ASTM F382-17
(Specification)Standard Specification and Test Method for Metallic Bone Plates
Standard Specification and Test Method for Metallic Bone Plates
ABSTRACT
This specification and test method establishes the consistent methods for classifying, and defining the geometric and performance characteristics of five types (cloverleaf, cobra head, reconstruction, straight, and tubular) of metallic bone plates used in the surgical internal fixation of the skeletal system. Also presented here are catalogs of standard specifications for material, labeling, and handling requirements, and standard test methods for measuring performance related mechanical (single cycle bend and bend fatigue) characteristics determined to be important to the in vivo performance of bone plates. This neither defines the levels of performance or case-specific clinical performance for bone plates, nor describes specific designs for bone plates.
SIGNIFICANCE AND USE
A2.5 Significance and Use
A2.5.1 This test method establishes a uniform four-point bending fatigue test to characterize and compare the fatigue performance of different bone plate designs. This test method may be used to determine a fatigue life of the bone plate at either a specific maximum bending moment or over a range of maximum bending moment conditions. Alternatively, the test method may be used to estimate a bone plate's fatigue strength for a specified number of fatigue cycles.
A2.5.2 This test method utilizes a simplified bone plate load model that may not be exactly representative of the in-situ loading configuration. The user should note that the test results generated by this test method can not be used to directly predict the in vivo performance of the bone plate being tested. The data generated from this test method can be used to conduct relative comparisons of different bone plate designs.
A2.5.3 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the devices being tested and their potential application.
A2.5.4 This test method assumes that the bone plate is manufactured from a material that exhibits linear-elastic material behavior. Therefore, the method is not applicable for testing bone plates made from materials that exhibit non-linear elastic behavior.
A2.5.5 This test method is restricted to the testing of bone plates within the lnear-elastic range of the material. . Therefore, the test method is not applicable for testing bone plates under conditions that would approach or exceed the bending strength of the bone plate being tested.
SCOPE
1.1 This specification and test method is intended to provide a comprehensive reference for bone plates used in the surgical internal fixation of the skeletal system. The standard establishes consistent methods to classify and define the geometric and performance characteristics of bone plates. The standard also presents a catalog of standard specifications that specify material; labeling and handling requirements; and standard test methods for measuring performance related mechanical characteristics determined to be important to the in vivo performance of bone plates.
1.2 It is not the intention of the standard to define levels of performance or case-specific clinical performance for bone plates, as insufficient knowledge is available to predict the consequences or their use in individual patients for specific activities of daily living. Futhermore, it is not the intention of the standard to describe or specify specific designs for bone plates used in the surgical internal fixation of the skeletal system.
1.3 This document may not be appropriate for all types of bone plates. The user is cautioned to consider the appropriateness of the standard in view of a particular bone plate and its potential application.
1.4 This document includes the following test methods used in determining the following bone plate mechanical performance characteristics:
1.4.1 Standard Test Method for Single Cycle Bend Testing of Metallic Bone Plates—Annex A1, and
1.4...
General Information
- Status
- Published
- Publication Date
- 31-Aug-2017
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.21 - Osteosynthesis
Relations
- Effective Date
- 15-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Apr-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Jun-2018
- Refers
ASTM F983-86(2018) - Standard Practice for Permanent Marking of Orthopaedic Implant Components - Effective Date
- 01-Feb-2018
- Effective Date
- 01-Jun-2014
- Refers
ASTM F983-86(2013) - Standard Practice for Permanent Marking of Orthopaedic Implant Components - Effective Date
- 01-Oct-2013
- Refers
ASTM F565-04(2013) - Standard Practice for Care and Handling of Orthopedic Implants and Instruments - Effective Date
- 01-Oct-2013
- Effective Date
- 01-Jun-2013
- Effective Date
- 15-Dec-2012
- Effective Date
- 15-Nov-2012
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 15-May-2012
Overview
ASTM F382-17 is an internationally recognized standard specification and test method for metallic bone plates used in surgical internal fixation of the skeletal system. Developed by ASTM International, this standard outlines consistent approaches for classifying and defining the geometric and performance characteristics of five primary types of metallic bone plates: cloverleaf, cobra head, reconstruction, straight, and tubular plates.
The document sets forth essential guidelines regarding the materials, labeling, and handling of these orthopedic devices, as well as standardized mechanical test methods that evaluate their structural and fatigue properties. While ASTM F382-17 supports uniformity and comparability across bone plate designs, it notably does not prescribe performance thresholds nor dictate case-specific clinical outcomes.
Key Topics
- Geometric Classification: The standard categorizes bone plates based on features such as cross-sectional shape (e.g., cloverleaf, cobra head) and anatomy-specific use cases.
- Material Specifications: It references appropriate ASTM alloy standards for surgical implant materials, supporting biocompatibility and mechanical reliability.
- Mechanical Testing:
- Single Cycle Bend Testing (Annex A1): Defines methods to measure bending stiffness, bending strength, and structural stiffness of bone plates.
- Bending Fatigue Testing (Annex A2): Standardizes fatigue life and strength assessments under repetitive loading, enabling comparative evaluation across different designs.
- Labeling and Handling: Requirements are set for proper manufacturer identification, material labeling, dimensional descriptors, and guidance on packaging to ensure traceability and device safety.
- Scope Limitations: The standard cautions users about its limits - it does not address all possible bone plate types or directly predict clinical in vivo performance, and results should be applied appropriately depending on plate design and intended application.
Applications
ASTM F382-17 finds practical value for stakeholders across the orthopedic and medical device industries:
- Design Validation: Manufacturers use the standard to confirm that new bone plate products meet consistent geometric and mechanical criteria, supporting regulatory approval submissions.
- Quality Assurance: By following standardized test methods, labs and production facilities can ensure reproducibility in measuring properties like bending stiffness and fatigue resistance of bone plates.
- Comparative Evaluation: The test protocols allow surgeons, clinicians, and engineers to comparatively assess different bone plate designs using uniform mechanical benchmarks.
- Procurement and Specification: Hospitals and procurement specialists refer to this standard when defining requirements for metallic bone plates, ensuring compatibility with existing surgical screws and expected performance.
- Education and Training: The standard serves as a reference for bioengineering curricula and surgeon education, fostering a shared understanding of device characteristics and mechanical behavior in orthopedic fixation.
Related Standards
Many ASTM and ISO standards complement ASTM F382-17, offering additional guidance and requirements for materials, testing, and orthopedic device safety:
- ASTM F67: Specification for unalloyed titanium for surgical implant applications
- ASTM F75: Specification for cobalt-chromium-molybdenum alloy castings for surgical implants
- ASTM F86: Practice for surface preparation and marking of metallic surgical implants
- ASTM F543: Specification and test methods for metallic medical bone screws
- ASTM F565: Practice for care and handling of orthopedic implants and instruments
- ASTM F136, F138, F139, F620, F621, F1472, F1713: Specifications for titanium and stainless steel implant materials
- ASTM E4, E122, E467, E1942, E1823: Standards related to mechanical testing equipment and fatigue testing
- ISO 9585: Determination of bending strength and stiffness of bone plates
- ISO 14602: Non-active surgical implants – particular requirements for osteosynthesis
Conclusion
By providing standardized geometric and performance classifications, mechanical test methods, and material guidelines, ASTM F382-17 ensures consistency, reliability, and safety in the development and evaluation of metallic bone plates for orthopedic surgery. Following this standard is essential for manufacturers, regulators, and clinicians seeking compliance with international best practices for surgical implant devices.
