ASTM E3269-21
(Test Method)Standard Test Method for Determination of the Mass Fraction of Particle-Bound Gold in Colloidal Gold Suspensions
Standard Test Method for Determination of the Mass Fraction of Particle-Bound Gold in Colloidal Gold Suspensions
SIGNIFICANCE AND USE
5.1 Au nano-objects in various shapes (that is, rods, particles) are increasingly used for a wide variety of applications. Medical applications of AuNPs, such as targeted drug delivery, tumor detection, and treatment are becoming more common (5). AuNPs have unique optical properties related to their size and their surface can be readily functionalized. Though Au is recognized to be inert and biocompatible in its bulk form, the behavior of Au nano-objects in biological systems and the environment must be tested to ensure their inertness and safety (6). It is important to know whether prepared and stored suspensions of AuNPs contain Au in its bound state (commonly Au (0) and particle adsorbed species) or ionized state (commonly, Au (I) or Au (III)) to attribute the biological response to the appropriate species. Krug, et al., concluded that the significance of toxicity studies is considerably reduced in those cases where the material properties of the nanomaterial suspensions were not characterized prior to and during the study (7). Furthermore, the analyte mass fraction of particle bound species is used with knowledge of particle size to compute particle number concentration.
SCOPE
1.1 This test method describes the use of inductively coupled plasma optical emission spectrometry (ICP-OES; also includes ICP-AES, where AES is atomic emission spectrometry) or inductively coupled plasma mass spectrometry (ICP-MS) for the determination of the mass fraction of particle bound gold (Au) in colloidal Au suspensions. Particle bound Au is defined as the mass of Au associated with the nanoparticle (NP) fraction and strongly adsorbed to the particle surface. Unbound Au is the fraction of Au in the native suspension not associated with the Au nanoparticle fraction that is, the dissolved Au existing in solution as a complex or free ion. The mass fraction of particle bound Au is determined by subtracting the mass fraction of unbound Au measured in acidified subsamples of the particle-free supernatant from the total Au mass fraction measured in acid-digested subsamples of the colloidal Au suspension. The particle-free supernatant is obtained after centrifugation of the colloidal Au suspension. This standard prescribes the use of an appropriate internal standard and calibration using either external standardization or single-point standard additions.
1.2 Colloidal gold suspensions with AuNP diameters ranging from 1 nm to 100 nm can be determined with this method.
1.3 The standard is not limited to particles with a uniform Au composition and may be applicable to a core-shell particle with a Au shell treatment.
1.4 This standard is specific to Au. The method may be applicable to other elements measurable by ICP-OES or ICP-MS but is limited to nanoparticles that are not reactive in aqueous suspension.
1.5 No detailed instructions for operating instrumentation are provided because of differences among various makes and models. Instead, the analyst shall follow the instructions provided by the manufacturer of their particular ICP-OES, ICP-MS or centrifuge instrument, especially with regard to optimization of the instrument settings.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurements are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2021
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.02 - Physical and Chemical Characterization
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Aug-2010
- Effective Date
- 01-Apr-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Apr-2008
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 15-Feb-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Jul-2005
Overview
ASTM E3269-21: Standard Test Method for Determination of the Mass Fraction of Particle-Bound Gold in Colloidal Gold Suspensions provides laboratories and industry professionals with a reliable method for quantifying the mass fraction of gold particles bound within colloidal gold nanoparticle (AuNP) suspensions. Issued by ASTM International, this standard responds to the growing use of gold nanoparticles in sectors such as medicine, environmental science, and nanotechnology, where precise characterization of AuNP content is essential for safety, quality control, and regulatory compliance.
The standard outlines procedures using advanced analytical techniques, specifically inductively coupled plasma optical emission spectrometry (ICP-OES, also known as ICP-AES) and inductively coupled plasma mass spectrometry (ICP-MS), to distinguish and accurately measure both particle-bound and unbound (dissolved) gold within the suspension.
Key Topics
Scope and Applicability
- The test method is designed for colloidal gold suspensions with AuNP diameters ranging from 1 nm to 100 nm.
- Applicable to colloidal systems that include different nanoparticle shapes and may also extend to core-shell structures with a gold shell.
- Focuses exclusively on gold, but the methodology may apply to other elements measurable by ICP-OES or ICP-MS in non-reactive nanoparticle suspensions.
Measurement Principles
- Particle-bound gold is defined as gold associated with or strongly adsorbed on the nanoparticle fraction.
- Differentiates between particle-bound and ionized or dissolved gold species.
- Utilizes centrifugation to separate nanoparticles from solution before analysis.
- Quantitative results are based on subtracting the mass fraction of unbound gold in the particle-free supernatant from the total mass fraction measured in the acid-digested colloidal suspension.
