ASTM D7754-23
(Test Method)Standard Test Method for Determination of Trace Oxygenates in Automotive Spark-Ignition Engine Fuel by Multidimensional Gas Chromatography
Standard Test Method for Determination of Trace Oxygenates in Automotive Spark-Ignition Engine Fuel by Multidimensional Gas Chromatography
SIGNIFICANCE AND USE
5.1 The analysis of trace oxygenates in automotive spark-ignition engine fuel has become routine in certain areas to ensure compliance whenever oxygenated fuels are used. In addition, test methods to measure trace levels of oxygenates in automotive spark-ignition fuel are necessary to assess product quality.
SCOPE
1.1 This test method covers the determination of trace oxygenates in automotive spark-ignition engine fuel. The method used is a multidimensional gas chromatographic method using 1,2-dimethoxy ethane as the internal standard. The oxygenates that are analyzed are: methyl-tertiary butyl ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl ether (DIPE), methanol, tertiary-amyl methyl ether (TAME), n-propanol, i-propanol, n-butanol, i-butanol, tert-butyl alcohol, sec-butyl alcohol, and tert-pentanol. Ethanol is usually not measured as a trace oxygenate since ethanol can be used as the main oxygenate compound in finished automotive spark-ignition fuels such as reformulated automotive spark-ignition fuels. The concentration range of the oxygenates covered in the ILS study was from 10 mg/kg to 2000 mg/kg. In addition this method is also suitable for the measurement of the C5 isomeric alcohols (2-methyl-1-butanol, 2-methyl-2-butanol) present from the fermentation of ethanol.
1.2 The ethanol blending concentration for which this test method applies ranges from 1 % to 15% by volume. Higher concentrations of ethanol coelute with methanol in the analytical column. Lower levels of ethanol, similar to the other oxygenate, can be calibrated and analyzed also. If higher ethanol concentrations are expected, the window cutting technique can be used to avoid ethanol from entering the analytical column and interfere with the determination of the other oxygenates of interest. Refer to Appendix X1 for details.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Alternative units, in common usage, are also provided to increase clarity and aid the users of this test method.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.04.0L - Gas Chromatography Methods
Relations
- Effective Date
- 01-Nov-2023
- Refers
ASTM D4175-23a - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 15-Dec-2023
- Refers
ASTM D4175-23e1 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
- Refers
ASTM D4175-23 - Standard Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants - Effective Date
- 01-Jul-2023
Overview
ASTM D7754-23, titled "Standard Test Method for Determination of Trace Oxygenates in Automotive Spark-Ignition Engine Fuel by Multidimensional Gas Chromatography," is an internationally recognized standard developed by ASTM International. This standard outlines a precise methodology for quantifying trace levels of various oxygenates in automotive spark-ignition engine fuel, using multidimensional gas chromatography (GC) with 1,2-dimethoxyethane as the internal standard.
With the increasing use of oxygenated fuels to meet emissions regulations and fuel quality standards, accurately detecting trace oxygenates in automotive fuels has become essential for regulatory compliance and quality assurance. This method is suitable for concentrations of oxygenates from 10 mg/kg up to 2000 mg/kg and accommodates ethanol blending scenarios from 1% up to 15% by volume.
Key Topics
- Trace Oxygenates Analyzed: The method quantifies key oxygenates such as methyl-tertiary butyl ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl ether (DIPE), methanol, tertiary-amyl methyl ether (TAME), n-propanol, isopropanol, n-butanol, isobutanol, tert-butyl alcohol, sec-butyl alcohol, tert-pentanol, and certain C5 isomeric alcohols.
- Multidimensional Gas Chromatography: Utilizes a two-column GC setup with a pre-column for hydrocarbon separation and an oxygen-selective analytical column for precise quantification of oxygenates.
- Internal Standard: 1,2-dimethoxyethane is used to improve analytical accuracy and correct for sample handling or instrument variation.
- Scope and Limitations: Ethanol is usually excluded from trace analysis, as it is often the main oxygenate in reformulated fuels. The method is adaptable for lower-level ethanol or for traces when higher ethanol content is held back from the analytical column.
- Units of Measurement: The standard utilizes SI units exclusively, while alternative common units may be listed for clarity.
Applications
- Fuel Quality Control: Ensures that finished automotive spark-ignition fuels meet regulatory and contractual oxygenate specifications, supporting product quality and engine performance.
- Regulatory Compliance: Provides reliable trace-level measurement of oxygenates, which is critical for compliance with air quality and fuel formulations regulations such as those enforced in reformulated gasoline markets.
- Production Optimization: Refineries and fuel producers use this standard to monitor and control blend compositions, ensuring consistent and legal products.
- Research and Development: Ideal for laboratories involved in fuel development, emission studies, or forensic fuel analysis where precise quantification of oxygenate content is required.
- Ethanol Blends: Applicable to fuels with ethanol content ranging from 1% to 15% by volume, aiding in analysis for E10, E15, and similar blends.
Related Standards
Practitioners often reference or use ASTM D7754-23 in conjunction with the following related standards:
- ASTM D4815: Test method for determination of MTBE, ETBE, TAME, DIPE, and C1-C4 alcohols in gasoline by GC.
- ASTM D4057: Practice for manual sampling of petroleum and petroleum products.
- ASTM D4175: Terminology relating to petroleum products, liquid fuels, and lubricants.
- ASTM D4307: Practice for preparation of liquid blends for use as analytical standards.
- ASTM D6304: Test method for determination of water in petroleum products by Karl Fischer titration.
- ASTM E355: Practice for gas chromatography terms and relationships.
Practical Value
ASTM D7754-23 is a critical tool for fuel laboratories and quality-control professionals in the petroleum and automotive industries. By ensuring trace detection of oxygenates at low levels, this standard supports fuel specification compliance, assists in identifying off-specification batches, and helps maintain environmental and engine protection standards through validated, repeatable gas chromatography procedures.
Using this international standard also facilitates cross-border trade and harmonization, as it reflects principles established by the World Trade Organization for technical barriers to trade, making it widely accepted and applicable across global fuel markets.
