ASTM D7359-23
(Test Method)Standard Test Method for Total Fluorine, Chlorine and Sulfur in Aromatic Hydrocarbons and Their Mixtures by Oxidative Pyrohydrolytic Combustion followed by Ion Chromatography Detection (Combustion Ion Chromatography-CIC)
Standard Test Method for Total Fluorine, Chlorine and Sulfur in Aromatic Hydrocarbons and Their Mixtures by Oxidative Pyrohydrolytic Combustion followed by Ion Chromatography Detection (Combustion Ion Chromatography-CIC)
SIGNIFICANCE AND USE
5.1 The total fluorine, chlorine, and sulfur contained in aromatic hydrocarbon matrices can contribute to emissions, be harmful to many catalytic chemical processes, and lead to corrosion. This test method can be used to determine total sulfur and halogens in aromatic hydrocarbons and their mixtures. The results can be used for compliance determinations when acceptable to a regulatory authority using performance based criteria.
SCOPE
1.1 This test method covers the individual determination of total fluorine, chlorine, and sulfur in aromatic hydrocarbons and their mixtures. Samples containing 0.10 mg/kg to 10 mg/kg of each element can be analyzed.
1.2 This method can be applied to sample concentrations outside the range of the scope by dilution of the sample in an appropriate solvent to bring the total concentrations of fluorine, chlorine, and sulfur within the range covered by the test method. However, it is the responsibility of the analyst to verify the solubility of the sample in the solvent and that the diluted sample results conform to the precision and accuracy of the method.
1.2.1 Special considerations must be made in order to attain detection limits below 1.0 mg/kg in a sample. The instrument must be clean and properly maintained to address potential sources of contamination, or carryover, or both. Multiple sequential injections shall be completed until a stable background is attained. A stable background is considered to be achieved when the analysis of a minimum of three consecutive system blanks have area counts equal to or less than 5 % RSD for the anions of interest.
1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2023
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.04 - Instrumental Analysis
Relations
- Effective Date
- 01-Jul-2019
- Effective Date
- 01-Nov-2017
- Effective Date
- 01-Nov-2016
- Effective Date
- 01-May-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Jan-2011
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Apr-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
Overview
ASTM D7359-23 is the standard test method for determining total fluorine, chlorine, and sulfur in aromatic hydrocarbons and their mixtures. Using oxidative pyrohydrolytic combustion followed by ion chromatography detection - a process also known as Combustion Ion Chromatography (CIC) - this method enables laboratories and industries to measure trace levels of these elements. These measurements are vital, as fluorine, chlorine, and sulfur can contribute to environmental emissions, damage catalytic processes, and promote corrosion in industrial systems. The ASTM D7359-23 standard is internationally recognized and developed according to global principles for standardization.
Key Topics
Key Focus Areas of ASTM D7359-23:
- Detection Range: Analyzes total fluorine, chlorine, and sulfur from 0.10 mg/kg to 10 mg/kg in samples.
- Applicability: Can be applied to concentrations outside this range via suitable sample dilution, provided solubility and analytical precision are confirmed.
- Technique: Involves oxidative pyrohydrolytic combustion at high temperatures (over 900°C), converting sample components to gaseous by-products, which are then absorbed and analyzed using ion chromatography.
- Calibration & Validation: Requires rigorous calibration procedures, stable system baselines, and standardization using certified reference materials.
- Interferences: Recognizes potential interferences from co-eluting anions, highlighting the need for thorough system cleanliness and appropriate solvent selection.
- Precision and Accuracy: Provides guidelines for repeatability and reproducibility, ensuring high-quality data across laboratories.
- Health & Safety: Emphasizes safety when handling hazardous substances and high temperatures; compliance with regulatory and best-practice guidelines is required.
Applications
Practical Uses of ASTM D7359-23:
- Quality Control in Manufacturing: Essential for chemical, petrochemical, and fuel manufacturers to verify and control halogen and sulfur content in products, thereby ensuring compatibility with sensitive catalytic processes and reducing corrosion risks.
- Regulatory Compliance: Supports compliance with environmental and product specifications by enabling trace component analysis, which can be reported to regulatory authorities as evidence of standards conformance.
- Research and Development: Useful in R&D labs for characterizing new aromatic hydrocarbon formulations and studying the effects of trace elements on chemical reactions.
- Process Optimization: Helps refineries and chemical plants monitor impurity levels, optimize production processes, and minimize maintenance by early detection of corrosive or catalyst-poisoning elements.
- Environmental Monitoring: Assists in emission studies and environmental risk assessments by providing accurate quantification of potentially harmful elements in hydrocarbon streams.
Related Standards
Reference to Additional Standards:
- ASTM D1193 - Specification for Reagent Water, ensuring water quality for analytical use.
- ASTM D3437 - Practice for Sampling and Handling Liquid Cyclic Products, recommended for proper sample collection.
- ASTM D3505 - Test Method for Density or Relative Density of Pure Liquid Chemicals.
- ASTM D6809 - Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons.
- ASTM E29 - Practice for Using Significant Digits in Test Data.
- ASTM E177 - Practice for Use of the Terms Precision and Bias in ASTM Test Methods.
- ASTM E288 & E969 - Specifications for glass volumetric flasks and pipets used in sample and standard preparation.
- OSHA Regulations (29 CFR 1910.1000 and 1910.1200) - For worker safety relating to hazardous chemicals.
Implementation of ASTM D7359-23 helps organizations ensure product integrity, meet environmental and regulatory demands, and safeguard processes against corrosion and catalyst damage by accurately detecting and controlling total fluorine, chlorine, and sulfur in aromatic hydrocarbons.