Calibration and Quality Control
- Requires internal standards and either external calibration or single-point standard additions.
- References best practices for preparing calibration blanks, standards, and ensuring matrix matching to minimize interferences.
- Recommends adherence to manufacturer guidance for instrument operation and emphasizes safety and environmental considerations.
Applications
The determination of particle-bound gold mass fraction in colloidal AuNP suspensions has critical implications in various fields:
- Medical and Life Sciences
- Supports the safe development and regulatory approval of AuNPs used in drug delivery, tumor imaging, and therapeutic treatments by enabling robust nanomaterial characterization and toxicity studies.
- Environmental Monitoring
- Facilitates assessment of nanoparticle behavior in environmental samples, aiding studies of transport, persistence, and potential ecological impact.
- Quality Assurance in Nanomaterials Production
- Provides manufacturers with a reference procedure for batch-release testing, quality control, and specification verification of gold nanoparticle products.
- Cutting-edge Research
- Enables reliable comparison of experimental results by standardizing the measurement of nanoparticle-bound gold, essential for research reproducibility and interlaboratory studies.
Related Standards
ASTM E3269-21 complements and references several important standards in the field of nanomaterial and elemental analysis:
- ASTM D5673 - Test Method for Elements in Water by Inductively Coupled Plasma–Mass Spectrometry
- ASTM D7035 - Test Method for Determination of Metals and Metalloids in Airborne Particulate Matter by Inductively Coupled Plasma Atomic Emission Spectrometry (ICP-AES)
- ISO Guide 30:2015 - Reference Materials - Terms and Definitions
- ISO 11885:2007 - Water Quality - Determination of Selected Elements by Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES)
- ISO/TS 19590:2017 - Nanotechnologies - Size Distribution and Concentration of Inorganic Nanoparticles in Aqueous Media via Single Particle ICP-MS
By following ASTM E3269-21, organizations can ensure enhanced traceability, comparability, and quality of analytical results when measuring colloidal gold nanoparticle suspensions, providing a foundation for innovation and safety in nanotechnology-related applications.
Buy Documents
ASTM E3269-21 - Standard Test Method for Determination of the Mass Fraction of Particle-Bound Gold in Colloidal Gold Suspensions
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E3269-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of the Mass Fraction of Particle-Bound Gold in Colloidal Gold Suspensions". This standard covers: SIGNIFICANCE AND USE 5.1 Au nano-objects in various shapes (that is, rods, particles) are increasingly used for a wide variety of applications. Medical applications of AuNPs, such as targeted drug delivery, tumor detection, and treatment are becoming more common (5). AuNPs have unique optical properties related to their size and their surface can be readily functionalized. Though Au is recognized to be inert and biocompatible in its bulk form, the behavior of Au nano-objects in biological systems and the environment must be tested to ensure their inertness and safety (6). It is important to know whether prepared and stored suspensions of AuNPs contain Au in its bound state (commonly Au (0) and particle adsorbed species) or ionized state (commonly, Au (I) or Au (III)) to attribute the biological response to the appropriate species. Krug, et al., concluded that the significance of toxicity studies is considerably reduced in those cases where the material properties of the nanomaterial suspensions were not characterized prior to and during the study (7). Furthermore, the analyte mass fraction of particle bound species is used with knowledge of particle size to compute particle number concentration. SCOPE 1.1 This test method describes the use of inductively coupled plasma optical emission spectrometry (ICP-OES; also includes ICP-AES, where AES is atomic emission spectrometry) or inductively coupled plasma mass spectrometry (ICP-MS) for the determination of the mass fraction of particle bound gold (Au) in colloidal Au suspensions. Particle bound Au is defined as the mass of Au associated with the nanoparticle (NP) fraction and strongly adsorbed to the particle surface. Unbound Au is the fraction of Au in the native suspension not associated with the Au nanoparticle fraction that is, the dissolved Au existing in solution as a complex or free ion. The mass fraction of particle bound Au is determined by subtracting the mass fraction of unbound Au measured in acidified subsamples of the particle-free supernatant from the total Au mass fraction measured in acid-digested subsamples of the colloidal Au suspension. The particle-free supernatant is obtained after centrifugation of the colloidal Au suspension. This standard prescribes the use of an appropriate internal standard and calibration using either external standardization or single-point standard additions. 1.2 Colloidal gold suspensions with AuNP diameters ranging from 1 nm to 100 nm can be determined with this method. 1.3 The standard is not limited to particles with a uniform Au composition and may be applicable to a core-shell particle with a Au shell treatment. 1.4 This standard is specific to Au. The method may be applicable to other elements measurable by ICP-OES or ICP-MS but is limited to nanoparticles that are not reactive in aqueous suspension. 1.5 No detailed instructions for operating instrumentation are provided because of differences among various makes and models. Instead, the analyst shall follow the instructions provided by the manufacturer of their particular ICP-OES, ICP-MS or centrifuge instrument, especially with regard to optimization of the instrument settings. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurements are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Au nano-objects in various shapes (that is, rods, particles) are increasingly used for a wide variety of applications. Medical applications of AuNPs, such as targeted drug delivery, tumor detection, and treatment are becoming more common (5). AuNPs have unique optical properties related to their size and their surface can be readily functionalized. Though Au is recognized to be inert and biocompatible in its bulk form, the behavior of Au nano-objects in biological systems and the environment must be tested to ensure their inertness and safety (6). It is important to know whether prepared and stored suspensions of AuNPs contain Au in its bound state (commonly Au (0) and particle adsorbed species) or ionized state (commonly, Au (I) or Au (III)) to attribute the biological response to the appropriate species. Krug, et al., concluded that the significance of toxicity studies is considerably reduced in those cases where the material properties of the nanomaterial suspensions were not characterized prior to and during the study (7). Furthermore, the analyte mass fraction of particle bound species is used with knowledge of particle size to compute particle number concentration. SCOPE 1.1 This test method describes the use of inductively coupled plasma optical emission spectrometry (ICP-OES; also includes ICP-AES, where AES is atomic emission spectrometry) or inductively coupled plasma mass spectrometry (ICP-MS) for the determination of the mass fraction of particle bound gold (Au) in colloidal Au suspensions. Particle bound Au is defined as the mass of Au associated with the nanoparticle (NP) fraction and strongly adsorbed to the particle surface. Unbound Au is the fraction of Au in the native suspension not associated with the Au nanoparticle fraction that is, the dissolved Au existing in solution as a complex or free ion. The mass fraction of particle bound Au is determined by subtracting the mass fraction of unbound Au measured in acidified subsamples of the particle-free supernatant from the total Au mass fraction measured in acid-digested subsamples of the colloidal Au suspension. The particle-free supernatant is obtained after centrifugation of the colloidal Au suspension. This standard prescribes the use of an appropriate internal standard and calibration using either external standardization or single-point standard additions. 1.2 Colloidal gold suspensions with AuNP diameters ranging from 1 nm to 100 nm can be determined with this method. 1.3 The standard is not limited to particles with a uniform Au composition and may be applicable to a core-shell particle with a Au shell treatment. 1.4 This standard is specific to Au. The method may be applicable to other elements measurable by ICP-OES or ICP-MS but is limited to nanoparticles that are not reactive in aqueous suspension. 1.5 No detailed instructions for operating instrumentation are provided because of differences among various makes and models. Instead, the analyst shall follow the instructions provided by the manufacturer of their particular ICP-OES, ICP-MS or centrifuge instrument, especially with regard to optimization of the instrument settings. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurements are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E3269-21 is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E3269-21 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D5673-15, ASTM E691-13, ASTM E691-11, ASTM D5673-10, ASTM D7035-10, ASTM D1129-10, ASTM E691-08, ASTM D7439-08, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D1193-06, ASTM D1129-06, ASTM E691-05, ASTM D5673-05. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E3269-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E3269 − 21
Standard Test Method for
Determination of the Mass Fraction of Particle-Bound Gold
in Colloidal Gold Suspensions
This standard is issued under the fixed designation E3269; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.6 The values stated in SI units are to be regarded as
standard. No other units of measurements are included in this
1.1 This test method describes the use of inductively
standard.
coupled plasma optical emission spectrometry (ICP-OES; also
1.7 This standard does not purport to address all of the
includes ICP-AES, where AES is atomic emission spectrom-
safety concerns, if any, associated with its use. It is the
etry) or inductively coupled plasma mass spectrometry (ICP-
responsibility of the user of this standard to establish appro-
MS) for the determination of the mass fraction of particle
priate safety, health, and environmental practices and deter-
bound gold (Au) in colloidal Au suspensions. Particle bound
mine the applicability of regulatory limitations prior to use.
Au is defined as the mass of Au associated with the nanopar-
1.8 This international standard was developed in accor-
ticle (NP) fraction and strongly adsorbed to the particle
dance with internationally recognized principles on standard-
surface. Unbound Au is the fraction of Au in the native
ization established in the Decision on Principles for the
suspension not associated with the Au nanoparticle fraction
Development of International Standards, Guides and Recom-
that is, the dissolved Au existing in solution as a complex or
mendations issued by the World Trade Organization Technical
free ion.The mass fraction of particle boundAu is determined
Barriers to Trade (TBT) Committee.
by subtracting the mass fraction of unbound Au measured in
acidified subsamples of the particle-free supernatant from the
2. Referenced Documents
totalAumassfractionmeasuredinacid-digestedsubsamplesof
the colloidal Au suspension. The particle-free supernatant is 2.1 ASTM Standards:
obtained after centrifugation of the colloidal Au suspension.