Buy Documents
ASTM F382-17 - Standard Specification and Test Method for Metallic Bone Plates
REDLINE ASTM F382-17 - Standard Specification and Test Method for Metallic Bone Plates
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F382-17 is a technical specification published by ASTM International. Its full title is "Standard Specification and Test Method for Metallic Bone Plates". This standard covers: ABSTRACT This specification and test method establishes the consistent methods for classifying, and defining the geometric and performance characteristics of five types (cloverleaf, cobra head, reconstruction, straight, and tubular) of metallic bone plates used in the surgical internal fixation of the skeletal system. Also presented here are catalogs of standard specifications for material, labeling, and handling requirements, and standard test methods for measuring performance related mechanical (single cycle bend and bend fatigue) characteristics determined to be important to the in vivo performance of bone plates. This neither defines the levels of performance or case-specific clinical performance for bone plates, nor describes specific designs for bone plates. SIGNIFICANCE AND USE A2.5 Significance and Use A2.5.1 This test method establishes a uniform four-point bending fatigue test to characterize and compare the fatigue performance of different bone plate designs. This test method may be used to determine a fatigue life of the bone plate at either a specific maximum bending moment or over a range of maximum bending moment conditions. Alternatively, the test method may be used to estimate a bone plate's fatigue strength for a specified number of fatigue cycles. A2.5.2 This test method utilizes a simplified bone plate load model that may not be exactly representative of the in-situ loading configuration. The user should note that the test results generated by this test method can not be used to directly predict the in vivo performance of the bone plate being tested. The data generated from this test method can be used to conduct relative comparisons of different bone plate designs. A2.5.3 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the devices being tested and their potential application. A2.5.4 This test method assumes that the bone plate is manufactured from a material that exhibits linear-elastic material behavior. Therefore, the method is not applicable for testing bone plates made from materials that exhibit non-linear elastic behavior. A2.5.5 This test method is restricted to the testing of bone plates within the lnear-elastic range of the material. . Therefore, the test method is not applicable for testing bone plates under conditions that would approach or exceed the bending strength of the bone plate being tested. SCOPE 1.1 This specification and test method is intended to provide a comprehensive reference for bone plates used in the surgical internal fixation of the skeletal system. The standard establishes consistent methods to classify and define the geometric and performance characteristics of bone plates. The standard also presents a catalog of standard specifications that specify material; labeling and handling requirements; and standard test methods for measuring performance related mechanical characteristics determined to be important to the in vivo performance of bone plates. 1.2 It is not the intention of the standard to define levels of performance or case-specific clinical performance for bone plates, as insufficient knowledge is available to predict the consequences or their use in individual patients for specific activities of daily living. Futhermore, it is not the intention of the standard to describe or specify specific designs for bone plates used in the surgical internal fixation of the skeletal system. 1.3 This document may not be appropriate for all types of bone plates. The user is cautioned to consider the appropriateness of the standard in view of a particular bone plate and its potential application. 1.4 This document includes the following test methods used in determining the following bone plate mechanical performance characteristics: 1.4.1 Standard Test Method for Single Cycle Bend Testing of Metallic Bone Plates—Annex A1, and 1.4...
ABSTRACT This specification and test method establishes the consistent methods for classifying, and defining the geometric and performance characteristics of five types (cloverleaf, cobra head, reconstruction, straight, and tubular) of metallic bone plates used in the surgical internal fixation of the skeletal system. Also presented here are catalogs of standard specifications for material, labeling, and handling requirements, and standard test methods for measuring performance related mechanical (single cycle bend and bend fatigue) characteristics determined to be important to the in vivo performance of bone plates. This neither defines the levels of performance or case-specific clinical performance for bone plates, nor describes specific designs for bone plates. SIGNIFICANCE AND USE A2.5 Significance and Use A2.5.1 This test method establishes a uniform four-point bending fatigue test to characterize and compare the fatigue performance of different bone plate designs. This test method may be used to determine a fatigue life of the bone plate at either a specific maximum bending moment or over a range of maximum bending moment conditions. Alternatively, the test method may be used to estimate a bone plate's fatigue strength for a specified number of fatigue cycles. A2.5.2 This test method utilizes a simplified bone plate load model that may not be exactly representative of the in-situ loading configuration. The user should note that the test results generated by this test method can not be used to directly predict the in vivo performance of the bone plate being tested. The data generated from this test method can be used to conduct relative comparisons of different bone plate designs. A2.5.3 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the devices being tested and their potential application. A2.5.4 This test method assumes that the bone plate is manufactured from a material that exhibits linear-elastic material behavior. Therefore, the method is not applicable for testing bone plates made from materials that exhibit non-linear elastic behavior. A2.5.5 This test method is restricted to the testing of bone plates within the lnear-elastic range of the material. . Therefore, the test method is not applicable for testing bone plates under conditions that would approach or exceed the bending strength of the bone plate being tested. SCOPE 1.1 This specification and test method is intended to provide a comprehensive reference for bone plates used in the surgical internal fixation of the skeletal system. The standard establishes consistent methods to classify and define the geometric and performance characteristics of bone plates. The standard also presents a catalog of standard specifications that specify material; labeling and handling requirements; and standard test methods for measuring performance related mechanical characteristics determined to be important to the in vivo performance of bone plates. 1.2 It is not the intention of the standard to define levels of performance or case-specific clinical performance for bone plates, as insufficient knowledge is available to predict the consequences or their use in individual patients for specific activities of daily living. Futhermore, it is not the intention of the standard to describe or specify specific designs for bone plates used in the surgical internal fixation of the skeletal system. 1.3 This document may not be appropriate for all types of bone plates. The user is cautioned to consider the appropriateness of the standard in view of a particular bone plate and its potential application. 1.4 This document includes the following test methods used in determining the following bone plate mechanical performance characteristics: 1.4.1 Standard Test Method for Single Cycle Bend Testing of Metallic Bone Plates—Annex A1, and 1.4...
ASTM F382-17 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F382-17 has the following relationships with other standards: It is inter standard links to ASTM E1823-24a, ASTM E1823-24, ASTM F2503-23e1, ASTM E1823-20, ASTM E1942-98(2018)e1, ASTM F983-86(2018), ASTM E4-14, ASTM F983-86(2013), ASTM F565-04(2013), ASTM F2503-13, ASTM E1823-12e, ASTM E1823-12d, ASTM E1823-12c, ASTM E1823-12b, ASTM E1823-12a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F382-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F382 − 17
Standard Specification and Test Method for
Metallic Bone Plates
This standard is issued under the fixed designation F382; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope using all of the described methods. Instead, the user should
only select test methods that are appropriate for a particular
1.1 This specification and test method is intended to provide
device design. In most instances, only a subset of the herein
a comprehensive reference for metallic bone plates used in the
described test methods will be required.
surgical internal fixation of the skeletal system. The standard
1.7 This standard does not purport to address all of the
establishes consistent methods to classify and define the
safety concerns, if any, associated with its use. It is the
geometric and performance characteristics of bone plates. The
responsibility of the user of this standard to establish appro-
standard also presents a catalog of standard specifications that
priate safety, health, and environmental practices and deter-
specify material; labeling and handling requirements; and
mine the applicability of regulatory limitations prior to use.
standard test methods for measuring performance related
1.8 This international standard was developed in accor-
mechanical characteristics determined to be important to the in
dance with internationally recognized principles on standard-
vivo performance of bone plates.
ization established in the Decision on Principles for the
1.2 It is not the intention of the standard to define levels of
Development of International Standards, Guides and Recom-
performance or case-specific clinical performance for bone
mendations issued by the World Trade Organization Technical
plates, as insufficient knowledge is available to predict the
Barriers to Trade (TBT) Committee.
consequences or their use in individual patients for specific
activities of daily living. Futhermore, it is not the intention of
2. Referenced Documents
the standard to describe or specify specific designs for bone
2.1 ASTM Standards:
plates used in the surgical internal fixation of the skeletal
E122 Practice for Calculating Sample Size to Estimate, With
system.