Buy Documents
ASTM D7359-23 - Standard Test Method for Total Fluorine, Chlorine and Sulfur in Aromatic Hydrocarbons and Their Mixtures by Oxidative Pyrohydrolytic Combustion followed by Ion Chromatography Detection (Combustion Ion Chromatography-CIC)
REDLINE ASTM D7359-23 - Standard Test Method for Total Fluorine, Chlorine and Sulfur in Aromatic Hydrocarbons and Their Mixtures by Oxidative Pyrohydrolytic Combustion followed by Ion Chromatography Detection (Combustion Ion Chromatography-CIC)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D7359-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Total Fluorine, Chlorine and Sulfur in Aromatic Hydrocarbons and Their Mixtures by Oxidative Pyrohydrolytic Combustion followed by Ion Chromatography Detection (Combustion Ion Chromatography-CIC)". This standard covers: SIGNIFICANCE AND USE 5.1 The total fluorine, chlorine, and sulfur contained in aromatic hydrocarbon matrices can contribute to emissions, be harmful to many catalytic chemical processes, and lead to corrosion. This test method can be used to determine total sulfur and halogens in aromatic hydrocarbons and their mixtures. The results can be used for compliance determinations when acceptable to a regulatory authority using performance based criteria. SCOPE 1.1 This test method covers the individual determination of total fluorine, chlorine, and sulfur in aromatic hydrocarbons and their mixtures. Samples containing 0.10 mg/kg to 10 mg/kg of each element can be analyzed. 1.2 This method can be applied to sample concentrations outside the range of the scope by dilution of the sample in an appropriate solvent to bring the total concentrations of fluorine, chlorine, and sulfur within the range covered by the test method. However, it is the responsibility of the analyst to verify the solubility of the sample in the solvent and that the diluted sample results conform to the precision and accuracy of the method. 1.2.1 Special considerations must be made in order to attain detection limits below 1.0 mg/kg in a sample. The instrument must be clean and properly maintained to address potential sources of contamination, or carryover, or both. Multiple sequential injections shall be completed until a stable background is attained. A stable background is considered to be achieved when the analysis of a minimum of three consecutive system blanks have area counts equal to or less than 5 % RSD for the anions of interest. 1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The total fluorine, chlorine, and sulfur contained in aromatic hydrocarbon matrices can contribute to emissions, be harmful to many catalytic chemical processes, and lead to corrosion. This test method can be used to determine total sulfur and halogens in aromatic hydrocarbons and their mixtures. The results can be used for compliance determinations when acceptable to a regulatory authority using performance based criteria. SCOPE 1.1 This test method covers the individual determination of total fluorine, chlorine, and sulfur in aromatic hydrocarbons and their mixtures. Samples containing 0.10 mg/kg to 10 mg/kg of each element can be analyzed. 1.2 This method can be applied to sample concentrations outside the range of the scope by dilution of the sample in an appropriate solvent to bring the total concentrations of fluorine, chlorine, and sulfur within the range covered by the test method. However, it is the responsibility of the analyst to verify the solubility of the sample in the solvent and that the diluted sample results conform to the precision and accuracy of the method. 1.2.1 Special considerations must be made in order to attain detection limits below 1.0 mg/kg in a sample. The instrument must be clean and properly maintained to address potential sources of contamination, or carryover, or both. Multiple sequential injections shall be completed until a stable background is attained. A stable background is considered to be achieved when the analysis of a minimum of three consecutive system blanks have area counts equal to or less than 5 % RSD for the anions of interest. 1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7359-23 is classified under the following ICS (International Classification for Standards) categories: 71.080.20 - Halogenated hydrocarbons. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7359-23 has the following relationships with other standards: It is inter standard links to ASTM E969-02(2019), ASTM E288-10(2017), ASTM D6809-02(2016), ASTM E177-14, ASTM E177-13, ASTM E691-13, ASTM E969-02(2012), ASTM E691-11, ASTM D3437-11, ASTM E177-10, ASTM D3437-10, ASTM E288-10, ASTM E177-08, ASTM E691-08, ASTM E29-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7359-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7359 − 23
Standard Test Method for
Total Fluorine, Chlorine and Sulfur in Aromatic
Hydrocarbons and Their Mixtures by Oxidative
Pyrohydrolytic Combustion followed by Ion
Chromatography Detection (Combustion Ion
Chromatography-CIC)
This standard is issued under the fixed designation D7359; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 1.5 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 This test method covers the individual determination of
responsibility of the user of this standard to establish appro-
total fluorine, chlorine, and sulfur in aromatic hydrocarbons
priate safety, health, and environmental practices and deter-
and their mixtures. Samples containing 0.10 mg ⁄kg to
mine the applicability of regulatory limitations prior to use.
10 mg ⁄kg of each element can be analyzed.
See Section 9.
1.2 This method can be applied to sample concentrations
1.6 This international standard was developed in accor-
outside the range of the scope by dilution of the sample in an
dance with internationally recognized principles on standard-
appropriate solvent to bring the total concentrations of fluorine,
ization established in the Decision on Principles for the
chlorine, and sulfur within the range covered by the test
Development of International Standards, Guides and Recom-
method. However, it is the responsibility of the analyst to
mendations issued by the World Trade Organization Technical
verify the solubility of the sample in the solvent and that the
Barriers to Trade (TBT) Committee.
diluted sample results conform to the precision and accuracy of
the method.
2. Referenced Documents
1.2.1 Special considerations must be made in order to attain
2.1 ASTM Standards:
detection limits below 1.0 mg ⁄kg in a sample. The instrument
D1193 Specification for Reagent Water
must be clean and properly maintained to address potential
D3437 Practice for Sampling and Handling Liquid Cyclic
sources of contamination, or carryover, or both. Multiple
Products
sequential injections shall be completed until a stable back-
D3505 Test Method for Density or Relative Density of Pure
ground is attained. A stable background is considered to be
Liquid Chemicals
achieved when the analysis of a minimum of three consecutive
D6809 Guide for Quality Control and Quality Assurance
system blanks have area counts equal to or less than 5 % RSD
Procedures for Aromatic Hydrocarbons and Related Ma-
for the anions of interest.
terials
1.3 In determining the conformance of the test results using
E29 Practice for Using Significant Digits in Test Data to
this method to applicable specifications, results shall be
Determine Conformance with Specifications
rounded off in accordance with the rounding-off method of
E177 Practice for Use of the Terms Precision and Bias in
Practice E29.