D1129Terminology Relating to Water
This standard prescribes the use of an appropriate internal D1193Specification for Reagent Water
standard and calibration using either external standardization
D4210Practice for Intralaboratory Quality Control Proce-
or single-point standard additions. dures and a Discussion on Reporting Low-Level Data
(Withdrawn 2002)
1.2 Colloidal gold suspensions with AuNP diameters rang-
D5673Test Method for Elements in Water by Inductively
ing from 1 nm to 100 nm can be determined with this method.
Coupled Plasma—Mass Spectrometry
1.3 The standard is not limited to particles with a uniform
D7035Test Method for Determination of Metals and Met-
Au composition and may be applicable to a core-shell particle
alloids in Airborne Particulate Matter by Inductively
with a Au shell treatment.
Coupled Plasma Atomic Emission Spectrometry (ICP-
1.4 This standard is specific to Au. The method may be AES)
applicable to other elements measurable by ICP-OES or D7439Test Method for Determination of Elements in Air-
ICP-MS but is limited to nanoparticles that are not reactive in borne Particulate Matter by Inductively Coupled Plasma-
aqueous suspension. –Mass Spectrometry
E691Practice for Conducting an Interlaboratory Study to
1.5 No detailed instructions for operating instrumentation
Determine the Precision of a Test Method
are provided because of differences among various makes and
E1613Test Method for Determination of Lead by Induc-
models. Instead, the analyst shall follow the instructions
tively Coupled Plasma Atomic Emission Spectrometry
provided by the manufacturer of their particular ICP-OES,
(ICP-AES), Flame Atomic Absorption Spectrometry
ICP-MS or centrifuge instrument, especially with regard to
optimization of the instrument settings.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This test method is under the jurisdiction of ASTM Committee E56 on contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Nanotechnology and is the direct responsibility of Subcommittee E56.02 on Standards volume information, refer to the standard’s Document Summary page on
Physical and Chemical Characterization. the ASTM website.
Current edition approved April 1, 2021. Published July 2021. DOI: 10.1520/ The last approved version of this historical standard is referenced on
E3269-21. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3269 − 21
(FAAS), or Graphite Furnace Atomic Absorption Spec- 3.1.6 continuing calibration blank, n—asolutioncontaining
trometry (GFAAS) Techniques (Withdrawn 2021) no analyte added, that is used to verify blank response and
4 freedom from carryover. E1613
2.2 ISO Standards:
3.1.7 continuing calibration verification (CCV), n—a solu-
ISO Guide 30:2015Reference materials — Selected terms
tion (or set of solutions) of known analyte concentration used
and definitions
to verify freedom from excessive instrumental drift; the con-
ISO 11885:2007Water quality — Determination of selected
centration is to be near the mid-range of a linear calibration
elements by inductively coupled plasma optical emission
curve and may be one of the actual calibration standards.
spectrometry (ICP-OES)
adapted from E1613
ISO 15202-3:2004Workplace air — Determination of met-
als and metalloids in airborne particulate matter by induc-
3.1.7.1 Discussion—The continuing calibration verification
tively coupled plasma atomic emission spectrometry —
must be matrix matched to the acid content present in the test
Part 3: Analysis
samples. It must be analyzed before and after all samples and
ISO 22036:2008Soil quality — Determination of trace
atafrequencyofnotlessthaneverytensamples.Themeasured
elementsinextractsofsoilbyinductivelycoupledplasma
value shall fall within 610 % of the known value.
— atomic emission spectrometry (ICP-AES)
3.1.8 Gold (Au) mass fraction, n—total mass fraction ofAu
ISO/TS 19590:2017(E) Nanotechnologies — Size distribu-
determined in the native suspension following complete diges-
tion and concentration of inorganic nanoparticles in aque-
tion or the sum of the mass fraction ofAu as determined in the
ous media via single particle inductively coupled plasma
dissolved(unboundstate)andparticleboundstateofasample.
mass spectrometry
adapted from ISO 11885:2007
3.1.9 intermediate stock standard solution, n—diluted solu-
3. Terminology
tion prepared from one or more of the primary stock standard
3.1 Definitions:
solutions. D1129
3.1.1 blank test solution, n—solution prepared in the same
3.1.10 internal standard, n—pure element(s) added in
way as the test sample solution but omitting the test portion.
known amount(s) to a solution. D5673
ISO 22036:2008
3.1.10.1 Discussion—The internal standard is used to mea-
3.1.2 calibration blank, n—volume of water containing the
sure the instrument response relative to the other analytes that
same acid matrix and internal standard as the calibration
are components of the same solution. The internal standard
standards,butwithouttheadditionofanystockorintermediate
should be an element that is not a sample component.
