TEST METHOD A—GAS SENSING ELECTRODE
and at pH 4 and 5, are listed below:
TEST METHOD
Interferences, mg/L pH 5 pH 4
HS10 7
−
NO (NO ) 161 24
2 2
8. Scope
−
HSO (SO ) 320 (as SO ) 48 (as SO )
3 2 2 2
HOAc (acetic acid) 372 216
8.1 This test method determines total or dissolved carbon
HCOOH (formic acid) 1841 345
dioxide (14.3) present as CO , carbonic acid, bicarbonate ion,
10.2 Samples containing sulfide can be treated with dilute
and carbonate ion in water, within the interference constraints
solutions of potassium dichromate (or the like), since sulfur is
specified.
not an interference for this test method. However, it is possible
that some organic material could be oxidized to CO by this
treatment,resultinginfalselyhighresults.Thesuitabilityofthe
Reagent Chemicals, American Chemical Society Specifications , American
test method for samples containing sulfide should be deter-
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
mined individually.
listed by the American Chemical Society, see Analar Standards for Laboratory
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
10.3 Water vapor is a potential electrode interference.Water
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
MD. can move across the membrane as water vapor, changing the
D513 − 16
concentration of the internal filling solution under the mem- 13.2 Dilute 10 mL of the buffer solution to 100 mL with
brane. Such changes will be seen as electrode drift. Water water using a volumetric flask. Transfer the contents of the
vaportransportisnotaproblemif(1)thetotalconcentrationof flask to a 150-mL beaker and add a stirring bar. Immerse the
dissolvedspeciesinsolution(Note1)isapproximatelyequalto electrode in the solution. Stir at a slow rate using the magnetic
that of the internal filling solution, and (2) electrode and stirrer.
sample temperatures are the same.
13.3 Using a volumetric pipette, add 0.5 mL of the 0.01 M
NaHCO standardsolutionandmixslowly.Allowthepotential
NOTE 1—The osmotic strength of a solution is related to the total
concentration of dissolved species in the solution. For example, the
reading to stabilize (approximately 10 min) and record the
osmotic strength of a solution containing 0.1 M hydrochloric acid, 0.1 M
potential (corresponds to 2.2 mg/L CO or 5.0 mg/L (as
aceticacid,and0.1 Msucroseisdeterminedasfollows:Hydrochloricacid
CaCO )).
dissociates to give 0.1 M hydrogen ion and 0.1 M chloride ion. The acetic
acid, because of the concentration of free hydrogen ion, is essentially
13.4 Using a volumetric pipette, add 0.5 mL of the 0.01 M
undissociated;thusgiving0.1Mofspecies.Likewise,theconcentrationof
NaHCO standardsolutionandmixslowly.Allowthepotential
sucrose species is 0.1 M . Therefore, the total osmotic strength is 0.4
reading to stabilize (approximately 5 min) and record the
osmolar.
potential (corresponds to 4.4 mg/L CO or 10.0 mg/L (as
10.4 Addition of carbon dioxide buffer (12.1) to samples of
CaCO )).
low osmotic strength automatically adjusts them to the correct
13.5 Using a volumetric pipette, add 0.9 mL of the 0.1 M
level. Samples with osmotic strength greater than approxi-
NaHCO standardsolutionandmixslowly.Allowthepotential
mately 1 M should be diluted before measurement to avoid
reading to stabilize (approximately 2 min) and record the
drifting associated with water vapor transport. Dilution should
potential (corresponds to 43.2 mg/L CO or 98.1 mg/L (as
not reduce the carbon dioxide level below 2.5 mg/L. Samples
CaCO )).
with osmotic strengths above 1 M that cannot be diluted can be 3
measured by adjusting the osmotic strength of the internal
13.6 Using a volumetric pipette, add 10 mL of the 0.1 M
filling solution. To adjust the total concentration of dissolved
NaHCO standardsolutionandmixslowly.Allowthepotential
species in the internal filling solution, add 0.425 g of reagent-
reading to stabilize (approximately 2 min) and record the
grade NaNO to 10 mL of internal filling solution.
potential (corresponds to 433 mg/L CO or 983 mg/L (as
CaCO )).