ASTM Test Methods
E288 Specification for Laboratory Glass Volumetric Flasks
1.4 The values stated in SI units are to be regarded as
E691 Practice for Conducting an Interlaboratory Study to
standard. No other units of measurement are included in this
Determine the Precision of a Test Method
standard.
E969 Specification for Glass Volumetric (Transfer) Pipets
This test method is under the jurisdiction of ASTM Committee D16 on
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi-
bility of Subcommittee D16.04 on Instrumental Analysis. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved April 1, 2023. Published April 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2008. Last previous edition approved in 2018 as D7359 – 18. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D7359-23. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7359 − 23
2.2 Other Documents: 3.1.10 calibration standard, n—standard prepared from the
OSHA Regulations, 29 CFR paragraphs 1910.1000 and working standard and subsequently used to calibrate the
1910.1200 instrument.
3.2 Abbreviations:
3. Terminology
3.2.1 CIC—combustion ion chromatography
3.1 Definitions:
3.2.2 Conc—concentration
3.1.1 combustion ion chromatography, n—an analytical sys-
3.2.3 CRM—certified reference material
tem consisting of pyrohydrolytic combustion followed by ion
chromatographic detection.
3.2.4 HCI—hydrogen chloride
3.1.2 oxidative pyrohydrolytic combustion, n—a process in
3.2.5 HF—hydrogen fluoride
which a sample undergoes combustion at temperatures greater
3.2.6 HX—hydrogen halide
than 900 °C in an oxygen-rich environment and in the presence
of excess water vapor not originating from the combustion of
3.2.7 IC—ion chromatograph or ion chromatography
the sample. In oxidative pyrohydrolytic combustion, the
3.2.8 SO —sulfur oxide (SO and SO )
sample is pyrolyzed into carbon dioxide, water, hydrogen
x 2 3
halides, and residual ash; typically elemental oxides.
3.2.9 SO —sulfur dioxide
3.1.3 halogens (X), n—the elements fluorine, chlorine,
3.2.10 SO —sulfur trioxide
bromine, and iodine.
3.2.11 SO —sulfate
3.1.4 hydrogen halide (HX), n—inorganic compounds with
the formula HX where X is one of the halogens: fluorine, 3.2.12 SRM—standard reference material
chlorine, bromine, and iodine. Hydrogen halides are gases that
3.2.13 Std—standard
dissolve in water to give acids.
3.1.5 sulfur oxide (SO ), n—refers to one or more of the
x 4. Summary of Test Method
following compounds:
4.1 A sample of known weight or volume is placed into a
3.1.5.1 sulfur dioxide (SO )
sample boat and introduced at a controlled rate into a high-
3.1.5.2 sulfur trioxide (SO )
temperature combustion tube. There the sample is combusted
3.1.5.3 sulfate (SO )
in an oxygen rich pyrohydrolytic environment. The gaseous
3.1.6 system blank, n—a combustion ion chromatography
by-products of the combusted sample are trapped in an
(CIC) analysis with no solvent or sample injection in which the
absorption medium where the hydrogen halides (HX) formed
-
same combustion, chromatography and time protocols are used
during combustion disassociate into their respective ions, X
as for the sample analysis, but without the combustion of a
while the sulfur oxides (SO ) formed are further oxidized to
X
2-
sample or solvent blank. The system blank must be equal to or
SO in the presence of an oxidizing agent. An aliquot of
less than 50 % (1/2) the area counts of the lowest calibration
known volume of the absorbing solution is then automatically
standard used for calibration and a maximum of 50 % (1/2) of
injected into an ion chromatograph (IC) by means of a sample
the area count of the solvent blank used in the preparation of
injection valve. The halide and sulfate anions are separated on
the calibration standards for the anions of interest.
the anion separation column of the IC. The conductivity of the
3.1.7 solvent blank, n—a combustion ion chromatography eluent is reduced with an anion suppression device prior to the
(CIC) analysis of the solvent used for preparation of the ion chromatograph’s conductivity detector, where the anions of
calibration standards in which the same combustion, interest are measured. Quantification of the fluorine, chlorine
chromatography, time protocols and injection volumes are used and sulfur in the original combusted sample is achieved by first
calibrating the system with a series of standards containing
as for the sample analysis. The solvent blank area count must
be less than or equal to two times (2×) the system blank and known amounts of fluorine, chlorine and sulfur and then
analyzing unknown samples under the same conditions as the
50 % (1/2) or less than the area counts of the lowest calibration
standard used in the calibration of the system for the anions of standards. The combined system of pyrohydrolytic combustion
followed by ion chromatographic detection is referred to as
interest.
Combustion Ion Chromatography (CIC).
3.1.8 stock standard solution, n—standard prepared from
primary standards and subsequently used to prepare the work-
5. Significance and Use
ing standard.
3.1.9 working standard solution, n—standard prepared from 5.1 The total fluorine, chlorine, and sulfur contained in
aromatic hydrocarbon matrices can contribute to emissions, be
the stock standard solution and subsequently used to prepare
the calibration standards. harmful to many catalytic chemical processes, and lead to
corrosion. This test method can be used to determine total
sulfur and halogens in aromatic hydrocarbons and their mix-
tures. The results can be used for compliance determinations
Available from U.S. Government Printing Office Superintendent of Documents,
when acceptable to a regulatory authority using performance
732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
www.access.gpo.gov. based criteria.