Buy Documents
ASTM D7754-23 - Standard Test Method for Determination of Trace Oxygenates in Automotive Spark-Ignition Engine Fuel by Multidimensional Gas Chromatography
REDLINE ASTM D7754-23 - Standard Test Method for Determination of Trace Oxygenates in Automotive Spark-Ignition Engine Fuel by Multidimensional Gas Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D7754-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Trace Oxygenates in Automotive Spark-Ignition Engine Fuel by Multidimensional Gas Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 The analysis of trace oxygenates in automotive spark-ignition engine fuel has become routine in certain areas to ensure compliance whenever oxygenated fuels are used. In addition, test methods to measure trace levels of oxygenates in automotive spark-ignition fuel are necessary to assess product quality. SCOPE 1.1 This test method covers the determination of trace oxygenates in automotive spark-ignition engine fuel. The method used is a multidimensional gas chromatographic method using 1,2-dimethoxy ethane as the internal standard. The oxygenates that are analyzed are: methyl-tertiary butyl ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl ether (DIPE), methanol, tertiary-amyl methyl ether (TAME), n-propanol, i-propanol, n-butanol, i-butanol, tert-butyl alcohol, sec-butyl alcohol, and tert-pentanol. Ethanol is usually not measured as a trace oxygenate since ethanol can be used as the main oxygenate compound in finished automotive spark-ignition fuels such as reformulated automotive spark-ignition fuels. The concentration range of the oxygenates covered in the ILS study was from 10 mg/kg to 2000 mg/kg. In addition this method is also suitable for the measurement of the C5 isomeric alcohols (2-methyl-1-butanol, 2-methyl-2-butanol) present from the fermentation of ethanol. 1.2 The ethanol blending concentration for which this test method applies ranges from 1 % to 15% by volume. Higher concentrations of ethanol coelute with methanol in the analytical column. Lower levels of ethanol, similar to the other oxygenate, can be calibrated and analyzed also. If higher ethanol concentrations are expected, the window cutting technique can be used to avoid ethanol from entering the analytical column and interfere with the determination of the other oxygenates of interest. Refer to Appendix X1 for details. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Alternative units, in common usage, are also provided to increase clarity and aid the users of this test method. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The analysis of trace oxygenates in automotive spark-ignition engine fuel has become routine in certain areas to ensure compliance whenever oxygenated fuels are used. In addition, test methods to measure trace levels of oxygenates in automotive spark-ignition fuel are necessary to assess product quality. SCOPE 1.1 This test method covers the determination of trace oxygenates in automotive spark-ignition engine fuel. The method used is a multidimensional gas chromatographic method using 1,2-dimethoxy ethane as the internal standard. The oxygenates that are analyzed are: methyl-tertiary butyl ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl ether (DIPE), methanol, tertiary-amyl methyl ether (TAME), n-propanol, i-propanol, n-butanol, i-butanol, tert-butyl alcohol, sec-butyl alcohol, and tert-pentanol. Ethanol is usually not measured as a trace oxygenate since ethanol can be used as the main oxygenate compound in finished automotive spark-ignition fuels such as reformulated automotive spark-ignition fuels. The concentration range of the oxygenates covered in the ILS study was from 10 mg/kg to 2000 mg/kg. In addition this method is also suitable for the measurement of the C5 isomeric alcohols (2-methyl-1-butanol, 2-methyl-2-butanol) present from the fermentation of ethanol. 1.2 The ethanol blending concentration for which this test method applies ranges from 1 % to 15% by volume. Higher concentrations of ethanol coelute with methanol in the analytical column. Lower levels of ethanol, similar to the other oxygenate, can be calibrated and analyzed also. If higher ethanol concentrations are expected, the window cutting technique can be used to avoid ethanol from entering the analytical column and interfere with the determination of the other oxygenates of interest. Refer to Appendix X1 for details. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Alternative units, in common usage, are also provided to increase clarity and aid the users of this test method. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7754-23 is classified under the following ICS (International Classification for Standards) categories: 75.160.20 - Liquid fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7754-23 has the following relationships with other standards: It is inter standard links to ASTM D7754-19, ASTM D4175-23a, ASTM D4175-23e1, ASTM D4175-23. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7754-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7754 − 23
Standard Test Method for
Determination of Trace Oxygenates in Automotive Spark-
Ignition Engine Fuel by Multidimensional Gas
Chromatography
This standard is issued under the fixed designation D7754; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
1.1 This test method covers the determination of trace
mine the applicability of regulatory limitations prior to use.
oxygenates in automotive spark-ignition engine fuel. The
1.5 This international standard was developed in accor-
method used is a multidimensional gas chromatographic
dance with internationally recognized principles on standard-
method using 1,2-dimethoxy ethane as the internal standard.
ization established in the Decision on Principles for the
The oxygenates that are analyzed are: methyl-tertiary butyl
Development of International Standards, Guides and Recom-
ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl
mendations issued by the World Trade Organization Technical
ether (DIPE), methanol, tertiary-amyl methyl ether (TAME),
Barriers to Trade (TBT) Committee.
n-propanol, i-propanol, n-butanol, i-butanol, tert-butyl alcohol,
sec-butyl alcohol, and tert-pentanol. Ethanol is usually not
2. Referenced Documents
measured as a trace oxygenate since ethanol can be used as the
2.1 ASTM Standards:
main oxygenate compound in finished automotive spark-
D4057 Practice for Manual Sampling of Petroleum and
ignition fuels such as reformulated automotive spark-ignition
Petroleum Products
fuels. The concentration range of the oxygenates covered in the
D4175 Terminology Relating to Petroleum Products, Liquid
ILS study was from 10 mg ⁄kg to 2000 mg ⁄kg. In addition this
Fuels, and Lubricants
method is also suitable for the measurement of the C5 isomeric
D4307 Practice for Preparation of Liquid Blends for Use as
alcohols (2-methyl-1-butanol, 2-methyl-2-butanol) present
Analytical Standards
from the fermentation of ethanol.
D4815 Test Method for Determination of MTBE, ETBE,
1.2 The ethanol blending concentration for which this test
TAME, DIPE, tertiary-Amyl Alcohol and C to C Alco-
1 4
method applies ranges from 1 % to 15% by volume. Higher
hols in Gasoline by Gas Chromatography
concentrations of ethanol coelute with methanol in the analyti-
D6304 Test Method for Determination of Water in Petro-
cal column. Lower levels of ethanol, similar to the other
leum Products, Lubricating Oils, and Additives by Cou-
oxygenate, can be calibrated and analyzed also. If higher
lometric Karl Fischer Titration
ethanol concentrations are expected, the window cutting tech-
E355 Practice for Gas Chromatography Terms and Relation-
nique can be used to avoid ethanol from entering the analytical
ships
column and interfere with the determination of the other
oxygenates of interest. Refer to Appendix X1 for details. 3. Terminology
3.1 Definitions:
1.3 The values stated in SI units are to be regarded as
3.1.1 This test method makes reference to common gas
standard. No other units of measurement are included in this
chromatographic procedures, terms, and relationships. Detailed
standard.
definitions of these can be found in Practice E355 or Termi-
1.3.1 Alternative units, in common usage, are also provided
nology D4175, or both.
to increase clarity and aid the users of this test method.
3.2 Definitions of Terms Specific to This Standard:
1.4 This standard does not purport to address all of the
3.2.1 electronic pressure control, n—electronic pneumatic
safety concerns, if any, associated with its use. It is the
control of carrier gas flows. Can be flow or pressure pro-
grammed to speed up elution of components.
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.04.0L on Gas Chromatography Methods. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2023. Published November 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2011. Last previous edition approved in 2019 as D7754 – 19. Standards volume information, refer to the standard’s Document Summary page on
DOI:10.1520/D7754-23. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7754 − 23
3.2.2 flame ionization detector (FID), n—detector used to 4. Summary of Test Method
analyze the components eluting from the column.
4.1 An appropriate internal standard of a product that is not
3.2.3 fluidic switch, n—device that reverses the directional
present in refinery streams, such as 1,2-dimethoxy ethane
flow in a union T altering the pressure at the midpoint. In its
(1,2-DME), is added to the sample, which is then introduced
simplest design it is also known as a Dean Switch.
into a gas chromatograph equipped with two columns and a
4-port switching valve. The sample first passes onto an apolar
3.2.4 inlet, n—capillary split/splitless inlet system operated
(non-polar) polydimethylsiloxane WCOT column that per-
in the split mode is recommended. Operate the inlet within its
forms a pre-separation of the trace oxygenates and elutes
linear range.
unwanted high boiling hydrocarbons to vent. The oxygenates
3.2.4.1 split ratio, n— in capillary gas chromatography, the
and the DME are transferred to the analytical oxygen selective
ratio of the total flow of carrier gas to the sample inlet versus
column by the switching valve. While the oxygenates and the
the flow of the carrier gas to the capillary column is expressed
DME are eluting from the analytical column, the inlet’s carrier
by:
gas is used to elute the hydrocarbons from the pre-column to
Split ratio 5 ~S1C!/C (1)
yield a stable baseline for the next analysis. The auxiliary
pressure controller is used to provide carrier gas to the
where:
analytical column during the analysis.