standard solution. adapted from D5673
3.1.11 laboratoryreagentblank,n—aliquotofreagentwater
3.1.3 calibration standards, n—series of known standard
that is treated exactly as a sample including exposure to all
solutions used by the analyst for calibration of the instrument
labware, equipment, solvents, reagents, internal standards, and
(that is, preparation of the analytical curve). D5673
surrogates that are used with other samples. D1129
3.1.3.1 Discussion—Matrix matching to the test sample
3.1.11.1 Discussion—The laboratory reagent blank is used
solution is used in the preparation of calibration standards and
to determine if test method analytes or other interferences are
calibration blank.
present in the laboratory environment, the reagents, or the
3.1.4 certified reference material (CRM), n—reference ma-
apparatus.
terial (RM) characterized by a metrologically valid procedure
3.1.12 mass fraction component X, n—defined as mass of X
for one or more specified properties, accompanied by an RM
divided by mass of solution.
certificate that provides the value of the specified property, its
3.1.13 matrix interference, n—effect of a matrix component
associated uncertainty, and a statement of metrological trace-
that might cause an analytical bias. adapted from D1129
ability. ISO Guide 30:2015
3.1.14 matrix matching, n—a technique used to minimize
3.1.5 colloidal suspension, n—any material in suspension
the effect of the test sample solution matrix on the analytical
with a nominal particle size less than 100 nm. D1129
results. ISO 15202-3:2014
3.1.5.1 Discussion—Other documents define colloidal as a
3.1.14.1 Discussion—Matrix matching involves preparing
state of subdivision, implying that the molecules or polymo-
calibration standard solutions in which the concentrations of
lecular particles dispersed in a medium have at least in one
acids and other major solvents and solutes are matched with
direction a dimension roughly between 1 nm and 1 µm, or that
those in the test solutions. With unknown sample matrices,
in a system discontinuities are found at distances of that order
exact matching is not possible. In this case, the technique of
(1) or in terms of being significantly affected by Brownian
standard additions and the use of an appropriate internal
(thermal) motion when suspended in a liquid (2).
standard help to compensate for multiplicative interference.
3.1.15 memory effect, n—signalfromanelementorisotopes
ofanelementinaprevioussamplethatcontributetothesignal
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
measured in a new sample. adapted from D5673
4th Floor, New York, NY 10036, http://www.ansi.org.
3.1.16 method detection limit (MDL), n—the minimum
The boldface numbers in parentheses refer to a list of references at the end of
this standard. concentration of an analyte that can be identified, measured
E3269 − 21
and reported with a 99 % confidence that the analyte concen- solutions are measured. Additional subsamples of the AuNP
trationisgreaterthanzero.Thisconfidencelevelisdetermined suspensions are centrifuged to remove the NPs from
from analysis of a sample in a given matrix containing the suspension, and the Au mass fractions of the acid digested
analyte. D5673 particle-freesuspensionfluidsolutionsaremeasured.Themass
fraction of AuNPs (bound Au) in the AuNP suspension is
3.1.16.1 Discussion—The MDL is also known as the limit
calculatedasthedifferencebetweentheAumassfractionvalue
of detection (LOD).
obtained for the digested samples and the Au mass fraction
3.1.17 method quantitation limit (MQL), n—the minimum
valueobtainedforthecorrespondingaciddigestedparticle-free
concentration of an analyte that can be measured within
suspension fluid solutions. This test method describes proce-
predefined goals for imprecision and bias, ordinarily taken to
duresfortheuseofICP-OESorICP-MSforestimatingtheAu
be at least ten times the standard deviation of the mean blank
mass fraction values in the digested suspension and particle-
signal. adapted from D7035
freesuspensionfluidsubsamples.Twomethodsofquantitation
3.1.17.1 Discussion—The MQL is also known as the limit are prescribed: (1) external calibration with use of internal
of quantitation (LOQ). standard, and (2) single-point standard additions with use of
3.1.17.2 Discussion—Calculation of the MDL and MQL in internal standard.
accordance with Test Method D7035 is prescribed in this
4.2 Basic knowledge of and experience with ICP-OES or
standard, though alternative approaches may be used if better
ICP-MS and centrifugation are assumed.
suited for the intended purpose of the measurement. Alterna-
tive approaches are described in Practice D4210 and Refs (3
5. Significance and Use
and 4).
5.1 Au nano-objects in various shapes (that is, rods, par-
3.1.18 particle-bound Au, n—Au associated with the nano-
ticles) are increasingly used for a wide variety of applications.
particle and strongly adsorbed to the particle surface.