Specified Precision, the Average for a Characteristic of a
1.3 This document may not be appropriate for all types of
Lot or Process
bone plates. The user is cautioned to consider the appropriate-
F565 Practice for Care and Handling of Orthopedic Implants
ness of the standard in view of a particular bone plate and its
and Instruments
potential application.
F983 Practice for Permanent Marking of Orthopaedic Im-
1.4 This document includes the following test methods used plant Components
in determining the following bone plate mechanical perfor-
F2503 Practice for Marking Medical Devices and Other
mance characteristics: Items for Safety in the Magnetic Resonance Environment
1.4.1 Standard Test Method for Single Cycle Bend Testing
2.2 ISO Standard:
of Metallic Bone Plates—Annex A1, and
ISO 9585 Implants for Surgery—Determination of Bending
1.4.2 Standard Test Method for Determining the Bending
Strength and Stiffness of Bone Plates
Fatigue Properties Of Metallic Bone Plates—Annex A2.
ISO 14602 Non-active surgical implants—Implants for Os-
teosynthesis particular requirements.
1.5 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
3. Terminology
standard.
3.1 Definitions—Geometric:
1.6 Multiple test methods are included in this standard.
3.1.1 auto compression—a type of bone plate that by its
However, it must be noted that the user is not obligated to test
design can generate a compressive force between adjacent
1 2
This specification and test method is under the jurisdiction of ASTM Commit- For referenced ASTM standards, visit the ASTM website, www.astm.org, or
tee F04 on Medical and Surgical Materials and Devices and is the direct contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
responsibility of Subcommittee F04.21 on Osteosynthesis. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Sept. 1, 2017. Published October 2017. Originally the ASTM website.
approved in 1973. Last previous edition approved in 2014 as F382 – 14. DOI: Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
10.1520/F0382-17. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F382 − 17
FIG. 1 Bone Plate Cross-sections
unconnected bone fragments through the use of one or more 3.2 Definitions—Mechanical/Structural:
ramped holes or another type of slot geometry. This ramp or
3.2.1 bending stiffness, K (N/mm)— of a bone plate, the
slot geometry contacts the underside of the screw head, and maximum slope of the linear elastic portion of the load versus
induces compressive force as the screw is inserted and tight-
load-point displacement curve for a bone plate when tested
ened to the bone plate. according to the test method of Annex A1.
3.1.2 bone plate—a metallic device with two or more holes
3.2.2 bending strength (N-m)— of a bone plate, the bending
or slot(s), or both, and a cross section that consists of at least
moment necessary to produce a 0.2 % offset displacement in
two dimensions (width and thickness) which generally are not
the bone plate when tested as described in Annex A1.
the same in magnitude. The device is intended to provide 2
3.2.3 bending structural stiffness, El (N-m )—of a bone
alignment and fixation of two or more bone sections, primarily
plate, the bone plate’s normalized effective bending stiffness
by spanning the fracture or defect. The device is typically fixed
that takes into consideration the effects of the test setup’s
to the bone through the use of bone screws or cerclage wire. A
configuration when tested according to the method described in
partial list of general types of bone plates is given in Section
Annex A1.
4.1.
3.2.4 fatigue life, n—the number of loading cycles of a
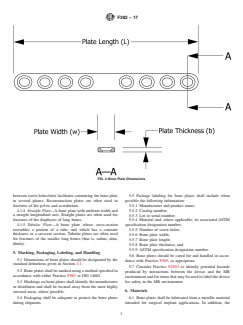
3.1.3 bone plate length, L (mm)—the linear dimension of the
specified character that a given specimen sustains before
bone plate measured along the longitudinal axis as illustrated in
failure of a specified nature occurs.
Fig. 2.
3.2.5 fatigue strength at N cycles—An estimate of the cyclic
3.1.4 bone plate thickness, b (mm)—the linear dimension of
forcing parameter (for example, load, moment, torque, stress,
the bone plate measured parallel to the screw hole axis as
and so on) at a given load ratio, for which 50 % of the
shown Figs. 1a, 1b, and 2. For a bone plate with a crescent
specimens within a given sample population would be ex-
section, the thickness is measured at the thickest point along
pected to survive N loading cycles.
the section.
3.1.5 bone plate width, w (mm)—the linear dimension of the
4. Classification
bone plate measured perpendicular to both the length and
4.1 Bone plates used in general orthopaedic surgery can be
thickness axes as shown in Fig. 2.
categorized into general types according to the following
3.1.6 contouring—the manipulation and bending of a bone
classifications:
plate, either pre-operatively or intra-operatively, to match the
4.1.1 Cloverleaf Plate—A bone plate that has one three-
anatomic geometry of the intended fixation location.
lobed end which contains screw holes.
3.1.7 crescent section—a bone plate cross-section shape
4.1.2 Cobra Head Plate—A bone plate that has one flared
(perpendicular to the long axis of the bone plate) where the
triangular or trapezoidal end which contains multiple screw
thickness is not constant along the section. Typically the
holes or slots, or both. This type of bone plate is often used for
section is thickest along the bone plate’s centerline and tapers
hip arthrodesis.
to a smaller thickness at the bone plate’s edges (see Fig. 1b).
4.1.3 Reconstruction Plate—A bone plate that does not have
3.1.8 uniform width—referring to a bone plate where the a uniform width, but usually has a smaller cross-section
width is constant along the bone plate’s length. between the screw holes or slots. The reduced cross-section
F382 − 17
FIG. 2 Bone Plate Dimensions
between screw holes/slots facilitates contouring the bone plate 5.5 Package labeling for bone plates shall include when
in several planes. Reconstruction plates are often used in possible the following information:
fractures of the pelvis and acetabulum. 5.5.1 Manufacturer and product name;
4.1.4 Straight Plate—A bone plate with uniform width and
5.5.2 Catalog number;
a straight longitudinal axis. Straight plates are often used for 5.5.3 Lot or serial number;
fractures of the diaphyses of long bones.
5.5.4 Material and, where applicable, its associated ASTM
4.1.5 Tubular Plate—A bone plate whose cross-section
specification designation number;
resembles a portion of a tube, and which has a constant
5.5.5 Number of screw holes;
thickness or a crescent section. Tubular plates are often used
5.5.6 Bone plate width;
for fractures of the smaller long bones (that is, radius, ulna,
5.5.7 Bone plate length;
fibula).