S = flow rate at the splitter vent, and
C = flow rate at the column outlet. 4.2 The eluted components Table 1 are detected by one or
two flame ionization detectors. In the single detector Configu-
3.2.5 low volume connector, n—special union for connect-
ration A (Fig. 1), only the components eluting from the
ing two lengths of tubing 1.6 mm inside diameter and smaller.
analytical column are analyzed. In the two detector Configu-
Sometimes this is referred to as zero dead volume union.
ration B (Fig. 2), detector one is used to monitor the apolar
3.2.6 multidimensional gas chromatography, n—gas chro-
elution and aid in setting “heart-cut” times for specific oxy-
matographic technique where using hardware (valves, pressure
genates while the second detector is used to monitor the
switches, etc.) in which selected components from one column
analytical column elution and also for the quantitation of the
(primary column) are transferred to a secondary column
oxygenates. The second detector response is proportional to the
differing in characteristics (film thickness, polarity, capacity,
oxygenates and DME components concentration. The signal is
etc.) from the first column.
recorded, the peak areas are measured, and the concentration of
3.2.7 WCOT column, n—wall-coated open tubular, a type of
each oxygenate is calculated with reference to the internal
capillary gas chromatographic column prepared by coating the
standard.
inside of the capillary wall with a specified thin film of
4.3 Alternatively, a fluidic switching system, Configuration
stationary phase. The coatings used are either 100 % polydi-
C (Figs. 3 and 4) may be used instead of valve switching. In
methyl siloxane or 5 % phenyl-polydimethylsiloxane.
this system, the two columns are joined by a zero dead volume
3.2.7.1 apolar column, n—polydimethylsiloxane nonpolar
(ZDV) tee purged by an auxiliary carrier source. At injection,
column used as a pre-column.
the auxiliary flow is low, and the inlet flow is sufficient so that
3.2.7.2 PLOT column-oxygen selective, n—porous-layer at the midpoint where the two columns join, the flow is the
open tubular which is an oxygenate selective capillary gas- required flow to transfer the oxygenates to the PLOT column.
solid chromatographic column. It is used as an analytical Thus, there is forward flow through the pre-column and the
column. analytical column. Once the oxygenates have passed through to
A
TABLE 1 Component List with Retention and Calibration Characteristics for WCOT/PLOT Column Set Using Conditions of Table 2
Component RT (min) Mol Wt BP (°C) Slope y-Int Corr. Coef.
ETBE 12.7 102.2 70 to 72 1.919 –0.02 0.999
MTBE 12.8 88.2 55 to 56 1.689 0.01 0.999
DIPE 12.9 102.2 68 to 69 2.124 –0.06 0.999
TAME 13.6 102.2 85 to 86 2.023 –0.02 0.999
Methanol 15.6 32.0 65 0.779 –0.09 0.997
Ethanol 18.7 46.1 78 1.352 0.19 0.999
iso-Propanol 22.2 60.1 81 to 83 1.504 –0.06 0.999
n-Propanol 22.2 60.1 97 . . .
t-Butanol 23.8 74.1 82 1.951 –0.12 0.999
s-Butanol 23.8 74.1 98 . . .
iso-Butanol 23.8 74.1 117 . . .
n-Butanol 24.4 74.1 118 1.906 –0.05 0.999
tert-Pentanol 25.1 88.1 102 2.148 –0.04 0.998
1,2-DME 26.0 90.1 85 . . .
A
For coeluting compounds the response is assigned to the first peak listed. Values may be different for different instruments.
D7754 − 23
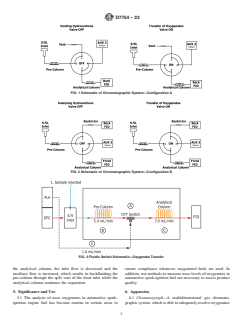
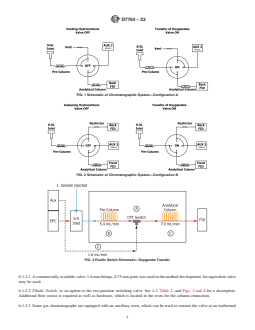
FIG. 1 Schematic of Chromatographic System—Configuration A
FIG. 2 Schematic of Chromatographic System—Configuration B
FIG. 3 Fluidic Switch Schematic—Oxygenate Transfer
the analytical column, the inlet flow is decreased and the ensure compliance whenever oxygenated fuels are used. In
auxiliary flow is increased, which results in backflushing the addition, test methods to measure trace levels of oxygenates in
pre-column through the split vent of the front inlet while the automotive spark-ignition fuel are necessary to assess product
analytical column continues the separation. quality.
5. Significance and Use 6. Apparatus
5.1 The analysis of trace oxygenates in automotive spark- 6.1 Chromatograph—A multidimensional gas chromato-
ignition engine fuel has become routine in certain areas to graphic system, which is able to adequately resolve oxygenates
D7754 − 23
FIG. 4 Fluidic Switch Schematic—Pre-column Backflush
and an internal standard and to eliminate hydrocarbon, as well Fig. 4. Carrier gas flow controllers (EPC) shall be capable of
as other interferences, is used for these analyses. The instru- precise control where the flow rates are low. Pressure control
ment is to be configured to operate using the approximate devices and gages shall be capable of precise control for the
conditions listed in Table 2. The system requires a column typical pressures required.
switching mechanism equivalent to Fig. 1 or Fig. 2 if using a 6.1.1 Detector—Two-flame ionization detectors are prefer-
valve system. If using a fluidic system then the fluidic switch ably used (Configuration B), although the analysis can be
and auxiliary flow control are required as shown in Fig. 3 and performed using only one detector (Configuration A and C).
TABLE 2 Chromatographic Conditions
Configuration Switching Valve Fluidic Switch
Carrier Gas Helium Helium
Injection Volume 1.0 μL 1.0 μL
Inlet: Split/Splitless (Split mode) Split/Splitless (Split mode)
Temperature 250 °C 250 °C
Split Ratio 10:1 10:1
A
Pressure 51.7 kPa, Constant Pressure 17.8 kPa, Flow Program Mode
Columns and Flows
Pre-column 30 m by 0.53 mm by 5.0 μm PDMS 15 m by 0.53 mm by 1.5 μm 5 % phenyl PDMS
7.5 mL/min @ 60 °C Initial Flow: 5.4 mL/min @ 60 °C
Hold for 1.5 min
Ramp: 90 mL/min to -5 mL/min
Hold until end of run
Analytical Column 10 m by 0.53 mm by 10 μm Oxygen Selective 10 m by 0.53 mm by 10 μm Oxygen Selective
7.5 mL/min@ 60 °C 7.0 mL/min constant flow
Oven:
Initial Temperature 60 °C 60 °C
Initial Hold 6.0 min 6.0 min
Ramp 1 10 °C ⁄min 10 °C ⁄min
Final Temperature 150 °C 150 °C
Final Hold 5.0 min 5.0 min
Ramp 2 10 °C ⁄min 10 °C ⁄min
Final Temperature 220 °C 220 °C
Final Hold 3.0 min 3.0 min
Total Time 30 min 30 min
Detector: FID FID
Temperature 275 °C 275 °C
Hydrogen 40 mL/min 40 mL/min
Air 450 mL/min 450 mL/min
Make-up (N2) 10 mL/min 10 mL/min
Valve Temperature 150 °C N/A
Auxiliary Pressure 73.1 kPa 10.3 kPa
Vent Restrictor 76 cm in length N/A
0.16 cm (O.D.) and 0.25 mm (I.D.) SS
Default Valve Times (for complete analysis):
Initial OFF N/A
0.50 min ON N/A
4.50 min OFF N/A
A
For Configuration A valve timing determination, 9.2, set the inlet pressure to 34.5 kPa.