MedicalapplicationsofAuNPs,suchastargeteddrugdelivery,
3.1.19 primary measurement standard, n—measurement
tumor detection, and treatment are becoming more common
standard that is designated or widely acknowledged as having
(5).AuNPs have unique optical properties related to their size
the highest metrological qualities and whose property value is
and their surface can be readily functionalized. Though Au is
accepted without reference to other standards of the same
recognized to be inert and biocompatible in its bulk form, the
property or quantity, within a specified context.
behavior of Au nano-objects in biological systems and the
ISO Guide 30:2015
environmentmustbetestedtoensuretheirinertnessandsafety
3.1.20 primarystockstandardsolution,n—solutionusedfor
(6). It is important to know whether prepared and stored
preparation of the intermediate stock standard solution or
suspensionsofAuNPscontainAuinitsboundstate(commonly
calibration standards, containing the analyte of interest at a
Au (0) and particle adsorbed species) or ionized state
certified concentration traceable to a primary measurement
(commonly, Au (I) or Au (III)) to attribute the biological
standard from a recognized Certified Reference Material
responsetotheappropriatespecies.Krug,etal.,concludedthat
producer. adapted from ISO 15202-3:2004
the significance of toxicity studies is considerably reduced in
3.1.21 spectral interference, n—an interference caused by those cases where the material properties of the nanomaterial
suspensions were not characterized prior to and during the
the signal from a species other than the analyte of interest.
adapted from D7035 study (7). Furthermore, the analyte mass fraction of particle
bound species is used with knowledge of particle size to
3.1.22 standard addition(s), n—aprocedureforthedetermi-
compute particle number concentration.
nation of the concentration of a particular species in a sample
by adding known amounts of that species to the sample
6. Interferences
solution and recording the change in signal.
adapted from D1129
6.1 Au is an element known to exhibit memory effects and
the potential exists for long signal stabilization and wash-out
3.1.23 test sample solution, n—solution prepared with the
times. Dilute nitric acid (HNO ) alone (1 % volume fraction
fraction (test portion) of the test sample according to the
HNO )isnotanappropriatediluentforAu.Theuseofadilute
appropriate specifications, such that it can be used for the 3
acid or acid mixture mimicking that used for Au dissolution,
envisaged measurement. ISO 11885:2007
for example hydrochloric acid (HCl) or aqua regia in combi-
3.1.24 unbound Au, n—dissolvedAu existing in solution as
nation with thiourea, has been shown to be effective in
a complex or free ion.
reducing memory effects (8).
3.1.24.1 Discussion—Unbound Au is the fraction of Au in
6.2 Spectral Interference—Precautions should be exercised
the native suspension not associated with the Au nanoparticle
to avoid those interferences normally associated with the
fraction or strongly adsorbed to the Au nanoparticle surface.
determination of Au and prescribed internal standards, copper
3.1.25 volume fraction component X, n—defined as volume
(Cu) or platinum (Pt) using ICP-OES or Pt using ICP-MS.
of X divided by volume of solution.
Blank, sample, and internal standard test solutions shall be
used to check for the absence of spectral interference.
4. Summary of Test Method
4.1 Subsamples ofAu nanoparticle (AuNP) suspensions are 6.3 Matrix Interference—ICP-OES and ICP-MS are subject
digested with acid, and the Au mass fractions of the resulting to matrix effects and the potential exists for signal drift and
E3269 − 21
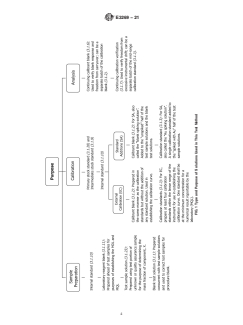
FIG. 1 Type and Purpose of Solutions Used in This Test Method
E3269 − 21
multiplicative interferences (signal enhancement or depres- 8.3 Concentrated HNO (trace metal grade).
sion). Properly chosen internal standards can reduce bias from
8.4 Concentrated HCl (trace metal grade).
these types of interferences. The method of standard additions
8.4.1 HCl diluent solution 5 % volume fraction HCl.
in combination with an internal standard is the most effective
NOTE4—Thisdiluentsolutionisusedtoimprovethechemicalstability
means to reduce systematic error caused by the matrix.
of sample and standard solutions as well as improve the stability of the
ICP-OESsignalprofileandreducethewashouttimeoftheAusignalfrom
7. Apparatus
the ICP-OES sample introduction system.
7.1 Clean, low-density polyethylene bottles (LDPE), 0.05 L
8.4.1.1 Prepare the 5 % volume fraction HCl diluent solu-
capacity with polypropylene screw cap closure or similar
tion as follows: dilute concentrated HCl with water by a
clean, metal-free polymer labware that is unreactive in con-
volumetric factor of 20.
centrated aqua regia.
8.5 Crystalline Thiourea:
7.2 Adjustable pipets capable of delivering liquid volumes
8.5.1 Thiourea diluent solution 0.5 % mass concentration
in the range 0.00015 L to 0.025 L.
thiourea in 2.4 % volume fraction HCl and 0.4 % volume
-7
7.3 Analytical balance, capable of weighing to1×10 kg
fraction HNO .