D7359 − 23
6. Interferences 7.11 Ion Chromatograph (IC) —An analytical system with
all required accessories including columns, suppressor and
6.1 Substances that co-elute with the anions of interest will
detector.
interfere. A high concentration of one anion can interfere with
7.11.1 Injection System, capable of delivering 20 μL to
other constituents if their retention times are close enough to
500 μL with a precision better than 1 % or as recommended for
affect the resolution of their peak.
this determination by the manufacturer. Larger volumes can be
used as long as the performance criteria of the method are not
7. Apparatus
degraded.
7.1 Autosampler, capable of accurately delivering a known
7.11.2 Pumping System, capable of delivering mobile phase
volume of sample, typically in the range of 10 μL to 100 μL,
flows between 0.2 mL ⁄min and 2.5 mL ⁄min with a precision
into the sample boat.
better than 2 %, or as recommended for this determination by
the manufacturer.
NOTE 1—The sample syringe should be rinsed with clean solvent
followed by a rinse with the next sample when changing from one vial to 7.11.3 Continuous Eluent Generation (optional), to auto-
another. Follow the manufacturer’s recommendation to minimize carry-
matically prepare and purify the eluent used in the ion
over.
chromatography. Electrolytic eluent generation and auto-buret
7.2 Balance, analytical, with sensitivity to 0.0001 g. preparation of eluent by means of in-line dilution of a stock
solution have been found satisfactory for this method. Other
7.3 Boat Inlet System—The system provides a sampling port
continuous eluent generation devices may be used if the
for the introduction of liquid samples into the sample boat and
precision and accuracy of the method are not degraded.
is connected to the inlet of the combustion tube. The system is
7.11.4 Anion Pre-concentration Column (optional), used for
swept by a humidified inert carrier gas and shall be capable of
anion pre-concentration and matrix elimination. Pre-
allowing the quantitative delivery of the material to be ana-
concentration enables larger volumes of absorbing solu-
lyzed into the oxidation zone at a controlled rate.
tion (1 mL to 3 mL) to be analyzed without the associated
7.4 Boat Inlet Cooler—Sample volatility requires an appa-
water dip. Matrix elimination refers to the elimination of any
ratus capable of cooling the sample boat prior to sample
unreacted hydrogen peroxide in the absorbing solution prior to
injection into the boat.
injection onto the guard and anion separator columns and
7.5 Gas Flow Control—The apparatus must be equipped potentially interfere with the fluoride peak resolution.
with flow controllers capable of maintaining a constant flow of 7.11.5 Guard Column, for protection of the analytical col-
oxygen and argon or helium carrier gas. umn from strongly retained constituents. Improved separation
is obtained with additional theoretical plates.
7.6 Furnace—An electric furnace which can maintain a
7.11.6 Anion Separator Column, capable of producing sat-
minimum temperature of 900 °C.
isfactory baseline separations of the anion peaks of interest as
7.7 Gas Absorption Unit, having an absorption tube with
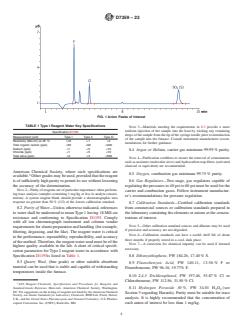
shown in Fig. 1.
sufficient capacity to hold a minimum of 5 mL which is
7.11.7 Anion Suppressor Device—Reduces the background
automatically filled with a known volume of absorption solu-
conductivity of the eluent after separation by the anion
tion by a built-in burette or other similar device. The gas
separator column. Both chemical and continuous electrolytic
absorption unit is interfaced to the IC and injects an aliquot of
suppressors have been found satisfactory for this method.
the absorption solution into the IC after the sample is com-
Other anion suppressor devices may be used as long as the
busted and the by-products of combustion are absorbed. The
precision and accuracy of the method are not degraded.
gas absorption unit rinses the absorption tube and the transfer
7.11.8 Data Acquisition System—An integrator or computer
lines from the combustion tube to the gas absorption unit with
data handling system capable of integrating the peak areas of
Type I reagent water (8.2) or other appropriate absorption
an ion chromatograph.
solution prior to sample combustion and after the absorption
7.12 Quartz or Ceramic Sample Boats of sufficient size to
solution is injected into the IC to minimize cross contamina-
hold 10 μL to 100 μL. The boat is filled with quartz wool or
tion.
other suitable material (8.3) to wick any remaining drops of the
7.8 Gas-Tight Sampling Syringe, of 10 μL, 25 μL, 50 μL,
sample from the tip of the syringe needle prior to introduction
100 μL, or 250 μL capacity and capable of accurately deliver-
of the sample into the furnace.
ing microliter quantities.
8. Reagents and Materials
7.9 Pyrohydrolytic Combustion Tube made of quartz and
capable of withstanding temperatures up to 1100 °C. The
8.1 Purity of Reagents—Reagent grade or higher purity
combustion tube must be of ample volume and may include chemicals shall be used for the preparation of all samples,
quartz wool or other suitable medium to provide sufficient
standards, eluent, and regenerator solutions. Unless otherwise
mixing and surface area to ensure complete combustion of the
indicated, it is intended that all reagents shall conform to the
sample. specification of the Committee on Analytical Reagents of the
7.10 Humidifier Delivery System, capable of delivering
Type 1 reagent water (8.2) to the combustion tube at a
Many different companies manufacture automatic ion chromatographs. Consult
controlled rate sufficient to provide a pyrohydrolytic environ-
the specific manufacturer instruction manuals for details regarding setup and
ment. operation.
D7359 − 23
FIG. 1 Anion Peaks of Interest
TABLE 1 Type I Reagent Water Key Specifications
NOTE 3—Materials meeting the requirements in 8.3 provide a more
uniform injection of the sample into the boat by wicking any remaining
Specification D1193
drops of the sample from the tip of the syringe needle prior to introduction
Measurement (unit) Type I Type II Type III
of the sample into the furnace. Consult instrument manufacturer recom-
Resistivity (MΩ·cm) at 25 °C >18 >1 >4
mendations for further guidance.