5.5.8 Bone plate thickness; and
5.5.9 ASTM specification designation number.
5. Marking, Packaging, Labeling, and Handling
5.6 Bone plates should be cared for and handled in accor-
5.1 Dimensions of bone plates should be designated by the
dance with Practice F565, as appropriate.
standard definitions given in Section 3.1.
5.7 Consider Practice F2503 to identify potential hazards
5.2 Bone plates shall be marked using a method specified in
produced by interactions between the device and the MR
accordance with either Practice F983 or ISO 14602.
environment and for terms that may be used to label the device
for safety in the MR environment.
5.3 Markings on bone plates shall identify the manufacturer
or distributor and shall be located away from the most highly
stressed areas, where possible. 6. Materials
5.4 Packaging shall be adequate to protect the bone plates 6.1 Bone plates shall be fabricated from a metallic material
during shipment. intended for surgical implant applications. In addition, the
F382 − 17
materials shall be biocompatible for the intended application. provides the primary means of stabilizing the bone fragments.
Materials should be chosen based on the design requirements Additionally, the bending stiffness of the bone plate may
of the particular device. ASTM subcommittee F04.12 main-
directly affect the rate and completeness of healing.
tains a number of specifications for materials that are suitable
7.2.1 The relevant bending properties (bending stiffness,
for surgical implant applications.
bending structural stiffness, and bending strength) shall be
determined using the standard test method of Annex A1.
7. General Requirements and Performance
Considerations 7.2.2 The relevant bending fatigue properties shall be deter-
mined in accordance with the methods described in Annex A2.
7.1 Geometric Considerations—Bone plates that are in-
tended to be used with bone screws shall have design features
8. Keywords
(screw holes or slots) that conform or appropriately fit the
corresponding bone screw.
8.1 bend testing—surgical implants; fatigue test; bone plate;
orthopedic medical devices—bone plates; surgical devices; test
7.2 Pending Properties—This is a critical characteristic of
bone plates for orthopedic applications since the bone plate methods—surgical implants
ANNEXES
(Mandatory Information)
A1. STANDARD TEST METHOD FOR SINGLE CYCLE BEND TESTING OF METALLIC BONE PLATES
A1.1 Scope: priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
A1.1.1 This test method describes methods for single cycle
bend testing in order to determine the intrinsic, structural
NOTE A1.1—There is currently an ISO standard (ISO 9585—Implants
for Surgery—Determination of Bending Strength and Stiffness of Bone
properties of metallic bone plates. The test method measures
Plates) that is similar, but not equivalent to this test method.
the bending stiffness, bending structural stiffness, and bending
strength of bone plates.
A1.2 Referenced Documents:
A1.1.2 This test method is intended to provide a means to
A1.2.1 ASTM Standards:
characterize mechanically different bone plate designs. It is not
E4 Practices for Load Verification of Testing Machines
the intention of this standard to define levels of performance
E122 Practice for Choice of Sample Size to Estimate the
for bone plates as insufficient knowledge is available to predict
Average Quality of a Lot or Process
the consequences of the use of particular bone plate designs.
A1.3 Terminology:
A1.1.3 This test method is intended to evaluate the bending
strength, bending structural stiffness, or the bending stiffness of
A1.3.1 Definitions:
the bone plate, and may not be appropriate for all situations.
A1.3.1.1 0.2 % offset displacement, q (mm)—permanent
When the structurally critical region of the bone plate is shown
deformation equal to 0.2 % of the center loading span distance.
to be located through a non-uniform region of the bone plate
(point B in Fig. A1.1).
(i.e., a peri-prosthetic, contoured plate), it may be necessary to
A1.3.1.2 bending strength (N-m)—of a bone plate, the
evaluate the bending strength, bending structural stiffness, or
bending moment necessary to produce a 0.2 % offset displace-
bending stiffness of this region of the bone plate using a
ment in the bone plate when tested as described in Section
different test method. This is because it may not be physically
A1.8 (the bending moment corresponding to point D in Fig.
possible to fit the non-uniform region between the loading
A1.1.). If the bone plate fractures before the proof point is
rollers of a four-point bend test. Structurally critical regions
attained the bending strength shall be defined as the bending
may be identified through such methods as hand calculations,
moment at fracture.
Finite Element Analysis, etc. Screw holes or other interlocking
A1.3.1.3 bending structural stiffness, (EI ) (N-m )—of a
features or contoured regions may be located at the proximal or e
bone plate, the bone plate’s normalized effective bending
distal extremities of a bone plate, and may result in structurally
stiffness that takes into consideration the effects of the test
critical regions at these locations.
setup’s configuration. For this test method, the bending struc-
A1.1.4 Units—The values stated in SI units are to be
tural stiffness is determined from the single cycle bending
regarded as standard. No other units of measurement are
response of the bone plate and the testing configuration.
included in this standard.
A1.3.1.4 bending stiffness, K (N/mm)—of a bone plate, the
A1.1.5 This standard does not purport to address all of the maximum slope of the linear elastic portion of the load versus
safety concerns, if any, associated with its use. It is the load-point curve when tested as described in section A1.8. (See
responsibility of the user of this standard to establish appro- the slope of line Om in Fig. A1.1).
F382 − 17
being tested. Such mechanical property data can be used to
conduct relative comparisons of different bone plates designs.
A1.5.3 The bending strength of the bone plate, as defined in
Section A1.3.1.2, identifies the bending moment that shall be
applied to the bone plate in order to produce a specific amount
of permanent deformation.
A1.5.4 The bending structural stiffness of the bone plate, as
defined in Section A1.3.1.3, is an indicator of the bone plate’s
stiffness that is independent of the test configuration. Bending
structural stiffness is simply related to the bone plate’s geom-
etry and the material used in manufacturing the bone plate.
A1.5.5 This test method assumes that linear-elastic material
behavior will be observed and therefore, the method is not
applicable for the testing of materials that exhibit non-linear
elastic behavior.
FIG. A1.1 Diagram Illustrating Methods for Determining the Bend-
A1.6 Apparatus:
ing Properties of Bone Plates
A1.6.1 The typical test configuration is illustrated in Fig.
A1.2.
A1.6.1.1 All loads shall be applied through rollers of equal
A1.3.1.5 bone plate width, w (mm)—the width of the bone
diameters within the range of 6 to 12 mm. The selected roller
plate as shown in Fig. A1.3.
diameter should not be greater than the distance between two
A1.3.1.6 center span, a (mm)—the distance between the two
adjacent screw holes in the bone plate to be tested.
loading rollers as shown in Fig. A1.2.
A1.6.1.2 Cylindrical rollers shall be used to test flat bone
A1.3.1.7 fracture load, F (N)—the applied load at the
max
plates and bone plates of curved cross-section, in which the
time when the bone plate fractures.
deviation from flatness at the center of the bone plate does not
A1.3.1.8 loading span, h (mm)—the distance between the
exceed w/6. Test other bone plates using rollers of profiled
loading roller and the nearest support as shown in Fig. A1.2.
form corresponding to the cross-section of the bone plate to be
A1.3.1.9 permanent deformation (mm)—the vertical dis-
tested (see Fig. A1.3).
placement of the point of load application remaining after the
A1.6.1.3 The loading and support rollers shall be positioned
applied load has been removed.
as follows:
A1.3.1.10 proof load, P (N)—the applied load at the inter-
A1.6.1.3.1 The loading rollers shall be positioned so that
section point of line. BC with the load versus load-point
two screw holes will be located between the loading rollers.
displacement curve (see Fig. A1.1).