7.4 Vortex mixer.
NOTE5—Thisdiluentsolutionisusedtoimprovethechemicalstability
of sample and standard solutions as well as improve the stability of the
7.5 Bath sonicator.
ICP-MSsignalprofileandreducewashouttimeoftheAusignalfromthe
7.6 ICP-OES instrument. ICP-MS sample introduction system.
8.5.1.1 Preparethethioureadiluentsolutionasfollows:Add
NOTE1—Differencesexistamongvariousmodelsandmanufacturersof
instruments. It is the responsibility of the analyst to verify that the 5.00 g of crystalline thiourea toa1L, tared, clean fluorinated
instrument configuration and operating conditions used satisfy the ana-
ethylene propylene (FEP) bottle followed by 0.5 L of water.
lytical requirements of this method and to maintain quality control data
Cap and mix to dissolve the thiourea. Add 0.022 L concen-
confirming instrument performance and analytical results.
trated HCl. Cap and mix. Add 0.0004 L concentrated HNO .
7.7 ICP-MS instrument (see Note 1).
Dilutetoafinalvolumeof1.00Lwithwater.Capandmix.The
solution is stable for one week.
7.8 Centrifuge (see Note 1).
7.8.1 The centrifuge must produce sufficient force to com-
8.6 Au primary stock standard solution, NIST Standard
pletely removeAuNPs from suspension for the given nominal
Reference Material (SRM) 3121Au Standard Solution, or any
particlesizedistributionofthesample.Ahigh-speedcentrifuge
similar Certified Reference Material (CRM) solution with
with relative centrifugal force (RCF) of 20 000×gor greater
certifiedAu mass fraction greater than or equal to 0.001 g/g in
(× g, Earth’s gravitational acceleration = 9.81 m/s) is needed
dilute HCl.
NOTE2—Falabella,etal.,pelletized5nm,20nmand60nmAuNPsby
NOTE 6—When diluted by a factor of 100, the solution should not
-1 -1 -1 -1
centrifuging at 1570 rad s (15 000 r·min ), 523 rad s (5000 r·min ),
containalevelofCuorPtorthechoseninternalstandardthatisdetectable
-1 -1
and 262 rad s (2000 r·min ), respectively for 20 min using a Beckman
by ICP-OES.
CoulterXL-A ultracentrifuge with a titanium 4 place rotor (9), but
NOTE 7—When diluted by a factor of 30 000, the solution should not
centrifugation conditions strongly depend on the geometry of the centri-
contain a level of Pt or the chosen internal standard that is detectable by
fuge (10). Longer centrifugation times and an RCF greater than 150 000
ICP-MS.
× g are generally needed to achieve sedimentation of AuNPs with
diameters < 5 nm. Eq. 1 in Section 13.1 can be used for guidance. 8.7 Cu primary stock standard solution or chosen internal
NOTE 3—The centrifuge should have a cooling option capable of
standard for ICP-OES shall contain a known mass fraction
maintaining samples at 20 °C for the duration of the centrifugation.
greater than or equal to 0.001 g/g in dilute HCl or HNO .
8. Reagents and Materials
NOTE 8—When diluted by a volumetric factor of 20, the solution must
not contain Au at a level that is detectable by ICP-OES.
8.1 Purity of Reagents—Unless otherwise indicated, it is
8.8 Pt primary stock standard solution or chosen internal
intended that all reagents shall conform to the specifications of
standard for ICP-MS shall contain a known mass fraction
theCommitteeonAnalyticalReagentsoftheAmericanChemi-
greater than or equal to 0.001 g/g in dilute HCl or HNO .
cal Society. Other grades may be used, provided it is first
ascertained that the reagent is of sufficiently high purity to
NOTE 9—When diluted by a volumetric factor of 30 000, the solution
permit its use without lessening the accuracy of the determi-
should not contain Au at a level that is detectable by ICP-MS.
nation.
8.9 Quality assurance sample,anRMorCRMconsistingof
8.2 Purity of Water—Unless otherwise indicated, references
a suspension of AuNPs similar to the suspensions to be
to water shall conform to the resistivity requirement of 18 analyzed and having an assigned value and uncertainty for the
MΩ·cm for Type I water in Specification D1193.
mass fraction of particle-bound Au.