D7754 − 23
The system shall have sufficient sensitivity and stability to replaceable glass deactivated liner (single-taper style) with
obtain a signal-to-noise ratio of at least 5 to 1 for a 1 mg ⁄kg deactivated glass wool at the bottom to retain non-vaporized
concentration of any oxygenate. In the fluidic system only one components). Split injection is necessary to maintain the actual
detector is used. chromatographed sample size within the limits of column and
6.1.2 Switching Valve—A switching valve, to be located detector efficiency and linearity.
within the gas chromatographic column oven or separate oven,
6.1.3.1 A microliter automatic syringe injector is used for
capable of performing the functions described in 9.2 and
introducing representative samples into the gas chromato-
illustrated in Fig. 1. The valve shall be of low volume design
graphic inlet and for adequate repeatability.
and not contribute significantly to chromatographic deteriora-
6.2 Data Acquisition System:
tion. Alternatively a Deans switching arrangement can also be
6.2.1 Computer—A data acquisition system containing a
used as shown in Fig. 3 and Fig. 4.
computer and data acquisition software is required.
6.1.2.1 A commercially available valve: 1.6 mm fittings,
6.2.2 Integrator—Alternatively, an integrator can be used to
0.75 mm ports was used in the method development. An
measure peak areas and to perform the analytical calculations.
equivalent valve may be used.
6.1.2.2 Fluidic Switch, as an option to the two-position
6.3 Column Class:
switching valve. See 4.3, Table 2, and Figs. 3 and 4 for a
6.3.1 Apolar (Non-polar) Pre-Column—This column per-
description. Additional flow source is required as well as
forms a pre-separation of the oxygenates and internal standard
hardware, which is located in the oven for the column
from hydrocarbons in the same boiling point range. Unless a
connection.
separate auxiliary oven is provided for it, the apolar column
6.1.2.3 Some gas chromatographs are equipped with an
shall perform at the same temperature as the polar column
auxiliary oven, which can be used to contain the valve at an
does.
isothermal temperature. In such a configuration, the two
6.3.1.1 WCOT Methyl Silicone Pre-Column—30 m long by
capillary columns are located in the main oven and connected
0.53 mm inside diameter fused silica column with a 5 μm film
to the valve by using low dead volume and inert stainless steel
thickness of cross-linked polydimethylsiloxane. With fluidic
tubing terminated in the GC oven.
switch (Configuration C) a 30 m long by 0.53 mm with a
6.1.2.4 An automatic valve switching device is used to
1.5 μm 5 % phenyl polydimethyl siloxane is recommended.
ensure repeatable switching times. Such a device is synchro-
6.3.2 Polar Analytical Column—Any column with equiva-
nized with injection and data collection times. For the pressure
lent or better chromatographic efficiency and selectivity to that
switching approach, automatic precise and stable pressure
described in 6.3.1 and which separation efficiency is illustrated
control shall be used. Fluidic systems require both a fluidic
in Figs. 5 and 6 can be used.
switch and a programmable auxiliary pressure source to
maintain and program flows. 6.3.2.1 LowOx or GS OxyPLOT Polar Analytical Column—
6.1.3 Injection System—The chromatograph is to be 10 m long by 0.53 mm inside diameter fused silica PLOT
equipped with a heated splitting-type inlet device containing a column with a 10 μm film thickness. These columns were used
FIG. 5 Oxygenate Elution Pattern, from WCOT Pre-column Only
D7754 − 23
NOTE 1—Calibration Standard—250 mg ⁄kg oxygenates, 7.5 % ethanol, and 200 mg/kg internal standard in isooctane.
FIG. 6 Oxygenate Elution Pattern from WCOT/PLOT Column Set Using Conditions in Table 2
in the method development to provide the precision and bias cut times (see Appendix X1). (Warning—isooctane and cy-
data referred to in Section 14. clohexane are flammable and may be harmful if inhaled. High
concentrations may cause unconsciousness or death).
7. Reagents and Materials
7.4 1,2-Dimethoxyethane (1,2-DME or ethylene glycol dim-
7.1 Gases:
ethyl ether)—Used as the internal standard. Use Reagent or
7.1.1 Helium, carrier gas, a minimum purity of 99.995 % is
Chromatography grade.
required. Oxygen scrubbers are recommended to safeguard the
WCOT columns. 8. Sampling
7.1.2 For the FID, hydrogen (99.9995 % with air as the
8.1 Every effort should be made to ensure that the sample is
remainder) and nitrogen (99.995 %, as make up) are used.
representative of the finished automotive spark-ignition fuel
(Warning—Observe high pressure precautions with all com-
from which it is taken. The use of multiple samples that are
pressed gases. Observe flammable gas precautions with hydro-
mixed or composite sampling is recommended. The use of
gen.)
epoxy-lined cans are recommended for storage or shipping of
7.2 Standards for Calibration and Identification—Standards the sample, or both. Follow the recommendations of Practice
D4057, when obtaining samples from bulk storage or pipelines.
of oxygenates and the internal standard are required for
establishing identification by retention time as well as calibra- Samples that contain free layer of water will require special
tion for quantitative analysis. These materials shall be of treatment. For the latter samples, it may be necessary to
known purity and free of the other components to be analyzed. separate and analyze the water and hydrocarbon phases sepa-
(Warning—These materials are flammable and can be harmful rately. The water phase may be determined from a separate
or fatal if ingested or inhaled.). The following oxygenates: method. For such analysis, it is necessary to know the amount
ethanol, methyl-tertiary butyl ether (MTBE), ethyl-tertiary of water and hydrocarbon phases to determine a total methanol
butyl ether (ETBE), diisopropyl ether (DIPE), methanol, content for the sample.
tertiary-amyl methyl ether (TAME), n-propanol, i-propanol,
8.2 Prior to analysis, allow the sample container as received
n-butanol, i-butanol, tert-butyl alcohol, sec-butyl alcohol, and
to equilibrate to ambient temperature.
tert-pentanol are to be used with the highest purity available
(98 % to 99 %). Ethanol is usually not measured as a trace 9. Preparation of Apparatus and Establishment of
sample component. Conditions
7.3 isooctane, or 2,2,4-Trimethylpentane—Used for prepa- 9.1 Determine Configuration—Refer to 4.2 to determine
ration of calibration standards and dilution of automotive which configuration the chromatographic system is designed
spark-ignition fuel samples. In some cases, cyclohexane may for the analysis. For a valve configuration the dual detector
be used provided it meets all of the requirements of the Configuration B (Fig. 2) is recommended due to the fact that
method. Using cyclohexane, since it elutes in the cut window the system can be assembled with no modifications to deter-
of the oxygenates, may cause difficulty in finding individual mine the cut times from the pre-column.
D7754 − 23
9.1.1 Figs. 1 and 2 are plumbing schematics for using a system that comes in contact with the sample; otherwise, peak
commercially available switching valve. This system is de- broadening will occur.
scribed primarily in the following sections–Configurations A 9.3.2 Vent Restrictor—The vent restrictor is intended to
and B. simulate the restriction caused by the PLOT column on the
pre-column. This is needed to ensure accurate cut time deter-
9.1.2 Figs. 3 and 4 are the plumbing schematics for using a
mination while the valve is in the OFF position (see Fig. 2). A
commercially available fluidic switching system.
piece of stainless steel tubing, 70 cm in length, 0.16 cm O.D.