11. Apparatus
13.7 Follow manufacturer instructions for calibrating
11.1 pH Meter, with expanded mV scale, or a selective ion
selective-ion meters with a direct reading of concentration
meter.
capabilities. For pH meters, generate a calibration curve by
11.2 CO Gas-Sensing Electrode.
2 creating a calibration curve by plotting potential values (on the
linear scale) versus concentration (on the logarithmic scale) on
11.3 Mixer, magnetic withTFE-fluorocarbon-coated stirring
semilogarithmic graph paper to obtain a calibration curve. The
bar or equivalent.
curve may be extended down to 2 mg/L and up to 800 mg/L
12. Reagents and Materials
CO . Commercially available meters may be used.
12.1 Buffer Solution—Dissolve 294 g of sodium citrate in
14. Procedure
approximately 700 mL of water in a 1-L volumetric flask.
AcidifythesolutiontopH4.5withconcentratedHCl(approxi- 14.1 Bring samples to the same temperature as the electrode
mately 90 mL) and dilute to the mark with water.
and standards.
12.2 Sodium Bicarbonate Solution, Standard (0.1 M)— 14.2 Place a known volume, V , (100 mL is convenient) of
s
Dissolve 8.40 g of sodium bicarbonate in water and dilute to
sample in 150-mL beaker and stir slowly. Immerse the elec-
1L. trode in the solution.
12.3 Sodium Bicarbonate Solution, Standard (0.01 M)— 14.3 Add 1 mL of buffer, V , for each 10 mL of sample.
b
Dilute 10.0 mL of sodium bicarbonate standard solution (0.1 Allow the potential reading to stabilize and record the value.
M) to 100 mL. Read the concentration measured, C , directly from the cali-
m
bration curve.
12.4 Filter Paper—Purchase suitable filter paper. Typically
the filter papers have a pore size of 0.45-µm membrane. 14.4 Determine the sample concentration, C , as follows:
s
Material such as fine-textured, acid-washed, ashless paper, or
V 1V
s b
C 5 C
glass fiber paper are acceptable. The user must first ascertain
s m
V
s
that the filter paper is of sufficient purity to use without
adversely affecting the bias and precision of the test method.
15. Precision and Bias
15.1 Precision—Theoverallandsingleoperatorprecisionof
13. Calibration and Standardization
this test method, within its designated range, varies with the
13.1 Assemble and check the electrode in accordance with
the manufacturer’s instructions.
Supporting data have been filed at ASTM International Headquarters and may
There are currently several manufacturers of the gas-sensing electrodes for beobtainedbyrequestingResearchReportRR:D19-1069.ContactASTMCustomer
carbon dioxide. Service at service@astm.org.
D513 − 16
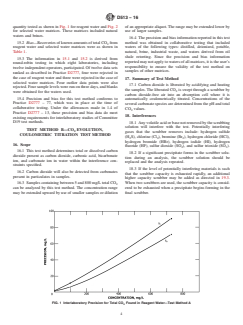
quantity tested as shown in Fig. 1 for reagent water and Fig. 2 of an appropriate aliquot.The range may be extended lower by
for selected water matrices. These matrices included natural use of larger samples.
waters and brines.
16.4 The precision and bias information reported in this test
15.2 Bias—RecoveriesofknownamountsoftotalCO from method was obtained in collaborative testing that included
reagent water and selected water matrices were as shown in waters of the following types: distilled, deionized, potable,
Table 1. natural, brine, industrial waste, and waters derived from oil
shale retorting. Since the precision and bias information
15.3 The information in 15.1 and 15.2 is derived from
reported may not apply to waters of all matrices, it is the user’s
round-robin testing in which eight laboratories, including
responsibility to ensure the validity of the test method on
twelve independent operators, participated. Of twelve data sets
samples of other matrices.
ranked as described in Practice D2777, four were rejected in
the case of reagent water and three were rejected in the case of
17. Summary of Test Method
selected water matrices. Four outlier data points were also
17.1 Carbon dioxide is liberated by acidifying and heating
rejected.Foursamplelevelswererunonthreedays,andblanks
the samples. The l
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
´1
Designation: D513 − 11 D513 − 16
Standard Test Methods for
Total and Dissolved Carbon Dioxide in Water
This standard is issued under the fixed designation D513; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—This test method was changed editorially in February 2012.