Total organic carbon (ppb) <50 <50 <200
Sodium (ppb) <1 <5 <10
8.4 Argon or Helium, carrier gas minimum 99.99 % purity.
Chloride (ppb) <1 <5 <10
Total silica (ppb) <3 <3 <500
NOTE 4—Purification scrubbers to ensure the removal of containments
such as moisture (molecular sieve) and hydrocarbon trap filters (activated
charcoal or equivalent) are recommended.
American Chemical Society, where such specifications are
8.5 Oxygen, combustion gas minimum 99.75 % purity.
available. Other grades may be used, provided that the reagent
8.6 Gas Regulators—Two-stage, gas regulators capable of
is of sufficiently high purity to permit its use without lessening
the accuracy of the determination. regulating the pressures to 40 psi to 60 psi must be used for the
NOTE 2—Purity of reagents are of particular importance when perform-
carrier and combustion gases. Follow instrument manufactur-
ing trace analysis (samples containing 1 mg ⁄kg or less in analyte concen-
er’s recommendations for pressure regulation.
tration). A system reagent blank should provide a chromatographic area
response no greater than 50 % (1/2) of the lowest calibration standard.
8.7 Calibration Standards—Certified calibration standards
8.2 Purity of Water—Unless otherwise indicated, references from commercial sources or calibration standards prepared in
to water shall be understood to mean Type 1 having 18 MΩ cm
the laboratory containing the elements or anions at the concen-
resistance and conforming to Specification D1193. Comply
trations of interest.
with all ion chromatograph instrument and column vendor
NOTE 5—Other calibration standard sources and diluents may be used
requirements for eluent preparation and handling (for example,
if precision and accuracy are not degraded.
filtering, degassing, and the like). The reagent water is critical
NOTE 6—Calibration standards can have a useful shelf life of about
to the performance, repeatability, reproducibility, and accuracy
three months if properly stored in a cool, dark place.
of the method. Therefore, the reagent water used must be of the
NOTE 7—A correction for chemical impurity can be used if deemed
highest quality available in the lab. A chart of critical specifi-
necessary.
cation parameters for Type I reagent water in accordance with
Specification D1193is listed in Table 1. 8.8 Dibenzothiophene, FW 184.26, 17.40 % S.
8.3 Quartz Wool, (fine grade) or other suitable absorbent
8.9 Fluorobenzoic Acid, FW 140.11, 13.56 % F or
material can be used that is stable and capable of withstanding
Fluorobenzene, FW 96.10, 19.77% F.
temperatures inside the furnace.
8.10 2,4,5 Trichlorophenol, FW 197.46, 53.87 % Cl or
Chlorobenzene, FW 112.56, 31.50 % Cl.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington, 8.11 Hydrogen Peroxide 30 %, FW 34.01 H O (see
2 2
DC. For suggestions on the testing of reagents not listed by the American Chemical
Section 9 regarding Hazards). Purity must be suitable for trace
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
analysis. It is highly recommended that the concentration of
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. each anion of interest be less than 1 mg/kg.
D7359 − 23
NOTE 8—Fluka TraceSelect Ultra, Fluka TraceSelect, and EMD
8.17.2 2,4,5 Trichlorophenol, (Chlorine), 0.1856 g target
Suprapur have all proven to work well for this method.
weight or Chlorobenzene, (Chlorine), 0.3174 g target weight.
8.12 Eluent Solution—Follow the specific guidelines for the
8.17.3 Dibenzothiophene, (Sulfur), 0.5748 g target weight.
preparation and use of the eluent solution from the manufac-
Calculation of Concentration of Stock Standard Solution
turer of the columns being used. Other concentrations may be
used if precision and accuracy of the method is not degraded.
Stock Standard Solution~μg/mL! 5 ~A! ~B! ~10 ! ~P!/~V! ~K!
The solutions recommended by the column manufacturer can
(1)
be purchased from qualified vendors as long as the
where:
performance, precision, and accuracy are not degraded (see
A = weight of the target compound in grams, g,
Section 9 regarding Hazards).
B = % concentration of the elements in the respective target
8.13 Suppressor Solutions:
compounds listed in 8.8, 8.9, and 8.10,
8.13.1 Chemical Suppressor Regenerant Solution—Follow
V = final diluted volume, mL,
the specific manufacturer guidelines for the preparation and use
P = % purity of target standard compounds listed in 8.8, 8.9,
of the suppressor solution. The manufacturer recommended
and 8.10, and
solutions can be purchased from qualified vendors as long as
K = 100 (correction to convert % to μg/g).
the performance, precision, and accuracy are not degraded.
8.18 Absorbing Solution—Dilute a sufficient amount of
8.13.2 Electrolytic Suppressor Current Setting—Follow the
hydrogen peroxide stock solution listed in 8.11 to achieve a
specific guidelines for the current setting from the vendor of
final concentration of approximately 100 μg/mL. (Approxi-
the suppressor being used based upon the flow rate and eluent
mately 0.7 mL of 30 % hydrogen peroxide added to 2 L of
concentration being used for the analysis.
absorbing solution will give a final concentration of approxi-
8.14 Solvent—The solvent of choice should be capable of
mately 100 μg/mL.) The use of hydrogen peroxide in the
dissolving the standard or sample. The solvent should contain
absorbing solution ensures that all SO species are converted
X
less than 0.05 mg/kg of the elements or anions of interest. The
to SO prior to detection by the IC. Hydrogen peroxide is not
blank value must be determined for each new bottle of solve
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7359 − 18 D7359 − 23
Standard Test Method for
Total Fluorine, Chlorine and Sulfur in Aromatic
Hydrocarbons and Their Mixtures by Oxidative
Pyrohydrolytic Combustion followed by Ion
Chromatography Detection (Combustion Ion
Chromatography-CIC)
This standard is issued under the fixed designation D7359; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the individual determination of total fluorine, chlorine, and sulfur in aromatic hydrocarbons and their
mixtures. Samples containing 0.100.10 mg ⁄kg to 1010 mg mg/kg ⁄kg of each element can be analyzed.