9.2 Configuration A (Single Detector Configuration):
and having 0.254 mm I.D., will approximate the column
9.2.1 Assembly—Refer to Fig. 1. First connect the polydi-
resistance.
methylsiloxane pre-column (6.3.1) to the detector directly to
9.3.3 Conditions—Establish the operating conditions listed
set the cut time (9.2.4) using low-volume connectors and inert
in Table 2. This gives example conditions for the columns
narrow bore tubing. It is important to minimize the volume of
systems used in the development of this test method. Modifi-
the chromatographic system that comes in contact with the
cations to column lengths etc. may require different operating
sample; otherwise, peak broadening will occur.
conditions. Check the system for leaks before proceeding
9.2.2 Vent Restrictor—The vent restrictor is intended to
further. Condition the system overnight before proceeding at an
simulate the restriction caused by the PLOT column on the
oven temperature of 200 °C.
pre-column. This is needed to ensure accurate cut time deter-
9.3.4 Setting Valve Times—With the valve in the OFF
mination while the valve is in the OFF position. A piece of
position determine first the retention time of the oxygenates
stainless steel tubing, 70 cm in length, 0.16 cm O.D. and
through the apolar. Subsequently the same is repeated but with
having 0.254 mm I.D., will approximate the column resistance.
the valve in the ON position.
9.2.3 Conditions—Establish the operating conditions listed
9.3.5 Switch the valve to the OFF position to monitor the
in Table 2. This gives example conditions for the columns
pre-column only. Inject 1 μL of a ~300 mg ⁄kg solution without
systems used in the development of this method. With the
ethanol or internal standard. To prepare this, add 20 μL of the
pre-column connected to the FID directly, the inlet pressure
solution made in 10.3 to 5 mL of isooctane. Record the
should be adjusted to ~31.0 kPa. This will set the column flow
chromatogram. Identify the peaks for each oxygenate using
to approximately 6.8 mL ⁄min at 60 °C. This is necessary since
Fig. 5. From this retention time data, set the oxygenate transfer
there is no simulation for the restriction caused by the PLOT
valve time ON to 0.5 min before the methanol starts eluting,
column when the pre-column is installed directly to the FID.
and valve time OFF to 0.5 min after the TAME peak returns to
Modifications to column lengths etc. may require different
baseline. The times should be incorporated into the analysis
operating conditions. Check the system for leaks before pro-
method before calibration is begun.
ceeding further. Condition the system overnight before pro-
9.4 Configuration C (Single Detector Configuration Using a
ceeding.
Fluidic Switch System:
9.2.4 Setting Valve Times—Once Configuration A is set up
9.4.1 The pre-column and the analytical column are linked
and the pre-column is connected to the detector, determine the
by fluidic switch (CFT) purged union (see Fig. 3). The iInlet
retention time of the oxygenates through the pre-column.
EPC delivers 5.4 mL ⁄min to both columns. The AUX EPC
9.2.5 Inject 1 μL of a ~300 mg ⁄kg solution without ethanol
delivers 1.6 mL ⁄min to the capillary flow technology (CFT)
or internal standard. To prepare this, add 20 μL of the solution
union. This increases the flow to the analytical column to
made in 10.3 to 5 mL of isooctane. Record the chromatogram,
7.0 mL ⁄min. Under these conditions the oxygenates are trans-
and identify the peaks for each oxygenate using Fig. 5. From
ferred in about 1.5 min.
this retention time data, set the oxygenate transfer valve time
9.4.2 High boiling point compounds are retained in the
ON to 0.5 min before the methanol starts eluting, and valve
pre-column. Oxygenates and low boiling point hydrocarbons
time OFF to 0.5 min after the TAME peak returns to baseline.
elute into analytical column. Oxygenates are trapped at the
The times should be incorporated into the analysis method
front of the analytical column. After TAME has eluted to the
before calibration is begun.
analytical column, the pre-column flow is reversed.
9.2.6 Reassemble the system by reinstalling the PLOT
9.4.3 Inlet EPC flow is reduced to 4 mL ⁄min (Fig. 4) and
column to the diagram in Fig. 1 and Table 2 using low-volume AUX EPC flow is increased to 11 mL ⁄min. Gas flow through
connectors and inert narrow bore tubing. It is important to the fluidic switch backflushes the pre-column in order to send
minimize the volume of the connections into and from the high boiling compounds out the front inlet split vent. Oven
valve. Proceed to place the Valve in the ON position so that the temperature program begins and oxygenates are separated on
apolar column and PLOT column are in series. Inject the the analytical column. A typical chromatogram using this
sample as described in 9.2.5 and leave the valve in the ON system is shown in Fig. 7.
position as determined in 9.2.5. Using the times determined in
9.5 Verification of Detectability—Inject a 5 mg ⁄kg calibra-
9.2.5 transfer the oxygenates to the PLOT column.
tion solution and ensure that a signal/noise level of at least 5 is
observed. Adjustment of the split ratio may be needed depend-
9.3 Configuration B (Dual Detector Configuration):
ing on the detector. See Fig. 8. The peaks for this level
9.3.1 Assembly—For Configuration B, connect the WCOT
(5 mg ⁄kg) have a height of about 2 pA.
and PLOT columns to the valve system as shown in Fig. 2
using low-volume connectors and inert narrow bore tubing. It 9.6 Conditioning—To protect the PLOT column, avoid in-
is important to minimize the volume of the chromatographic jecting samples until the valve times are properly optimized
D7754 − 23
NOTE 1—Conditions in Table 2. Calibration Standard – mg/kg internal standard in isooctane.
FIG. 7 Oxygenate Elution Pattern from WCOT/PLOT Column Set Using the Fluidic Switch System
FIG. 8 Signal to Noise Verification—5 mg/kg Oxygenate/1.0 % Ethanol Calibration Standard with Conditions of Table 2.
usi
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7754 − 19 D7754 − 23
Standard Test Method for
Determination of Trace Oxygenates in Automotive Spark-
Ignition Engine Fuel by Multidimensional Gas
Chromatography
This standard is issued under the fixed designation D7754; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of trace oxygenates in automotive spark-ignition engine fuel. The method used is
a multidimensional gas chromatographic method using 1,2-dimethoxy ethane as the internal standard. The oxygenates that are
analyzed are: methyl-tertiary butyl ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl ether (DIPE), methanol,
tertiary-amyl methyl ether (TAME), n-propanol, i-propanol, n-butanol, i-butanol, tert-butyl alcohol, sec-butyl alcohol, and
tert-pentanol. Ethanol is usually not measured as a trace oxygenate since ethanol can be used as the main oxygenate compound
in finished automotive spark-ignition fuels such as reformulated automotive spark-ignition fuels. The concentration range of the
oxygenates covered in the ILS study was from 10 mg ⁄kg to 2000 mg ⁄kg. In addition this method is also suitable for the
measurement of the C5 isomeric alcohols (2-methyl-1-butanol, 2-methyl-2-butanol) present from the fermentation of ethanol.
1.2 The ethanol blending concentration for which this test method applies ranges from 1 % to 15% by volume. Higher
concentrations of ethanol coelute with methanol in the analytical column. Lower levels of ethanol, similar to the other oxygenate,
can be calibrated and analyzed also. If higher ethanol concentrations are expected, the window cutting technique can be used to
avoid ethanol from entering the analytical column and interfere with the determination of the other oxygenates of interest. Refer
to Appendix X1 for details.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Alternative units, in common usage, are also provided to increase clarity and aid the users of this test method.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.04.0L on Gas Chromatography Methods.