1. Scope*
1.1 These test methods cover the measurement of total or dissolved carbon dioxide present as carbon dioxide (CO ), carbonic
acid, bicarbonate ion, and carbonate ion in water:
Range Sections
Test Method A (Gas Sensing Electrode) 2 to 800 mg/L 8 to 15
Test Method B (CO Evolution, Coulometric Titration) 5 to 800 mg/L 16 to 24
Range Sections
Test Method A (Gas Sensing Electrode) 2 to 800 mg/L 8 to 15
Test Method B (CO Evolution, Coulometric 5 to 800 mg/L 16 to 24
Titration)
1.2 Carbon dioxide may also be detected from carbonates present in particulates in samples.
1.3 Test Method A is applicable to various natural waters and brines.
1.4 Test Method B is applicable to natural waters, brines, and various industrial waters as delineated in 16.4.
1.5 It is the user’s responsibility to ensure the validity of these test methods on waters of untested matrices.
1.6 Several test methods were discontinued from this standard in 1988. Refer to Appendix X1 for historical information.
1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
D1066 Practice for Sampling Steam
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D1293 Test Methods for pH of Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
E200 Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis
3. Terminology
3.1 DefinitionsDefinitions:—For
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129. definitions of terms used in these test methods,
refer to Terminology D1129.
These test methods are under the jurisdiction of ASTM Committee D19 on Water and are the direct responsibility of Subcommittee D19.05 on Inorganic Constituents
in Water.
Current edition approved April 1, 2011June 15, 2016. Published April 2011June 2016. Originally approved in 1938. Last previous edition approved in 20062011 as
ɛ1
D513 – 06.D513 – 11 . DOI: 10.1520/D0513-11E01.10.1520/D0513-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D513 − 16
4. Significance and Use
4.1 Carbon dioxide is a major respiration product of plants and animals and a decomposition product of organic matter and
certain minerals. The atmosphere averages about 0.04 vol % of CO . Surface waters generally contain less than 10 mg/L, except
at local points of abnormal organic or mineral decomposition; however, underground water, particularly deep waters, may contain
several hundred mg/L.
4.2 When dissolved in water, CO contributes significantly to corrosion of water-handling systems. This is particularly
troublesome in steam condensate systems. Loss of CO from an aqueous system can disturb the carbonate equilibrium and result
in calcite encrustation of confining surfaces. Scaling of water heaters is a good example. Because of the delicate balance between
corrosion and encrustation tendencies, much care must be given to control of CO and related species in water systems.
Recarbonation of municipal supplies during final stages of softening and amine neutralization of steam condensate are applied for
these purposes.
5. Purity of Reagents
5.1 Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all reagents shall conform
to the specifications of the Committee on Analytical Reagents of the American Chemical Society. Other grades may be used,
provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy of the
determination.
5.2 Unless otherwise indicated, references to water shall be understood to mean water conforming to Type I of Specification
D1193. Other reagent water types may be used provided it is first ascertained that the water is of sufficiently high purity to permit
its use without adversely affecting the bias and precision of the test method. Additionally, for those test methods requiring water
free of CO , refer to 8.2 of Practice E200.
6. Precautions
6.1 Warning—Carbon dioxide is easily lost from solution during transit and storage of samples. It is also possible for total CO
concentration to increase after sampling due to solution of finely divided calcium carbonate as a result of temperature or pressure
changes.
7. Sampling
7.1 Collect the sample in accordance with PracticesPractice D1066 and Practices D3370, as applicable.
7.2 Filter samples when they are collected if particulates are present that may contain carbonates if dissolved species only are
to be determined. When aliquots of sample are taken from sample bottles containing particulates, the bottle must be shaken or
otherwise homogenized to ensure a representative sample is taken. When particulates form in samples due to changes in
temperature, pH, etc., after the sample has been collected, these particulates must be included in tests of the sample. Care must
be used to avoid loss of CO during any homogenization of filtration of samples. Do not filter samples unless it is required to
remove potentially interfering particulates.
7.3 Use a hard, glass, chemically resistant bottle for collecting the sample.
7.4 Fill the sample bottle completely, with no air space remaining beneath the cap, and store the sample at a temperature below
that at which it was collected until the determination is made.
TEST METHOD A—GAS SENSING ELECTRODE SENSING ELECTRODE TEST METHOD
8. Scope
8.1 This test method determines total or dissolved carbon dioxide (14.3) present as CO , carbonic acid, bicarbonate ion, and
carbonate ion in water, within the interference constraints specified.
8.2 Samples containing 2 to 800 mg/L total CO can be analyzed by this test method. The concentration range may be extended
by dilution of an appropriate aliquot.