1.2 This method can be applied to sample concentrations outside the range of the scope by dilution of the sample in an appropriate
solvent to bring the total concentrations of fluorine, chlorine, and sulfur within the range covered by the test method. However,
it is the responsibility of the analyst to verify the solubility of the sample in the solvent and that the diluted sample results conform
to the precision and accuracy of the method.
1.2.1 Special considerations must be made in order to attain detection limits below 1.01.0 mg mg/kg ⁄kg in a sample. The
instrument must be clean and properly maintained to address potential sources of contamination, of contamination, or carryover,
or both. Multiple sequential injections shall be completed until a stable background is attained. A stable background is considered
to be achieved when the analysis of a minimum of three consecutive system blanks have area counts equal to or less than 5 % RSD
for the anions of interest.
1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off
in accordance with the rounding-off method of Practice E29.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. See Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsibility of
Subcommittee D16.04 on Instrumental Analysis.
Current edition approved Nov. 1, 2018April 1, 2023. Published December 2018April 2023. Originally approved in 2008. Last previous edition approved in 20142018 as
D7359 – 14a. DOI: 10.1520/D7359-18.18. DOI: 10.1520/D7359-23.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7359 − 23
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D3437 Practice for Sampling and Handling Liquid Cyclic Products
D3505 Test Method for Density or Relative Density of Pure Liquid Chemicals
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E288 Specification for Laboratory Glass Volumetric Flasks
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E969 Specification for Glass Volumetric (Transfer) Pipets
2.2 Other Documents:
OSHA Regulations, 29 CFR paragraphs 1910.1000 and 1910.1200
3. Terminology
3.1 Definitions:
3.1.1 combustion ion chromatography, n—an analytical system consisting of pyrohydrolytic combustion followed by ion
chromatographic detection.
3.1.2 oxidative pyrohydrolytic combustion, n—a process in which a sample undergoes combustion at temperatures greater than
900°C900 °C in an oxygen rich oxygen-rich environment and in the presence of excess water vapor not originating from the
combustion of the sample. In oxidative pyrohydrolytic combustion, the sample is pyrolyzed into carbon dioxide, water, hydrogen
halides, and residual ash; typically elemental oxides.
3.1.3 halogens (X), n—the elements fluorine, chlorine, bromine, and iodine.
3.1.4 hydrogen halide (HX), n—are inorganic compounds with the formula HX where X is one of the halogens: fluorine, chlorine,
bromine, and iodine. Hydrogen halides are gases that dissolve in water to give acids.
3.1.5 sulfur oxide (SO ), n—refers to one or more of the following compounds:
x
3.1.5.1 sulfur dioxide (SO )
3.1.5.2 sulfur trioxide (SO )
3.1.5.3 sulfate (SO )
3.1.6 system blank, n—a combustion ion chromatography (CIC) analysis with no solvent or sample injection in which the same
combustion, chromatography and time protocols are used as for the sample analysis, but without the combustion of a sample or
solvent blank. The system blank must be equal to or less than 50 % (1/2) the area counts of the lowest calibration standard used
for calibration and a maximum of 50 % (1/2) of the area count of the solvent blank used in the preparation of the calibration
standards for the anions of interest.
3.1.7 solvent blank, n—a combustion ion chromatography (CIC) analysis of the solvent used for preparation of the calibration
standards in which the same combustion, chromatography, time protocols and injection volumes are used as for the sample
analysis. The solvent blank area count must be less than or equal to two times (2×) the system blank and 50 % (1/2) or less than
the area counts of the lowest calibration standard used in the calibration of the system for the anions of interest.
3.1.8 stock standard solution, n—standard prepared from primary standards and subsequently used to prepare the working
standard.
3.1.9 working standard solution, n—standard prepared from the stock standard solution and subsequently used to prepare the
calibration standards.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from U.S. Government Printing Office Superintendent of Documents, 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
www.access.gpo.gov.
D7359 − 23
3.1.10 calibration standard, n—standard prepared from the working standard and subsequently used to calibrate the instrument.
3.2 Abbreviations:
3.2.1 CIC—combustion ion chromatography
3.2.2 Conc—concentration
3.2.3 CRM—certified reference material
3.2.4 HCI—hydrogen chloride
3.2.5 HF—hydrogen fluoride
3.2.6 HX—hydrogen halide
3.2.7 IC—ion chromatograph or ion chromatography
3.2.8 SO —sulfur oxide (SO and SO )
x 2 3
3.2.9 SO —sulfur dioxide
3.2.10 SO —sulfur trioxide
3.2.11 SO —sulfate
3.2.12 Std—standard
3.2.12 SRM—standard reference material
3.2.13 Std—standard
4. Summary of Test Method
4.1 A sample of known weight or volume is placed into a sample boat and introduced at a controlled rate into a high temperature
high-temperature combustion tube. There the sample is combusted in an oxygen rich pyrohydrolytic environment. The gaseous
by-products of the combusted sample are trapped in an absorption medium where the hydrogen halides (HX) formed during
- 2-
combustion disassociate into their respective ions, X while the sulfur oxides (SO ) formed are further oxidized to SO in the
X 4
presence of an oxidizing agent. An aliquot of known volume of the absorbing solution is then automatically injected into an ion
chromatograph (IC) by means of a sample injection valve. The halide and sulfate anions are separated on the anion separation
column of the IC. The conductivity of the eluent is reduced with an anion suppression device prior to the ion chromatograph’s
conductivity detector, where the anions of interest are measured. Quantification of the fluorine, chlorine and sulfur in the original
combusted sample is achieved by first calibrating the system with a series of standards containing known amounts of fluorine,
chlorine and sulfur and then analyzing unknown samples under the same conditions as the standards. The combined system of
pyrohydrolytic combustion followed by ion chromatographic detection is referred to as Combustion Ion Chromatography (CIC).