Current edition approved Nov. 1, 2019Nov. 1, 2023. Published December 2019November 2023. Originally approved in 2011. Last previous edition approved in 20162019
as D7754 – 16.D7754 – 19. DOI:10.1520/D7754-19.DOI:10.1520/D7754-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7754 − 23
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4175 Terminology Relating to Petroleum Products, Liquid Fuels, and Lubricants
D4307 Practice for Preparation of Liquid Blends for Use as Analytical Standards
D4815 Test Method for Determination of MTBE, ETBE, TAME, DIPE, tertiary-Amyl Alcohol and C to C Alcohols in
1 4
Gasoline by Gas Chromatography
D6304 Test Method for Determination of Water in Petroleum Products, Lubricating Oils, and Additives by Coulometric Karl
Fischer Titration
E355 Practice for Gas Chromatography Terms and Relationships
3. Terminology
3.1 Definitions:
3.1.1 This test method makes reference to common gas chromatographic procedures, terms, and relationships. Detailed definitions
of these can be found in Practice E355 or Terminology D4175, or both.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 electronic pressure control, n—electronic pneumatic control of carrier gas flows. Can be flow or pressure programmed to
speed up elution of components.
3.2.2 flame ionization detector (FID), n—detector used to analyze the components eluting from the column.
3.2.3 fluidic switch, n—device that reverses the directional flow in a union T altering the pressure at the midpoint. In its simplest
design it is also known as a Dean Switch.
3.2.4 inlet, n—capillary split/splitless inlet system operated in the split mode is recommended. Operate the inlet within its linear
range.
3.2.4.1 split ratio, n— in capillary gas chromatography, the ratio of the total flow of carrier gas to the sample inlet versus the
flow of the carrier gas to the capillary column is expressed by:
Split ratio 5 ~S1C!/C (1)
where:
S = flow rate at the splitter vent, and
C = flow rate at the column outlet.
3.2.5 low volume connector, n—special union for connecting two lengths of tubing 1.6 mm inside diameter and smaller.
Sometimes this is referred to as zero dead volume union.
3.2.6 multidimensional gas chromatography, n—gas chromatographic technique where using hardware (valves, pressure switches,
etc.) in which selected components from one column (primary column) are transferred to a secondary column differing in
characteristics (film thickness, polarity, capacity, etc.) from the first column.
3.2.7 WCOT column, n—wall-coated open tubular, a type of capillary gas chromatographic column prepared by coating the inside
of the capillary wall with a specified thin film of stationary phase. The coatings used are either 100 % polydimethyl siloxane or
5 % phenyl-polydimethylsiloxane.
3.2.7.1 apolar column, n—polydimethylsiloxane nonpolar column used as a pre-column.
3.2.7.2 PLOT column-oxygen selective, n—porous-layer open tubular which is an oxygenate selective capillary gas-solid
chromatographic column. It is used as an analytical column.
4. Summary of Test Method
4.1 An appropriate internal standard of a product that is not present in refinery streams, such as 1,2-dimethoxy ethane (1,2-DME),
is added to the sample, which is then introduced into a gas chromatograph equipped with two columns and a 4-port switching
valve. The sample first passes onto an apolar (non-polar) polydimethylsiloxane WCOT column that performs a pre-separation of
the trace oxygenates and elutes unwanted high boiling hydrocarbons to vent. The oxygenates and the DME are transferred to the
analytical oxygen selective column by the switching valve. While the oxygenates and the DME are eluting from the analytical
D7754 − 23
column, the inlet’s carrier gas is used to elute the hydrocarbons from the pre-column to yield a stable baseline for the next analysis.
The auxiliary pressure controller is used to provide carrier gas to the analytical column during the analysis.
4.2 The eluted components Table 1 are detected by one or two flame ionization detectors. In the single detector Configuration A
(Fig. 1), only the components eluting from the analytical column are analyzed. In the two detector Configuration B (Fig. 2),
detector one is used to monitor the apolar elution and aid in setting “heart-cut” times for specific oxygenates while the second
detector is used to monitor the analytical column elution and also for the quantitation of the oxygenates. The second detector
response is proportional to the oxygenates and DME components concentration. The signal is recorded, the peak areas are
measured, and the concentration of each oxygenate is calculated with reference to the internal standard.
4.3 Alternatively, a fluidic switching system, Configuration C (Figs. 3 and 4) may be used instead of valve switching. In this
system, the two columns are joined by a zero dead volume (ZDV) tee purged by an auxiliary carrier source. At injection, the
auxiliary flow is low, and the inlet flow is sufficient so that at the midpoint where the two columns join, the flow is the required
flow to transfer the oxygenates to the PLOT column. Thus, there is forward flow through the pre-column and the analytical column.
Once the oxygenates have passed through to the analytical column, the inlet flow is decreased and the auxiliary flow is increased,
which results in backflushing the pre-column through the split vent of the front inlet while the analytical column continues the
separation.
5. Significance and Use
5.1 The analysis of trace oxygenates in automotive spark-ignition engine fuel has become routine in certain areas to ensure
compliance whenever oxygenated fuels are used. In addition, test methods to measure trace levels of oxygenates in automotive
spark-ignition fuel are necessary to assess product quality.
6. Apparatus
6.1 Chromatograph—A multidimensional gas chromatographic system, which is able to adequately resolve oxygenates and an
internal standard and to eliminate hydrocarbon, as well as other interferences, is used for these analyses. The instrument is to be
configured to operate using the approximate conditions listed in Table 2. The system requires a column switching mechanism
equivalent to Fig. 1 or Fig. 2 if using a valve system. If using a fluidic system then the fluidic switch and auxiliary flow control
are required as shown in Fig. 3 and Fig. 4. Carrier gas flow controllers (EPC) shall be capable of precise control where the flow
rates are low. Pressure control devices and gages shall be capable of precise control for the typical pressures required.
6.1.1 Detector—Two-flame ionization detectors are preferably used (Configuration B), although the analysis can be performed
using only one detector (Configuration A and C). The system shall have sufficient sensitivity and stability to obtain a signal-to-noise
ratio of at least 5 to 1 for a 1 mg ⁄kg concentration of any oxygenate. In the fluidic system only one detector is used.
6.1.2 Switching Valve—A switching valve, to be located within the gas chromatographic column oven or separate oven, capable
of performing the functions described in 9.2 and illustrated in Fig. 1. The valve shall be of low volume design and not contribute
significantly to chromatographic deterioration. Alternatively a Deans switching arrangement can also be used as shown in Fig. 3
and Fig. 4.
A
TABLE 1 Component List with Retention and Calibration Characteristics for WCOT/PLOT Column Set Using Conditions of Table 2
Component RT (min) Mol Wt BP (°C) Slope y-Int Corr. Coef.
ETBE 12.7 102.2 70 to 72 1.919 –0.02 0.999
MTBE 12.8 88.2 55 to 56 1.689 0.01 0.999
DIPE 12.9 102.2 68 to 69 2.124 –0.06 0.999
TAME 13.6 102.2 85 to 86 2.023 –0.02 0.999
Methanol 15.6 32.0 65 0.779 –0.09 0.997
Ethanol 18.7 46.1 78 1.352 0.19 0.999
iso-Propanol 22.2 60.1 81 to 83 1.504 –0.06 0.999
n-Propanol 22.2 60.1 97 . . .
t-Butanol 23.8 74.1 82 1.951 –0.12 0.999
s-Butanol 23.8 74.1 98 . . .
iso-Butanol 23.8 74.1 117 . . .
n-Butanol 24.4 74.1 118 1.906 –0.05 0.999
tert-Pentanol 25.1 88.1 102 2.148 –0.04 0.998
1,2-DME 26.0 90.1 85 . . .