Record the center span distance.
A1.3.1.11 proof point displacement (mm)—the load-point
A1.6.1.3.2 The support rollers shall be located equal dis-
displacement associated with the bone plate’s bending strength
tances away from the adjacent loading roller so that two screw
(see point A in Fig. A1.1).
holes will be located between the adjacent loading and support
A1.3.1.12 total deformation (mm)—the vertical displace-
rollers. Record the distance between the loading roller and
ment of the point of application of the load when specified load
nearest support roller.
is applied.
A1.6.1.3.3 The recommended testing configuration locates
A1.4 Summary of Test Method:
the two loading rollers at approximately the one-third points
between the supporting rollers.
A1.4.1 Bone plates are subjected to a single cycle four-point
A1.6.1.3.4 The applied load shall be shared equally by both
bending load. The bending stiffness, bending structural
stiffness, and bending strength of the bone plate are then loading rollers.
derived from the test record generated during the test and the A1.6.1.4 Machines used for the bending test shall conform
testing configuration. to the requirements of Practice E4.
A1.6.2 The user is strongly encouraged to obtain bone plate
A1.5 Significance and Use:
test specimens of sufficient length that can be tested using the
A1.5.1 This bend test is used to determine values for the
methods described in A1.6.1. However, alternative test con-
mechanical response of bone plates to a specific type of
figurations can be used to determine the single cycle bending
bending load. The information resulting from this test method
properties of bone plates that do not lend themselves to the
can give the surgeon some insight into the mechanical response
configuration of Sections A1.6.1 and A1.8.1. The user should
of a given bone plate.
bear in mind that the results obtained using the alternative
method described below are not directly comparable to those
A1.5.2 Since the loading on the bone plate in situ will, in
general differ from the loading configuration used in this obtained using the preferred method.
method, the results obtained from this test method cannot be A1.6.2.1 Bone plates that do not have a sufficiently long
used directly to predict in vivo performance of the bone plate section of symmetry or do not have a section of symmetry can
F382 − 17
FIG. A1.2 Test Configuration
FIG. A1.3 Roller Profiling Requirements
be attached to rigid extension segments. The rigid extension
segments can be used to effectively lengthen the bone plate so
that the bone plate can be tested with the four-point bend test
method (see Fig. A1.4 for an illustration). For these tests, the
following requirements apply.
A1.6.2.1.1 The rigid extension segments shall be designed
so that they do not interfere with the bone plate’s deformation
during the single cycle bend test.
A1.6.2.1.2 The loading rollers shall contact the rigid exten-
sion segments of the test setup during the test.
A1.6.2.1.3 At the completion of the single cycle bend test,
the bone plate anchor shall be examined in order to determine
if the indicated permanent deformation can be related to the
FIG. A1.4 Bone Plate with Rigid Extension Segments
mechanical performance of the anchoring system.
A1.6.2.2 Alternative test configurations utilized in deter-
mining the single cycle bending properties of bone plates shall
be described in the test report. A1.7 Sampling:
F382 − 17
really represents an average of the EI over the center span region.
A1.7.1 Determine sample size using the methods outlined in
e
Practice E122.
A1.8.3.1.4 Calculate the 0.2 % offset displacement from the
expression:
A1.7.2 Bone plates of different lengths but nominally iden-
tical cross sections, and made of the same material, may be
q 5 0.002 × a (A1.2)
used to constitute a sample.
where:
A1.8 Procedure:
a = the center span distance.
A1.8.1 Place the bone plate in the testing fixture and
A1.8.3.1.5 On the load versus load-point displacement dia-
position it in accordance with the following:
gram mark OB equal to q. Then draw line BC parallel to Om.
A1.8.1.1 Place the bone plate so that the loading rollers are
A1.8.3.1.6 Locate the proof load at the intersection point of
in contact with the surface of the bone plate intended to be in
line BC with the load versus load-point displacement curve.
contact with the bone.
A1.8.3.1.7 Calculate the bending strength of the bone plate
A1.8.1.2 If the bone plate is symmetrical, place it symmetri-
from the following equation:
cally with the two innermost screw holes between the loading
Ph
~ !
rollers.
bending strength 5 (A1.3)
A1.8.1.3 If the bone plate has a central screw hole, place it
with the central screw hole and one other screw hole symmetri-
where:
cally between the loading rollers.
P = the proof load, and
A1.8.1.4 If the bone plate is asymmetrical, place it with two
h = the loading span distance.
screw holes between the loading rollers so that the position of
A1.8.3.1.8 If the bone plate fractures prior to where the load
the fracture for which it is intended to be used is between the
versus load-point displacement curve intersects the offset line
loading rollers.
BC, calculate the bending strength from the expression:
A1.8.1.5 Ensure that the loading rollers are not in contact
with parts of the bone plate where there is a screw hole.
F × h
max
bending strength 5 (A1.4)
Wherever possible, the support rollers should not be in contact
with parts of the bone plate which include a screw hole.
where:
A1.8.1.6 Align the long axis of the bone plate so that it is
F = the fracture load, and
perpendicular to the axes of the rollers. max
h = the loading span distance.
A1.8.2 Apply loads of increasing magnitude, and generate a
NOTE A1.4—It should be noted that these bending strength equations
load versus load-point displacement diagram either auto-
are only valid while the bone plate under test is exhibiting linear elastic
graphically or from numeric data acquired during the test.
behavior. The user is cautioned of this fact since this method may produce
bending strength results that may not necessarily be equal to the
NOTE A1.2—Displacement-controlled testing is strongly preferred over
corresponding theoretical calculations.
load-controlled testing. The measured deformation behavior past the yield
point can be different for load-controlled testing due to non-linear
A1.9 Report:
displacement rates.
A1.9.1 Report the following information:
A1.8.3 Determine the bending stiffness, bending structural
A1.9.1.1 Adequate description of the test material, includ-
stiffness, and bending strength for each tested bone plate
ing the number of bone plates tested;
according to the method that follows:
A1.9.1.2 Adequate description of the test configuration;
A1.8.3.1 A load versus load-point displacement curve (see
A1.9.1.3 The center span and loading span dimensions (h
Fig. A1.1) is produced either autographically or from numeri-
and a);
cal data acquired during the test.
A1.8.3.1.1 On the load versus load-point displacement dia- A1.9.1.4 The 0.2 % offset displacement, q, used to deter-
gram generated for the test, draw a best fit straight line (Om) mine the bending strength;
through the initial (linear) portion of the load versus load-point
A1.9.1.5 Mean and standard deviations of the bending
displacement curve.
stiffness values for the set of bone plates tested;
A1.8.3.1.2 Determine the bone plate’s bending stiffness by
A1.9.1.6 Mean and standard deviations of the bending
calculating the slope of the line, Om, drawn in Section 8.3.1.1.
structural stiffness values for the set of bone plates tested;
A1.8.3.1.3 De
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F382 − 14 F382 − 17
Standard Specification and Test Method for
Metallic Bone Plates
This standard is issued under the fixed designation F382; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This specification and test method is intended to provide a comprehensive reference for bone plates used in the surgical
internal fixation of the skeletal system. The standard establishes consistent methods to classify and define the geometric and
performance characteristics of bone plates. The standard also presents a catalog of standard specifications that specify material;
labeling and handling requirements; and standard test methods for measuring performance related mechanical characteristics
determined to be important to the in vivo performance of bone plates.