8.3 Samples should be analyzed immediately. If this is not possible, preserve by making them slightly alkaline (pH between 8
and 9) using carbonate-free NaOH solution and store them in a tightly capped vessel. The latter step prevents absorption of CO
from the air.
8.4 The precision and bias were obtained on reagent water and a water matrix of choice that included natural waters and brines.
It is the responsibility of the analyst to determine the acceptability of this test method for the water being analyzed.
Reagent Chemicals, American Chemical Society Specifications , American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D513 − 16
9. Summary of Test Method
9.1 Carbon dioxide is liberated by acidification of the sample to pH 5.0. The carbon dioxide electrode uses a gas-permeable
membrane to separate the sample solution from the electrode internal solution. Dissolved carbon dioxide in the sample solution
diffuses through the membrane until an equilibrium is reached between the partial pressure of CO in the sample solution and the
CO in the internal filling solution. In any given sample, the partial pressure of CO will be proportional to the concentration of
2 2
CO . The diffusion of CO across the membrane affects the concentration of hydrogen ions in the internal filling solution:
2 2
→
1 2
CO 1H O H 1HCO
2 2 3
←
9.2 The hydrogen ion concentration of the internal solution is measured by the pH electrode located behind the membrane. Since
the hydrogen ion concentration is directly related to CO concentration, the electrode response is Nernstian.
9.3 Samples are treated prior to measurement with a buffer solution that sets the pH between 4.8 and 5.2. At this pH,
interferences are minimized and the various ionic forms are converted to CO (see Section 10).
10. Interferences
10.1 Volatile weak acids are potential positive electrode interferences. Concentrations of these interfering species that cause a
10 % error at 44 mg/L CO or 100 mg/L (as CaCO ) and at pH 4 and 5, are listed below:
2 3
Interferences, mg/L pH 5 pH 4
H S 10 7
−
NO (NO ) 161 24
2 2
−
HSO (SO ) 320 (as SO ) 48 (as SO )
3 2 2 2
HOAc (acetic acid) 372 216
HCOOH (formic acid) 1841 345
10.2 Samples containing sulfide can be treated with dilute solutions of potassium dichromate (or the like), since sulfur is not
an interference for this test method. However, it is possible that some organic material could be oxidized to CO by this treatment,
resulting in falsely high results. The suitability of the test method for samples containing sulfide should be determined individually.
10.3 Water vapor is a potential electrode interference. Water can move across the membrane as water vapor, changing the
concentration of the internal filling solution under the membrane. Such changes will be seen as electrode drift. Water vapor
transport is not a problem if (1) the total concentration of dissolved species in solution (Note 1) is approximately equal to that of
the internal filling solution, and (2) electrode and sample temperatures are the same.
NOTE 1—The osmotic strength of a solution is related to the total concentration of dissolved species in the solution. For example, the osmotic strength
of a solution containing 0.1 M hydrochloric acid, 0.1 M acetic acid, and 0.1 M sucrose is determined as follows: Hydrochloric acid dissociates to give
0.1 M hydrogen ion and 0.1 M chloride ion. The acetic acid, because of the concentration of free hydrogen ion, is essentially undissociated; thus giving
0.1 M of species. Likewise, the concentration of sucrose species is 0.1 M . Therefore, the total osmotic strength is 0.4 osmolar.
10.4 Addition of carbon dioxide buffer (12.1) to samples of low osmotic strength automatically adjusts them to the correct level.
Samples with osmotic strength greater than approximately 1 M should be diluted before measurement to avoid drifting associated
with water vapor transport. Dilution should not reduce the carbon dioxide level below 2.5 mg/L. Samples with osmotic strengths
above 1 M that cannot be diluted can be measured by adjusting the osmotic strength of the internal filling solution. To adjust the
total concentration of dissolved species in the internal filling solution, add 0.425 g of reagent-grade NaNO to 10 mL of internal
filling solution.
11. Apparatus
11.1 pH Meter, with expanded mV scale, or a selective ion meter.
11.2 CO Gas-Sensing Electrode.
11.3 Mixer, magnetic with TFE-fluorocarbon-coated stirring bar or equivalent.
12. Reagents and Materials
12.1 Buffer Solution—Dissolve 294 g of sodium citrate in approximately 700 mL of water in a 1-L volumetric flask. Acidify the
solution to pH 4.5 with concentrated HCl (approximately 90 mL) and dilute to the mark with water.