A
For coeluting compounds the response is assigned to the first peak listed. Values may be different for different instruments.
D7754 − 23
FIG. 1 Schematic of Chromatographic System—Configuration A
FIG. 2 Schematic of Chromatographic System—Configuration B
FIG. 3 Fluidic Switch Schematic—Oxygenate Transfer
6.1.2.1 A commercially available valve: 1.6 mm fittings, 0.75 mm ports was used in the method development. An equivalent valve
may be used.
6.1.2.2 Fluidic Switch, as an option to the two-position switching valve. See 4.3, Table 2, and Figs. 3 and 4 for a description.
Additional flow source is required as well as hardware, which is located in the oven for the column connection.
6.1.2.3 Some gas chromatographs are equipped with an auxiliary oven, which can be used to contain the valve at an isothermal
D7754 − 23
FIG. 4 Fluidic Switch Schematic—Pre-column Backflush
TABLE 2 Chromatographic Conditions
Configuration Switching Valve Fluidic Switch
Carrier Gas Helium Helium
Injection Volume 1.0 μL 1.0 μL
Inlet: Split/Splitless (Split mode) Split/Splitless (Split mode)
Temperature 250 °C 250 °C
Split Ratio 10:1 10:1
A
Pressure 51.7 kPa, Constant Pressure 17.8 kPa, Flow Program Mode
Columns and Flows
Pre-column 30 m by 0.53 mm by 5.0 μm PDMS 15 m by 0.53 mm by 1.5 μm 5 % phenyl PDMS
7.5 mL/min @ 60 °C Initial Flow: 5.4 mL/min @ 60 °C
Hold for 1.5 min
Ramp: 90 mL/min to -5 mL/min
Hold until end of run
Analytical Column 10 m by 0.53 mm by 10 μm Oxygen Selective 10 m by 0.53 mm by 10 μm Oxygen Selective
7.5 mL/min@ 60 °C 7.0 mL/min constant flow
Oven:
Initial Temperature 60 °C 60 °C
Initial Hold 6.0 min 6.0 min
Ramp 1 10 °C ⁄min 10 °C ⁄min
Final Temperature 150 °C 150 °C
Final Hold 5.0 min 5.0 min
Ramp 2 10 °C ⁄min 10 °C ⁄min
Final Temperature 220 °C 220 °C
Final Hold 3.0 min 3.0 min
Total Time 30 min 30 min
Detector: FID FID
Temperature 275 °C 275 °C
Hydrogen 40 mL/min 40 mL/min
Air 450 mL/min 450 mL/min
Make-up (N2) 10 mL/min 10 mL/min
Valve Temperature 150 °C N/A
Auxiliary Pressure 73.1 kPa 10.3 kPa
Vent Restrictor 76 cm in length N/A
0.16 cm (O.D.) and 0.25 mm (I.D.) SS
Default Valve Times (for complete analysis):
Initial OFF N/A
0.50 min ON N/A
4.50 min OFF N/A
A
For Configuration A valve timing determination, 9.2, set the inlet pressure to 34.5 kPa.
temperature. In such a configuration, the two capillary columns are located in the main oven and connected to the valve by using
low dead volume and inert stainless steel tubing terminated in the GC oven.
6.1.2.4 An automatic valve switching device is used to ensure repeatable switching times. Such a device is synchronized with
injection and data collection times. For the pressure switching approach, automatic precise and stable pressure control shall be
used. Fluidic systems require both a fluidic switch and a programmable auxiliary pressure source to maintain and program flows.
6.1.3 Injection System—The chromatograph is to be equipped with a heated splitting-type inlet device containing a replaceable
D7754 − 23
glass deactivated liner (single-taper style) with deactivated glass wool at the bottom to retain non-vaporized components). Split
injection is necessary to maintain the actual chromatographed sample size within the limits of column and detector efficiency and
linearity.
6.1.3.1 A microliter automatic syringe injector is used for introducing representative samples into the gas chromatographic inlet
and for adequate repeatability.
6.2 Data Acquisition System:
6.2.1 Computer—A data acquisition system containing a computer and data acquisition software is required.
6.2.2 Integrator—Alternatively, an integrator can be used to measure peak areas and to perform the analytical calculations.
6.3 Column Class:
6.3.1 Apolar (Non-polar) Pre-Column—This column performs a pre-separation of the oxygenates and internal standard from
hydrocarbons in the same boiling point range. Unless a separate auxiliary oven is provided for it, the apolar column shall perform
at the same temperature as the polar column does.
6.3.1.1 WCOT Methyl Silicone Pre-Column—30 m long by 0.53 mm inside diameter fused silica column with a 5 μm film
thickness of cross-linked polydimethylsiloxane. With fluidic switch (Configuration C) a 30 m long by 0.53 mm with a 1.5 μm 5 %
phenyl polydimethyl siloxane is recommended.
6.3.2 Polar Analytical Column—Any column with equivalent or better chromatographic efficiency and selectivity to that described
in 6.3.1 and which separation efficiency is illustrated in Figs. 5 and 6 can be used.
6.3.2.1 LowOx or GS OxyPLOT Polar Analytical Column—10 m long by 0.53 mm inside diameter fused silica PLOT column with
a 10 μm film thickness. These columns were used in the method development to provide the precision and bias data referred to
in Section 14.
7. Reagents and Materials
7.1 Gases:
FIG. 5 Oxygenate Elution Pattern, from WCOT Pre-column Only
D7754 − 23
NOTE 1—Calibration Standard—250 mg ⁄kg oxygenates, 7.5 % ethanol, and 200 mg/kg internal standard in isooctane.
FIG. 6 Oxygenate Elution Pattern from WCOT/PLOT Column Set Using Conditions in Table 2
7.1.1 Helium, carrier gas, a minimum purity of 99.995 % is required. Oxygen scrubbers are recommended to safeguard the WCOT
columns.
7.1.2 For the FID, hydrogen (99.9995 % with air as the remainder) and nitrogen (99.995 %, as make up) are used.
(Warning—Observe high pressure precautions with all compressed gases. Observe flammable gas precautions with hydrogen.)
7.2 Standards for Calibration and Identification—Standards of oxygenates and the internal standard are required for establishing
identification by retention time as well as calibration for quantitative analysis. These materials shall be of known purity and free
of the other components to be analyzed. (Warning—These materials are flammable and can be harmful or fatal if ingested or
inhaled.). The following oxygenates: ethanol, methyl-tertiary butyl ether (MTBE), ethyl-tertiary butyl ether (ETBE), diisopropyl
ether (DIPE), methanol, tertiary-amyl methyl ether (TAME), n-propanol, i-propanol, n-butanol, i-butanol, tert-butyl alcohol,
sec-butyl alcohol, and tert-pentanol are to be used with the highest purity available (98 % to 99 %). Ethanol is usually not measured
as a trace sample component.
7.3 isooctane, or 2,2,4-Trimethylpentane—Used for preparation of calibration standards and dilution of automotive spark-ignition
fuel samples. In some cases, cyclohexane may be used provided it meets all of the requirements of the method. Using cyclohexane,
since it elutes in the cut window of the oxygenates, may cause difficulty in finding individual cut times (see Appendix X1).
(Warning—isooctane and cyclohexane are flammable and may be harmful if inhaled. High concentrations may cause
unconsciousness or death).