1.2 It is not the intention of the standard to define levels of performance or case-specific clinical performance for bone plates,
as insufficient knowledge is available to predict the consequences or their use in individual patients for specific activities of daily
living. Futhermore, it is not the intention of the standard to describe or specify specific designs for bone plates used in the surgical
internal fixation of the skeletal system.
1.3 This document may not be appropriate for all types of bone plates. The user is cautioned to consider the appropriateness
of the standard in view of a particular bone plate and its potential application.
1.4 This document includes the following test methods used in determining the following bone plate mechanical performance
characteristics:
1.4.1 Standard Test Method for Single Cycle Bend Testing of Metallic Bone Plates—Annex A1, and
1.4.2 Standard Test Method for Determining the Bending Fatigue Properties Of Metallic Bone Plates—Annex A2.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 Multiple test methods are included in this standard. However, it must be noted that the user is not obligated to test using
all of the described methods. Instead, the user should only select test methods that are appropriate for a particular device design.
In most instances, only a subset of the herein described test methods will be required.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E122 Practice for Calculating Sample Size to Estimate, With Specified Precision, the Average for a Characteristic of a Lot or
Process
F67 Specification for Unalloyed Titanium, for Surgical Implant Applications (UNS R50250, UNS R50400, UNS R50550, UNS
R50700)
F75 Specification for Cobalt-28 Chromium-6 Molybdenum Alloy Castings and Casting Alloy for Surgical Implants (UNS
R30075)
F86 Practice for Surface Preparation and Marking of Metallic Surgical Implants
This specification and test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of
Subcommittee F04.21 on Osteosynthesis.
Current edition approved Nov. 1, 2014Sept. 1, 2017. Published January 2014October 2017. Originally approved in 1973. Last previous edition approved in 20082014 as
ε1
F382 – 99 (2008)F382 – 14. . DOI: 10.1520/F0382-14.10.1520/F0382-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F382 − 17
FIG. 1 Bone Plate Cross-sections
F90 Specification for Wrought Cobalt-20Chromium-15Tungsten-10Nickel Alloy for Surgical Implant Applications (UNS
R30605)
F136 Specification for Wrought Titanium-6Aluminum-4Vanadium ELI (Extra Low Interstitial) Alloy for Surgical Implant
Applications (UNS R56401)
F138 Specification for Wrought 18Chromium-14Nickel-2.5Molybdenum Stainless Steel Bar and Wire for Surgical Implants
(UNS S31673)
F139 Specification for Wrought 18Chromium-14Nickel-2.5Molybdenum Stainless Steel Sheet and Strip for Surgical Implants
(UNS S31673)
F543 Specification and Test Methods for Metallic Medical Bone Screws
F565 Practice for Care and Handling of Orthopedic Implants and Instruments
F620 Specification for Titanium Alloy Forgings for Surgical Implants in the Alpha Plus Beta Condition
F621 Specification for Stainless Steel Forgings for Surgical Implants
F983 Practice for Permanent Marking of Orthopaedic Implant Components
F1295F2503 Specification for Wrought Titanium-6Aluminum-7Niobium Alloy for Surgical Implant Applications (UNS
R56700)Practice for Marking Medical Devices and Other Items for Safety in the Magnetic Resonance Environment
F1314 Specification for Wrought Nitrogen Strengthened 22 Chromium–13 Nickel–5 Manganese–2.5 Molybdenum Stainless
Steel Alloy Bar and Wire for Surgical Implants (UNS S20910)
F1472 Specification for Wrought Titanium-6Aluminum-4Vanadium Alloy for Surgical Implant Applications (UNS R56400)
F1713 Specification for Wrought Titanium-13Niobium-13Zirconium Alloy for Surgical Implant Applications (UNS R58130)
2.2 ISO Standard:
ISO 9585 Implants for Surgery—Determination of Bending Strength and Stiffness of Bone Plates
ISO 14602 Non-active surgical implants—Implants for Osteosynthesis particular requirements.
3. Terminology
3.1 Definitions—Geometric:
3.1.1 auto compression—a type of bone plate that by its design can generate a compressive force between adjacent unconnected
bone fragments through the use of one or more ramped holes or another type of slot geometry. This ramp or slot geometry contacts
the underside of the screw head, and induces compressive force as the screw is inserted and tightened to the bone plate.
3.1.2 bone plate—a metallic device with two or more holes or slot(s), or both, and a cross section that consists of at least two
dimensions (width and thickness) which generally are not the same in magnitude. The device is intended to provide alignment and
fixation of two or more bone sections, primarily by spanning the fracture or defect. The device is typically fixed to the bone through
the use of bone screws or cerclage wire. A partial list of general types of bone plates is given in Section 4.1.
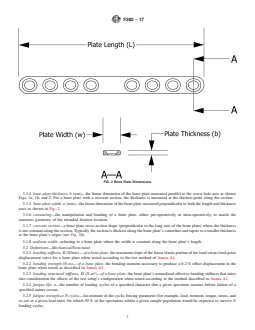
3.1.3 bone plate length, L (mm)—the linear dimension of the bone plate measured along the longitudinal axis as illustrated in
Fig. 2.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
F382 − 17
FIG. 2 Bone Plate Dimensions
3.1.4 bone plate thickness, b (mm)—the linear dimension of the bone plate measured parallel to the screw hole axis as shown
Figs. 1a, 1b, and 2. For a bone plate with a crescent section, the thickness is measured at the thickest point along the section.
3.1.5 bone plate width, w (mm)—the linear dimension of the bone plate measured perpendicular to both the length and thickness
axes as shown in Fig. 2.
3.1.6 contouring—the manipulation and bending of a bone plate, either pre-operatively or intra-operatively, to match the
anatomic geometry of the intended fixation location.
3.1.7 crescent section—a bone plate cross-section shape (perpendicular to the long axis of the bone plate) where the thickness
is not constant along the section. Typically the section is thickest along the bone plate’s centerline and tapers to a smaller thickness
at the bone plate’s edges (see Fig. 1b).
3.1.8 uniform width—referring to a bone plate where the width is constant along the bone plate’s length.
3.2 Definitions—Mechanical/Structural:
3.2.1 bending stiffness, K (N/mm)— of a bone plate, the maximum slope of the linear elastic portion of the load versus load-point
displacement curve for a bone plate when tested according to the test method of Annex A1.
3.2.2 bending strength (N-m)— of a bone plate, the bending moment necessary to produce a 0.2 % offset displacement in the
bone plate when tested as described in Annex A1.
3.2.3 bending structural stiffness, El (N-m )—of a bone plate, the bone plate’s normalized effective bending stiffness that takes
into consideration the effects of the test setup’s configuration when tested according to the method described in Annex A1.
3.2.4 fatigue life, n—the number of loading cycles of a specified character that a given specimen sustains before failure of a
specified nature occurs.
3.2.5 fatigue strength at N cycles—An estimate of the cyclic forcing parameter (for example, load, moment, torque, stress, and
so on) at a given load ratio, for which 50 % of the specimens within a given sample population would be expected to survive N
loading cycles.