5. Significance and Use
5.1 The total fluorine, chlorine, and sulfur contained in aromatic hydrocarbon matrices can contribute to emissions, be harmful
to many catalytic chemical processes, and lead to corrosion. This test method can be used to determine total sulfur and halogens
in aromatic hydrocarbons and their mixtures. The results can be used for compliance determinations when acceptable to a
regulatory authority using performance based criteria.
D7359 − 23
6. Interferences
6.1 Substances that co-elute with the anions of interest will interfere. A high concentration of one anion can interfere with other
constituents if their retention times are close enough to affect the resolution of their peak.
7. Apparatus
7.1 Autosampler, capable of accurately delivering a known volume of sample, typically in the range of 10 to 100 μL, 10 μL to
100 μL, into the sample boat.
NOTE 1—The sample syringe should be rinsed with clean solvent followed by a rinse with the next sample when changing from one vial to another. Follow
the manufacturer’s recommendation to minimize carryover.
7.2 Balance, analytical, with sensitivity to 0.0001 g.0.0001 g.
7.3 Boat Inlet System—The system provides a sampling port for the introduction of liquid samples into the sample boat and is
connected to the inlet of the combustion tube. The system is swept by a humidified inert carrier gas and shall be capable of allowing
the quantitative delivery of the material to be analyzed into the oxidation zone at a controlled rate.
7.4 Boat Inlet Cooler—Sample volatility requires an apparatus capable of cooling the sample boat prior to sample injection into
the boat.
7.5 Gas Flow Control—The apparatus must be equipped with flow controllers capable of maintaining a constant flow of oxygen
and argon or helium carrier gas.
7.6 Furnace—An electric furnace which can maintain a minimum temperature of 900°C.900 °C.
7.7 Gas Absorption Unit, having an absorption tube with sufficient capacity to hold a minimum of 5 mL which is automatically
filled with a known volume of absorption solution by a built-in burette or other similar device. The gas absorption unit is interfaced
to the IC and injects an aliquot of the absorption solution into the IC after the sample is combusted and the by-products of
combustion are absorbed. The gas absorption unit rinses the absorption tube and the transfer lines from the combustion tube to the
gas absorption unit with Type I reagent water (8.2) or other appropriate absorption solution prior to sample combustion and after
the absorption solution is injected into the IC to minimize cross contamination.
7.8 Gas-Tight Sampling Syringe, of 10, 25, 50, 100, or 250- μL 10 μL, 25 μL, 50 μL, 100 μL, or 250 μL capacity and capable of
accurately delivering microliter quantities.
7.9 Pyrohydrolytic Combustion Tube made of quartz and capable of withstanding temperatures up to 1100°C.1100 °C. The
combustion tube must be of ample volume and may include quartz wool or other suitable medium to provide sufficient mixing and
surface area to ensure complete combustion of the sample.
7.10 Humidifier Delivery System, capable of delivering Type 1 reagent water (8.2) to the combustion tube at a controlled rate
sufficient to provide a pyrohydrolytic environment.
7.11 Ion Chromatograph (IC),(IC) —anAn analytical system with all required accessories including columns, suppressor and
detector.
7.11.1 Injection System, capable of delivering 20 to 500 μL 20 μL to 500 μL with a precision better than 1 % or as recommended
for this determination by the manufacturer. Larger volumes can be used as long as the performance criteria of the method are not
degraded.
7.11.2 Pumping System, capable of delivering mobile phase flows between 0.20.2 mL ⁄min and 2.52.5 mL mL/min ⁄min with a
precision better than 2 %, 2 %, or as recommended for this determination by the manufacturer.
Many different companies manufacture automatic ion chromatographs. Consult the specific manufacturer instruction manuals for details regarding setup and operation.
D7359 − 23
7.11.3 Continuous Eluent Generation (optional), to automatically prepare and purify the eluent used in the ion chromatography.
Electrolytic eluent generation and auto-buret preparation of eluent by means of in-line dilution of a stock solution have been found
satisfactory for this method. Other continuous eluent generation devices may be used if the precision and accuracy of the method
are not degraded.
7.11.4 Anion Pre-concentration Column (optional), used for anion pre-concentration and matrix elimination. Pre-concentration
enables larger volumes of absorbing solution (1 to 3 mL) solution (1 mL to 3 mL) to be analyzed without the associated water dip.
Matrix elimination refers to the elimination of any unreacted hydrogen peroxide in the absorbing solution prior to injection onto
the guard and anion separator columns and potentially interfere with the fluoride peak resolution.
7.11.5 Guard Column, for protection of the analytical column from strongly retained constituents. Improved separation is obtained
with additional theoretical plates.
7.11.6 Anion Separator Column, capable of producing satisfactory baseline separations of the anion peaks of interest as shown in
Fig. 1.
7.11.7 Anion Suppressor Device, Device—reducesReduces the background conductivity of the eluent after separation by the anion
separator column. Both chemical and continuous electrolytic suppressors have been found satisfactory for this method. Other anion
suppressor devices may be used as long as the precision and accuracy of the method are not degraded.
7.11.8 Data Acquisition System, System—anAn integrator or computer data handling system capable of integrating the peak areas
of an ion chromatographchromatograph.