9. Hazards
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
9.1 Concentrated HCl and HNO are corrosive. The vapor
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
of each is an irritant. HNO acid is oxidizing. When mixed in
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
an approximate 3:1 molar ratio of concentrated HCl:HNO ,
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma- 3
copeial Convention, Inc. (USPC), Rockville, MD. aqua regia is formed. Aqua regia solutions are extremely
E3269 − 21
NOTE12—IfaroughestimateoftheAumassfractioninthetestsample
corrosive and may result in explosion (when mixed with
isunavailable,preliminaryanalysesshouldbeperformedtoobtainarough
organic compounds) and skin burns if not handled with
estimate.
extreme caution. Avoid exposure by contact with the skin or
10.2.2 Prepare a calibration blank solution in the same
eyes, or by inhalation of fumes. Use suitable personal protec-
manner as the calibration solutions but without the addition of
tiveequipment(includingimpermeablegloves,safetygoggles,
any stock or intermediate standard solution.
and laboratory coat) as established by a hazard assessment
when working with concentrated acids and aqua regia. Open-
NOTE 13—The calibration blank solution is used in establishing the
vesselsampledissolutionshouldbeperformedinafumehood.
calibration curve.
9.1.1 When preparing aqua regia, add HNO to HCl slowly.
10.2.3 Known amount of internal standard (prepared as in
9.1.1.1 An exothermic reaction will occur upon mixing the
accordancewith10.1)shouldbeaddedtothecalibrationblank
two acids.
andeachcalibrationsolutionataconcentrationlevelofsimilar
nominal concentration to the test samples and within the linear
9.2 The diluent, 5 % volume fraction HCl is corrosive.
range of the instrument. Record the mass fraction of internal
Avoid exposure by contact with skin or by inhalation. Use
standard in the calibration blank and each calibration solution.
suitablepersonalprotectiveequipment(gloves,safetygoggles,
10.2.4 At least four calibration standards shall be used to
and laboratory coat) as established by a hazard assessment for
establish the calibration curve. One standard shall be at the
HCl.
minimumconcentrationforanumericalresultreportablebythe
9.3 Thediluent,0.5%massconcentrationthioureain2.4%
laboratory. This approach permits corrective actions if results
volume fraction HCl and 0.4 % volume fraction HNO is
of the continuing calibration blank exceeds this concentration.
corrosive. Avoid exposure by contact with skin or by inhala-
10.3 Preparation of Calibration Solutions for Calibration
tion.Usesuitablepersonalprotectiveequipment(gloves,safety
by Single-Point Standard Additions:
goggles, and laboratory coat) as established by a hazard
10.3.1 Au Spiking Solution—From the stock or intermediate
assessment for HCl, HNO , and thiourea.
stock standard, prepare a fresh standard solution containing a
known mass fraction of Au. The mass fraction of Au in the
10. Preparation of Calibration Standards
spiking solution should be approximately equal to three times
10.1 Prepare intermediate stock standard solutions of Au
the average of the estimated mass fraction values ofAu in the
and the appropriate internal standard from the primary stock
suspensionstobeanalyzed.AftertheAuspikeisadded,theAu
standard solutions as required using the appropriate diluent
mass fraction of the samples should be increased by at least a
solution.
factor of two. The diluted standard solution will hereafter be
NOTE 10—For improved accuracy and precision, the user should referred to as the “Au spiking solution.”
prepare calibration and internal standard solutions on a mass fraction
NOTE14—IfaroughestimateoftheAumassfractioninthetestsample
basis.
isunavailable,preliminaryanalysesshouldbeperformedtoobtainarough
10.1.1 Cu or Pt or other appropriate element chosen on the
estimate.
basis of a performed correlation study should be used as the
10.3.2 Blank Spiking Solution—Prepare a calibration blank
internal standard for ICP-OES analysis. The HCl diluent
solution in the same manner as the Au spiking solution but
solution (see 8.4.1) should be used in the preparation of the
without the addition of any stock or intermediate standard
intermediateAu and internal standard stock standard solutions
solution.Thisblanksolutionwillhereafterbereferredtoasthe
for ICP-OES analysis.
“blank spiking solution.”
10.1.2 Ptorotherappropriateelementchosenonthebasisof
NOTE 15—The “blank spiking solution” can additionally be used as a
a performed correlation study should be used as the internal
continuing calibration blank which is used to verify blank response and
standard for ICP-MS analysis. The thiourea acid diluent
freedom from carryover.
solution (see 8.5.1) should be used in the preparation of
intermediate stock standard Au and Pt solutions for ICP-MS
11. Sampling
analysis.
11.1 Resuspension:
NOTE 11—A second set of intermediate stock standard solutions
11.1.1 If the sample was stored in a refrigerator, allow it to
prepared by serial dilution of the first set of intermediate stock standard
come to room temperature. Prior to opening the sample
solutions may be required for ICP-MS analysis to attain the appropriate
container, make sure the suspension is thoroughly mixed.
concentration range for preparation of the final set of calibration standard
Invert the container multiple times to ensure complete resus-
solutions used to establish the calibration curve.
pension of any settled
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...