F382 − 17
4. Classification
4.1 Bone plates used in general orthopaedic surgery can be categorized into general types according to the following
classifications:
4.1.1 Cloverleaf Plate—A bone plate that has one three-lobed end which contains screw holes.
4.1.2 Cobra Head Plate—A bone plate that has one flared triangular or trapezoidal end which contains multiple screw holes or
slots, or both. This type of bone plate is often used for hip arthrodesis.
4.1.3 Reconstruction Plate—A bone plate that does not have a uniform width, but usually has a smaller cross-section between
the screw holes or slots. The reduced cross-section between screw holes/slots facilitates contouring the bone plate in several planes.
Reconstruction plates are often used in fractures of the pelvis and acetabulum.
4.1.4 Straight Plate—A bone plate with uniform width and a straight longitudinal axis. Straight plates are often used for
fractures of the diaphyses of long bones.
4.1.5 Tubular Plate—A bone plate whose cross-section resembles a portion of a tube, and which has a constant thickness or a
crescent section. Tubular plates are often used for fractures of the smaller long bones (that is, radius, ulna, fibula).
5. Marking, Packaging, Labeling, and Handling
5.1 Dimensions of bone plates should be designated by the standard definitions given in Section 3.1.
5.2 Bone plates shall be marked using a method specified in accordance with either Practice F983 or ISO 14602ISO 14602.
ISO 14602.
5.3 Markings on bone plates shall identify the manufacturer or distributor and shall be located away from the most highly
stressed areas, where possible.
5.4 Packaging shall be adequate to protect the bone plates during shipment.
5.5 Package labeling for bone plates shall include when possible the following information:
5.5.1 Manufacturer and product name;
5.5.2 Catalog number;
5.5.3 Lot or serial number;
5.5.4 Material and, where applicable, its associated ASTM specification designation number;
5.5.5 Number of screw holes;
5.5.6 Bone plate width;
5.5.7 Bone plate length;
5.5.8 Bone plate thickness; and
5.5.9 ASTM specification designation number.
5.6 Bone plates should be cared for and handled in accordance with Practice F565, as appropriate.
5.7 Consider Practice F2503 to identify potential hazards produced by interactions between the device and the MR environment
and for terms that may be used to label the device for safety in the MR environment.
6. Materials
6.1 All bone plates made of materials which have an ASTM committee F04 standard designation shall meet those requirements
given in the ASTM standards. A majority of materials having ASTM specifications can be found in the list of referenced ASTM
standards of Section 2.1.
6.1 Bone plates of forged Specificationshall be fabricated F136 shall meet the requirements of Specificationfrom a metallic
material intended for surgical implant applications. In addition, the materials shall be biocompatible for the intended application.
Materials should be chosen based on the design requirements of the F620.particular device. ASTM committee F04.12 maintains
a number of metallic material specifications suitable for surgical implant applications.
6.3 Bone plates of forged Specification F138 shall meet the requirements of Specification F621.
7. General Requirements and Performance Considerations
7.1 Geometric Considerations—Bone plates that are intended to be used with bone screws shall have design features (screw
holes or slots) that conform or appropriately fit the corresponding bone screw.
7.2 Pending Properties—This is a critical characteristic of bone plates for orthopedic applications since the bone plate provides
the primary means of stabilizing the bone fragments. Additionally, the bending stiffness of the bone plate may directly affect the
rate and completeness of healing.
7.2.1 The relevant bending properties (bending stiffness, bending structural stiffness, and bending strength) shall be determined
using the standard test method of Annex A1.
7.2.2 The relevant bending fatigue properties shall be determined in accordance with the methods described in Annex A2.
F382 − 17
8. Keywords
8.1 bend testing—surgical implants; fatigue test; bone plate; orthopedic medical devices—bone plates; surgical devices; test
methods—surgical implants
ANNEXES
A1. STANDARD TEST METHOD FOR SINGLE CYCLE BEND TESTING OF METALLIC BONE PLATES
A1.1 Scope:
A1.1.1 This test method describes methods for single cycle bend testing in order to determine the intrinsic, structural properties
of metallic bone plates. The test method measures the bending stiffness, bending structural stiffness, and bending strength of bone
plates.
A1.1.2 This test method is intended to provide a means to characterize mechanically different bone plate designs. It is not the
intention of this standard to define levels of performance for bone plates as insufficient knowledge is available to predict the
consequences of the use of particular bone plate designs.
A1.1.3 This test method is intended to evaluate the bending strength, bending structural stiffness, or the bending stiffness of the
bone plate, and may not be appropriate for all situations. When the structurally critical region of the bone plate is shown to be
located through a non-uniform region of the bone plate (i.e., a peri-prosthetic, contoured plate), it may be necessary to evaluate
the bending strength, bending structural stiffness, or bending stiffness of this region of the bone plate using a different test method.
This is because it may not be physically possible to fit the non-uniform region between the loading rollers of a four-point bend
test. Structurally critical regions may be identified through such methods as hand calculations, Finite Element Analysis, etc. Screw
holes or other interlocking features or contoured regions may be located at the proximal or distal extremities of a bone plate, and
may result in structurally critical regions at these locations.
A1.1.4 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this
standard.
A1.1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.use.
NOTE A1.1—There is currently an ISO standard (ISO 9585—Implants for Surgery—Determination of Bending Strength and Stiffness of Bone Plates) that
is similar, but not equivalent to this test method.
A1.2 Referenced Documents:
A1.2.1 ASTM Standards:
E4 Practices for Load Verification of Testing Machines
E122 Practice for Choice of Sample Size to Estimate the Average Quality of a Lot or Process
A1.3 Terminology:
A1.3.1 Definitions:
A1.3.1.1 0.2 % offset displacement, q (mm)—permanent deformation equal to 0.2 % of the center loading span distance. (point B
in Fig. A1.1).
F382 − 17
FIG. A1.1 Diagram Illustrating Methods for Determining the Bending Properties of Bone Plates
A1.3.1.2 bending strength (N-m)—of a bone plate, the bending moment necessary to produce a 0.2 % offset displacement in the
bone plate when tested as described in Section A1.8 (the bending moment corresponding to point D in Fig. A1.1.). If the bone plate
fractures before the proof point is attained the bending strength shall be defined as the bending moment at fracture.
A1.3.1.3 bending structural stiffness, (EI ) (N-m )—of a bone plate, the bone plate’s normalized effective bending stiffness that
e
takes into consideration the effects of the test setup’s configuration. For this test method, the bending structural stiffness is
determined from the single cycle bending response of the bone plate and the testing configuration.
A1.3.1.4 bending stiffness, K (N/mm)—of a bone plate, the maximum slope of the linear elastic portion of the load versus
load-point curve when tested as described in section A1.8. (See the slope of line Om in Fig. A1.1).
FIG. A1.2 Test Configuration
F382 − 17
A1.3.1.5 bone plate width, w (mm)—the width of the bone plate as shown in Fig. A1.3.
A1.3.1.6 center span, a (mm)—the distance between the two loading rollers as shown in Fig. A1.2.
A1.3.1.7 fracture load, F (N)—the applied load at the time when the bone plate fractures.
max
A1.3.1.8 loading span, h (mm)—the distance between the loading roller and the nearest support as shown in Fig. A1.2.
A1.3.1.9 permanent deformation (mm)—the vertical displacement of the point of load application remaining after the applied load
has been removed.