7.12 Quartz or Ceramic Sample Boats of sufficient size to hold 1010 μL to 100 μL. The boat is filled with quartz wool or other
suitable material (8.3) to wick any remaining drops of the sample from the tip of the syringe needle prior to introduction of the
sample into the furnace.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade or higher purity chemicals shall be used for the preparation of all samples, standards,
eluent, and regenerator solutions. Unless otherwise indicated, it is intended that all reagents shall conform to the specification of
FIG. 1 Anion Peaks of Interest
D7359 − 23
TABLE 1 Type I Reagent Water Key Specifications
Specification D1193–06 (2011)
Measurement (unit) Type I Type II Type III
Resistivity (M·cm) at 25°C >18 >1 >4
Resistivity (M·cm) at 25 °C >18 >1 >4
Total organic carbon (ppb) <50 <50 <200
Sodium (ppb) <1 <5 <10
Chloride (ppb) <1 <5 <10
Total silica (ppb) <3 <3 <500
the Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades
may be used, provided that the reagent is of sufficiently high purity to permit its use without lessening the accuracy of the
determination.
NOTE 2—Purity of reagents are of particular importance when performing trace analysis (samples containing 11 mg mg/kg ⁄kg or less in analyte
concentration). A system reagent blank should provide a chromatographic area response no greater than 50 % 50 % (1/2) of the lowest calibration
standard.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean Type 1 having 18 MΩ cm
resistance and conforming to Specification D1193. Comply with all ion chromatograph instrument and column vendor
requirements for eluent preparation and handling (for example, filtering, degassing, and the like). The reagent water is critical to
the performance, repeatability, reproducibility, and accuracy of the method. Therefore, the reagent water used must be of the
highest quality available in the lab. A chart of critical specification parameters for Type I reagent water in accordance with
Specification D1193–06 (2011) is listed in Table 1.
8.3 Quartz Wool, (fine grade) or other suitable absorbent material can be used that is stable and capable of withstanding
temperatures inside the furnace.
NOTE 3—Materials meeting the requirements in 8.3 provide a more uniform injection of the sample into the boat by wicking any remaining drops of the
sample from the tip of the syringe needle prior to introduction of the sample into the furnace. Consult instrument manufacturer recommendations for
further guidance.
8.4 Argon or Helium, carrier gas minimum 99.99 % purity.
NOTE 4—Purification scrubbers to ensure the removal of containments such as moisture (molecular sieve) and hydrocarbon trap filters (activated charcoal
or equivalent) are recommended.
8.5 Oxygen, combustion gas minimum 99.75 % purity.
8.6 Gas Regulators, Regulators—two-stage,Two-stage, gas regulators capable of regulating the pressures to 40 to 60 psi 40 psi to
60 psi must be used for the carrier and combustion gases. Follow instrument manufacturer’s recommendations for pressure
regulation.
8.7 Calibration Standards, Standards—certifiedCertified calibration standards from commercial sources or calibration standards
prepared in the laboratory containing the elements or anions at the concentrations of interest.
NOTE 5—Other calibration standard sources and diluents may be used if precision and accuracy are not degraded.
NOTE 6—Calibration standards can have a useful shelf life of about three months if properly stored in a cool, dark place.
NOTE 7—A correction for chemical impurity can be used if deemed necessary.
8.8 Dibenzothiophene, FW 184.26, 17.40 % S.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
D7359 − 23
8.9 Fluorobenzoic Acid, Fluorobenzoic Acid, FW 140.11, 13.56 % F or Fluorobenzene, FW 96.10, 19.77% F.
8.10 2,4,5 Trichlorophenol, 2,4,5 Trichlorophenol, FW 197.46, 53.87 % Cl or Chlorobenzene, FW 112.56, 31.50 % Cl.
8.11 Hydrogen Peroxide 30 %, FW 34.01 H O (see Section 9 regarding Hazards). Purity must be suitable for trace analysis. It
2 2
is highly recommended that the concentration of each anion of interest be less than 1 mg/kg.
6 7
NOTE 8—Fluka TraceSelect Ultra, Fluka TraceSelect, and EMD Suprapur have all proven to work well for this method.
8.12 Eluent Solution—Follow the specific guidelines for the preparation and use of the eluent solution from the manufacturer of
the columns being used. Other concentrations may be used if precision and accuracy of the method is not degraded. The solutions
recommended by the column manufacturer can be purchased from qualified vendors as long as the performance, precision, and
accuracy are not degraded (see Section 9 regarding Hazards).
8.13 Suppressor Solutions:
8.13.1 Chemical Suppressor Regenerant Solution—Follow the specific manufacturer guidelines for the preparation and use of the
suppressor solution. The manufacturer recommended solutions can be purchased from qualified vendors as long as the
performance, precision, and accuracy are not degraded.
8.13.2 Electrolytic Suppressor Current Setting—Follow the specific guidelines for the current setting from the vendor of the
suppressor being used based upon the flow rate and eluent concentration being used for the analysis.
8.14 Solvent—The solvent of choice should be capable of dissolving the standard or sample. The solvent should contain less than
0.05 mg/kg of the elements or anions of interest. The blank value must be determined for each new bottle of solvent. Suggested
solvents include, but are not limited to, iso-octane, xylene, toluene, and methanol.
8.15 Volumetric Flasks–Type Class A, in accordance with Specification E288 at the volume required for the preparation of
standards, reagents, and solutions.
8.16 Volumetric Pipets–Type Class A, in accordance with Specification E969 at the volume required for preparation of standards,
reagents, and solutions.
8.17 Stock Standard Solution(s) of approximately 1000 1000 μg μg/mL—⁄mL—Prepare stock standard solution(s) by accurately
weighing to within 10 % of the target weights for any or all of the target standard compound(s) listed in 8.17.1 (choose one of the
options for Fluorine), 8.17.2 (choose one of the options for Chlorine), and 8.17.3 into a 100 mL Type Class A volumetric flask.
Dilute to volume with the selected solvent listed in 8.14. Calculate the actual concentration of the stock standard solution(s) for
each element by using the formula in Eq 1with the actual recorded weight of the target compound used for each element. This stock
standar
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...