12.2 Sodium Bicarbonate Solution, Standard (0.1 M)—Dissolve 8.40 g of sodium bicarbonate in water and dilute to 1 L.
12.3 Sodium Bicarbonate Solution, Standard (0.01 M)—
Dilute 10.0 mL of sodium bicarbonate standard solution (0.1 M) to 100 mL.
12.4 Filter Paper—Purchase suitable filter paper. Typically the filter papers have a pore size of 0.45-μm membrane. Material
such as fine-textured, acid-washed, ashless paper, or glass fiber paper are acceptable. The user must first ascertain that the filter
paper is of sufficient purity to use without adversely affecting the bias and precision of the test method.
There are currently several manufacturers of the gas-sensing electrodes for carbon dioxide.
D513 − 16
13. Calibration and Standardization
13.1 Assemble and check the electrode in accordance with the manufacturer’s instructions.
13.2 Dilute 10 mL of the buffer solution to 100 mL with water using a volumetric flask. Transfer the contents of the flask to
a 150-mL beaker and add a stirring bar. Immerse the electrode in the solution. Stir at a slow rate using the magnetic stirrer.
13.3 Using a volumetric pipette, add 0.5 mL of the 0.01 M NaHCO standard solution and mix slowly. Allow the potential
reading to stabilize (approximately 10 min) and record the potential (corresponds to 2.2 mg/L CO or 5.0 mg/L (as CaCO )).
2 3
13.4 Using a volumetric pipette, add 0.5 mL of the 0.01 M NaHCO standard solution and mix slowly. Allow the potential
reading to stabilize (approximately 5 min) and record the potential (corresponds to 4.4 mg/L CO or 10.0 mg/L (as CaCO )).
2 3
13.5 Using a volumetric pipette, add 0.9 mL of the 0.1 M NaHCO standard solution and mix slowly. Allow the potential reading
to stabilize (approximately 2 min) and record the potential (corresponds to 43.2 mg/L CO or 98.1 mg/L (as CaCO )).
2 3
13.6 Using a volumetric pipette, add 10 mL of the 0.1 M NaHCO standard solution and mix slowly. Allow the potential reading
to stabilize (approximately 2 min) and record the potential (corresponds to 433 mg/L CO or 983 mg/L (as CaCO )).
2 3
13.7 Plot Follow manufacturer instructions for calibrating selective-ion meters with a direct reading of concentration
capabilities. For pH meters, generate a calibration curve by creating a calibration curve by plotting potential values (on the linear
scale) versus concentration (on the logarithmic scale) on semilogarithmic graph paper to obtain a calibration curve. The curve may
be extended down to 2 mg/L and up to 800 mg/L CO . Commercially available meters may be used.
14. Procedure
14.1 Bring samples to the same temperature as the electrode and standards.
14.2 Place a known volume, V , (100 mL is convenient) of sample in 150-mL beaker and stir slowly. Immerse the electrode in
s
the solution.
14.3 Add 1 mL of buffer, V , for each 10 mL of sample. Allow the potential reading to stabilize and record the value. Read the
b
concentration measured, C , directly from the calibration curve.
m
14.4 Determine the sample concentration, C , as follows:
s
V 1V
s b
C 5 C
s m
V
s
15. Precision and Bias
15.1 Precision—The overall and single operator precision of this test method, within its designated range, varies with the
quantity tested as shown in Fig. 1 for reagent water and Fig. 2 for selected water matrices. These matrices included natural waters
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:XXX-XXX RR:D19-1069. Contact ASTM
Customer Service at service@astm.org.
FIG. 1 Interlaboratory Precision for Total CO Found in Reagent Water—Test Method A
D513 − 16
FIG. 2 Interlaboratory Precision for Total CO Found in Selected Water Matrices—Test Method A
and brines.
15.2 Bias—Recoveries of known amounts of total CO from reagent water and selected water matrices were as shown in Table
1.
15.3 The information in 15.1 and 15.2 is derived from round-robin testing in which eight laboratories, including twelve
independent operators, participated. Of twelve data sets ranked as described in Practice D2777, four were rejected in the case of
reagent water and three were rejected in the case of selected water matrices. Four outlier data points were also rejected. Four
sample levels were run on three days, and blanks were obtained for the waters used.
15.4 Precision and bias for this test method conforms to Practice D2777 – 77, which was in place at the time of collaborative
testing. Under the allowances made in 1.4 of Practice D2777 – 08,13, these precision and bias data do meet existing requirements
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...