A1.3.1.10 proof load, P (N)—the applied load at the intersection point of line. BC with the load versus load-point displacement
curve (see Fig. A1.1).
A1.3.1.11 proof point displacement (mm)—the load-point displacement associated with the bone plate’s bending strength (see
point A in Fig. A1.1).
A1.3.1.12 total deformation (mm)—the vertical displacement of the point of application of the load when specified load is applied.
A1.4 Summary of Test Method:
A1.4.1 Bone plates are subjected to a single cycle four-point bending load. The bending stiffness, bending structural stiffness, and
bending strength of the bone plate are then derived from the test record generated during the test and the testing configuration.
A1.5 Significance and Use:
A1.5.1 This bend test is used to determine values for the mechanical response of bone plates to a specific type of bending load.
The information resulting from this test method can give the surgeon some insight into the mechanical response of a given bone
plate.
A1.5.2 Since the loading on the bone plate in situ will, in general differ from the loading configuration used in this method, the
results obtained from this test method cannot be used directly to predict in vivo performance of the bone plate being tested. Such
mechanical property data can be used to conduct relative comparisons of different bone plates designs.
A1.5.3 The bending strength of the bone plate, as defined in Section A1.3.1.2, identifies the bending moment that shall be applied
to the bone plate in order to produce a specific amount of permanent deformation.
A1.5.4 The bending structural stiffness of the bone plate, as defined in Section A1.3.1.3, is an indicator of the bone plate’s stiffness
that is independent of the test configuration. Bending structural stiffness is simply related to the bone plate’s geometry and the
material used in manufacturing the bone plate.
FIG. A1.3 Roller Profiling Requirements
F382 − 17
A1.5.5 This test method assumes that linear-elastic material behavior will be observed and therefore, the method is not applicable
for the testing of materials that exhibit non-linear elastic behavior.
A1.6 Apparatus:
A1.6.1 The typical test configuration is illustrated in Fig. A1.2.
A1.6.1.1 All loads shall be applied through rollers of equal diameters within the range of 6 to 12 mm. The selected roller diameter
should not be greater than the distance between two adjacent screw holes in the bone plate to be tested.
A1.6.1.2 Cylindrical rollers shall be used to test flat bone plates and bone plates of curved cross-section, in which the deviation
from flatness at the center of the bone plate does not exceed w/6. Test other bone plates using rollers of profiled form corresponding
to the cross-section of the bone plate to be tested (see Fig. A1.3).
A1.6.1.3 The loading and support rollers shall be positioned as follows:
A1.6.1.3.1 The loading rollers shall be positioned so that two screw holes will be located between the loading rollers. Record the
center span distance.
A1.6.1.3.2 The support rollers shall be located equal distances away from the adjacent loading roller so that two screw holes will
be located between the adjacent loading and support rollers. Record the distance between the loading roller and nearest support
roller.
A1.6.1.3.3 The recommended testing configuration locates the two loading rollers at approximately the one-third points between
the supporting rollers.
A1.6.1.3.4 The applied load shall be shared equally by both loading rollers.
A1.6.1.4 Machines used for the bending test shall conform to the requirements of Practice E4.
A1.6.2 The user is strongly encouraged to obtain bone plate test specimens of sufficient length that can be tested using the methods
described in A1.6.1. However, alternative test configurations can be used to determine the single cycle bending properties of bone
plates that do not lend themselves to the configuration of Sections A1.6.1 and A1.8.1. The user should bear in mind that the results
obtained using the alternative method described below are not directly comparable to those obtained using the preferred method.
A1.6.2.1 Bone plates that do not have a sufficiently long section of symmetry or do not have a section of symmetry can be attached
to rigid extension segments. The rigid extension segments can be used to effectively lengthen the bone plate so that the bone plate
can be tested with the four-point bend test method (see Fig. A1.4 for an illustration). For these tests, the following requirements
apply.
FIG. A1.4 Bone Plate with Rigid Extension Segments
F382 − 17
A1.6.2.1.1 The rigid extension segments shall be designed so that they do not interfere with the bone plate’s deformation during
the single cycle bend test.
A1.6.2.1.2 The loading rollers shall contact the rigid extension segments of the test setup during the test.
A1.6.2.1.3 At the completion of the single cycle bend test, the bone plate anchor shall be examined in order to determine if the
indicated permanent deformation can be related to the mechanical performance of the anchoring system.
A1.6.2.2 Alternative test configurations utilized in determining the single cycle bending properties of bone plates shall be
described in the test report.
A1.7 Sampling:
A1.7.1 Determine sample size using the methods outlined in Practice E122.
A1.7.2 Bone plates of different lengths but nominally identical cross sections, and made of the same material, may be used to
constitute a sample.
A1.8 Procedure:
A1.8.1 Place the bone plate in the testing fixture and position it in accordance with the following:
A1.8.1.1 Place the bone plate so that the loading rollers are in contact with the surface of the bone plate intended to be in contact
with the bone.
A1.8.1.2 If the bone plate is symmetrical, place it symmetrically with the two innermost screw holes between the loading rollers.
A1.8.1.3 If the bone plate has a central screw hole, place it with the central screw hole and one other screw hole symmetrically
between the loading rollers.
A1.8.1.4 If the bone plate is asymmetrical, place it with two screw holes between the loading rollers so that the position of the
fracture for which it is intended to be used is between the loading rollers.
A1.8.1.5 Ensure that the loading rollers are not in contact with parts of the bone plate where there is a screw hole. Wherever
possible, the support rollers should not be in contact with parts of the bone plate which include a screw hole.
A1.8.1.6 Align the long axis of the bone plate so that it is perpendicular to the axes of the rollers.
A1.8.2 Apply loads of increasing magnitude, and generate a load versus load-point displacement diagram either auto-graphically
or from numeric data acquired during the test.
NOTE A1.2—Displacement-controlled testing is strongly preferred over load-controlled testing. The measured deformation behavior past the yield point
can be different for load-controlled testing due to non-linear displacement rates.
A1.8.3 Determine the bending stiffness, bending structural stiffness, and bending strength for each tested bone plate according to
the method that follows:
A1.8.3.1 A load versus load-point displacement curve (see Fig. A1.1) is produced either autographically or from numerical data
acquired during the test.
A1.8.3.1.1 On the load versus load-point displacement diagram generated for the test, draw a best fit straight line (Om) through
the initial (linear) portion of the load versus load-point displacement curve.
A1.8.3.1.2 Determine the bone plate’s bending stiffness by calculating the slope of the line, Om, drawn in Section 8.3.1.1.
F382 − 17
A1.8.3.1.3 Determine the bone plate’s bending structural stiffness with the following expression:
2h13a Kh
~ !
EI 5 (A1.1)
e
where:
K = the bending stiffness,
a = the center span distance, and
h = the loading span distance.
NOTE A1.3—Since the test method requires the inclusion of screw holes in the center span region, the bending structural stiffness of the bone plate really
represents an average of the EI over the center span region.
e
A1.8.3.1.4 Calculate the 0.2 % offset displacement from the expression:
q5 0.002 3a (A1.2)
where:
a = the center span distance.
A1.8.3.1.5 On the load versus load-point displacement diagram mark OB equal to q. Then draw line BC parallel to Om.
A1.8.3.1.6 Locate
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...