7.4 1,2-Dimethoxyethane (1,2-DME or ethylene glycol dimethyl ether)—Used as the internal standard. Use Reagent or
Chromatography grade.
8. Sampling
8.1 Every effort should be made to ensure that the sample is representative of the finished automotive spark-ignition fuel from
which it is taken. The use of multiple samples that are mixed or composite sampling is recommended. The use of epoxy-lined cans
are recommended for storage or shipping of the sample, or both. Follow the recommendations of Practice D4057, when obtaining
samples from bulk storage or pipelines. Samples that contain free layer of water will require special treatment. For the latter
samples, it may be necessary to separate and analyze the water and hydrocarbon phases separately. The water phase may be
D7754 − 23
determined from a separate method. For such analysis, it is necessary to know the amount of water and hydrocarbon phases to
determine a total methanol content for the sample.
8.2 Prior to analysis, allow the sample container as received to equilibrate to ambient temperature.
9. Preparation of Apparatus and Establishment of Conditions
9.1 Determine Configuration—Refer to 4.2 to determine which configuration the chromatographic system is designed for the
analysis. For a valve configuration the dual detector Configuration B (Fig. 2) is recommended due to the fact that the system can
be assembled with no modifications to determine the cut times from the pre-column.
9.1.1 Figs. 1 and 2 are plumbing schematics for using a commercially available switching valve. This system is described
primarily in the following sections–Configurations A and B.
9.1.2 Figs. 3 and 4 are the plumbing schematics for using a commercially available fluidic switching system.
9.2 Configuration A (Single Detector Configuration):
9.2.1 Assembly—Refer to Fig. 1. First connect the polydimethylsiloxane pre-column (6.3.1) to the detector directly to set the cut
time (9.2.4) using low-volume connectors and inert narrow bore tubing. It is important to minimize the volume of the
chromatographic system that comes in contact with the sample; otherwise, peak broadening will occur.
9.2.2 Vent Restrictor—The vent restrictor is intended to simulate the restriction caused by the PLOT column on the pre-column.
This is needed to ensure accurate cut time determination while the valve is in the OFF position. A piece of stainless steel tubing,
70 cm in length, 0.16 cm O.D. and having 0.254 mm I.D., will approximate the column resistance.
9.2.3 Conditions—Establish the operating conditions listed in Table 2. This gives example conditions for the columns systems
used in the development of this method. With the pre-column connected to the FID directly, the inlet pressure should be adjusted
to ~31.0 kPa. This will set the column flow to approximately 6.8 mL ⁄min at 60 °C. This is necessary since there is no simulation
for the restriction caused by the PLOT column when the pre-column is installed directly to the FID. Modifications to column
lengths etc. may require different operating conditions. Check the system for leaks before proceeding further. Condition the system
overnight before proceeding.
9.2.4 Setting Valve Times—Once Configuration A is set up and the pre-column is connected to the detector, determine the retention
time of the oxygenates through the pre-column.
9.2.5 Inject 1 μL of a ~300 mg ⁄kg solution without ethanol or internal standard. To prepare this, add 20 μL of the solution made
in 10.3 to 5 mL of isooctane. Record the chromatogram, and identify the peaks for each oxygenate using Fig. 5. From this retention
time data, set the oxygenate transfer valve time ON to 0.5 min before the methanol starts eluting, and valve time OFF to 0.5 min
after the TAME peak returns to baseline. The times should be incorporated into the analysis method before calibration is begun.
9.2.6 Reassemble the system by reinstalling the PLOT column to the diagram in Fig. 1 and Table 2 using low-volume connectors
and inert narrow bore tubing. It is important to minimize the volume of the connections into and from the valve. Proceed to place
the Valve in the ON position so that the apolar column and PLOT column are in series. Inject the sample as described in 9.2.5 and
leave the valve in the ON position as determined in 9.2.5. Using the times determined in 9.2.5 transfer the oxygenates to the PLOT
column.
9.3 Configuration B (Dual Detector Configuration):
9.3.1 Assembly—For Configuration B, connect the WCOT and PLOT columns to the valve system as shown in Fig. 2 using
low-volume connectors and inert narrow bore tubing. It is important to minimize the volume of the chromatographic system that
comes in contact with the sample; otherwise, peak broadening will occur.
9.3.2 Vent Restrictor—The vent restrictor is intended to simulate the restriction caused by the PLOT column on the pre-column.
This is needed to ensure accurate cut time determination while the valve is in the OFF position (see Fig. 2). A piece of stainless
steel tubing, 70 cm in length, 0.16 cm O.D. and having 0.254 mm I.D., will approximate the column resistance.
D7754 − 23
9.3.3 Conditions—Establish the operating conditions listed in Table 2. This gives example conditions for the columns systems
used in the development of this test method. Modifications to column lengths etc. may require different operating conditions. Check
the system for leaks before proceeding further. Condition the system overnight before proceeding at an oven temperature of 200 °C.
9.3.4 Setting Valve Times—With the valve in the OFF position determine first the retention time of the oxygenates through the
apolar. Subsequently the same is repeated but with the valve in the ON position.
9.3.5 Switch the valve to the OFF position to monitor the pre-column only. Inject 1 μL of a ~300 mg ⁄kg solution without ethanol
or internal standard. To prepare this, add 20 μL of the solution made in 10.3 to 5 mL of isooctane. Record the chromatogram.
Identify the peaks for each oxygenate using Fig. 5. From this retention time data, set the oxygenate transfer valve time ON to
0.5 min before the methanol starts eluting, and valve time OFF to 0.5 min after the TAME peak returns to baseline. The times
should be incorporated into the analysis method before calibration is begun.
9.4 Configuration C (Single Detector Configuration Using a Fluidic Switch System:
9.4.1 The pre-column and the analytical column are linked by fluidic switch (CFT) purged union (see Fig. 3). The iInlet EPC
delivers 5.4 mL ⁄min to both columns. The AUX EPC delivers 1.6 mL ⁄min to the capillary flow technology (CFT) union. This
increases the flow to the analytical column to 7.0 mL ⁄min. Under these conditions the oxygenates are transferred in about 1.5 min.
9.4.2 High boiling point compounds are retained in the pre-column. Oxygenates and low boiling point hydrocarbons elute into
analytical column. Oxygenates are trapped at the front of the analytical column. After TAME has eluted to the analytical column,
the pre-column flow is reversed.
9.4.3 Inlet EPC flow is reduced to 4 mL ⁄min (Fig. 4) and AUX EPC flow is increased to 11 mL ⁄min. Gas flow through the fluidic
switch backflushes the pre-column in order to send high boiling compounds out the front inlet split vent. Oven temperature
program begins and oxygenates are separated on the analytical column. A typical chromatogram using this system is shown in Fig.
7.
9.5 Verification of Detectability—Inject a 5 mg ⁄kg calibration solution and ensure that a signal/noise level of at least 5 is observed.
Adjustment of the split ratio may be needed depending on the detector. See Fig. 8. The peaks for this level (5 mg ⁄kg) have a height
of about 2 pA.
9.6 Conditioning—To protect the PLOT column, avoid injecting samples until the valve times are properly optimized using
calibration standards. It is recommended that when all of the analyses are completed, the GC oven temperature be maintained at
220 °C, with the pre-column carrier head pressure maintained at 138 kPa using the electronic pressure controller for at least several
NOTE 1—Conditions in Table 2. Calibration Standard – mg/kg internal standard in isooctane.
FIG. 7 Oxygenate Elution Pattern from WCOT/PLOT Column Set Using the Fluidic Switch System
--------------
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...