ASTM F3446-20
(Test Method)Standard Test Method for Determination of Frictional Torque and Friction Factor for Hip Implants Using an Anatomical Motion Hip Simulator
Standard Test Method for Determination of Frictional Torque and Friction Factor for Hip Implants Using an Anatomical Motion Hip Simulator
SIGNIFICANCE AND USE
5.1 This test procedure provides a method of evaluating the frictional torque and friction factor of artificial hip joint bearings under the stated in-vitro test conditions.
5.2 Friction is not simply a materials property. The specimen system and the effects on its friction are multi-factorial, including the materials and processing of the components, the design and assembly of the components, the test parameters, and environmental factors (lubricant, temperature, etc.).
5.3 The procedure may be used as a standardized method of measuring friction for a particular system, or as a method of investigating the effects of specific test parameters such as hip sizes, designs, radial clearance, different lubricants, clamping (nonuniform sphericity), misalignments during installation, etc.
5.4 The procedure may be used to study the variation of friction with time as the specimens wear, which is particularly useful for samples that undergo a transition from “run-in” to “steady-state” wear behavior. Since the motion and load waveforms are identical to those specified in ISO 14242-1:2014, standardized friction and wear measurements may be combined and viewed in the correct perspective where they affect each other.
5.5 Frictional torque, and in particular the maximum value, are useful to assess the torques that may compromise fixation, or cause disassociation of modular components in acetabular cup or liner/shell assemblies through a lever-out or torsion-out mechanism.
5.6 Friction factor is a useful parameter for comparison of materials and designs, and provides insights into the lubrication regime operating in the implant system. Friction factor measurement may also be able to detect acetabular liner deformation (clamping referred to earlier).
SCOPE
1.1 This test procedure provides a method of evaluating the frictional torque and friction factor of artificial hip joint bearings used in Total Hip Replacement systems. The method presented here was based on a published study, first as a conference paper in 2008 (1)2 and then as a peer-reviewed journal paper (2). The method is compatible with and is capable of being carried out during actual wear testing of total hip replacement implants on wear simulators equipped with multiple degrees of freedom force and moment sensors.
1.2 Although the methodology described does not replicate all physiological loading conditions, it is a means of in-vitro comparison of the frictional torque and friction factor of artificial hip joint bearings used in Total Hip Replacement systems under the stated test conditions.
1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Nov-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Nov-2015
- Effective Date
- 01-Jun-2013
- Refers
ASTM F86-12a - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 01-Dec-2012
- Effective Date
- 15-Aug-2012
- Refers
ASTM F86-12 - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 15-May-2012
- Effective Date
- 01-May-2012
- Effective Date
- 01-Jun-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Jul-2010
- Effective Date
- 01-Jan-2010
- Effective Date
- 01-Dec-2009
- Effective Date
- 15-Nov-2009
- Effective Date
- 01-Mar-2006
Overview
ASTM F3446-20 is an international standard developed by ASTM for the determination of frictional torque and friction factor in hip implants using an anatomical motion hip simulator. This standard outlines test methods designed to evaluate artificial hip joint bearings under in-vitro conditions, primarily in the context of total hip replacement (THR) systems. The approach enables researchers and manufacturers to systematically assess the friction characteristics of hip implants, considering variables such as component materials, design, assembly, and environmental factors like lubrication and temperature.
Friction has significant implications for the performance and longevity of hip prostheses. Assessing both frictional torque and friction factor is essential to ensure implant safety, optimize materials and designs, and predict potential modes of failure, such as component loosening or the disassociation of modular joints.
Key Topics
Frictional Torque and Friction Factor Measurement
- Quantifies the resistance experienced by hip implant surfaces under simulated physiological loads.
- Provides a standardized procedure for comparing different designs, materials, and assembly methods.
Test Setup and Procedures
- Utilizes a hip simulator capable of multiple degrees of freedom and equipped with force and moment sensors.
- In-vitro testing is carried out under defined lubricant and temperature conditions, reflecting aspects of human joint operation.
Assessment of Multi-Factorial Influences
- Evaluates the effects of variables including component material, surface processing, design geometry, radial clearance, and type of lubricants.
- Allows investigation into the impact of clamping, assembly misalignments, and environmental parameters.
Wear and Friction Evolution
- Enables the study of friction changes over time, capturing both the initial run-in phase and steady-state operation.
- Data can be correlated with wear assessments to provide a comprehensive view of implant durability and performance.
Data Reporting and Analysis
- Requires extensive documentation, including time-averaged and maximum frictional values, wear correlations, and clear traceability of test specimens.
- Specifies systematic approaches for handling and interpreting data, including exclusion criteria for low-load conditions.
Applications
Hip Implant Development and Quality Control
- Provides manufacturers and researchers with a consistent method for evaluating friction in THR systems.
- Facilitates material comparisons and optimization of implant geometries.
Implant Safety Assessment
- Supports risk analysis for implant fixation integrity, detecting frictional conditions that could compromise the stability of acetabular cups or liners.
Regulatory and Comparative Studies
- Supplies validated methodologies for third-party testing and benchmarking according to internationally recognized protocols.
- Assists in complying with regulatory requirements and supports the development of evidence for clinical application.
Tribological Research
- Enables the study of joint mechanics and wear, contributing to innovation in biomaterials and implant design.
Related Standards
- ASTM F86 - Surface Preparation and Marking of Metallic Surgical Implants
- ASTM F732 - Wear Testing of Polymeric Materials Used in Total Joint Prostheses
- ASTM F2025 - Gravimetric Measurement of Polymeric Components for Wear Assessment
- ASTM G40 - Terminology Relating to Wear and Erosion
- ISO 14242-1 - Wear of Total Hip-Joint Prostheses: Loading and Displacement Parameters
- ISO 14242-2 - Methods of Measurement for Wear Testing Machines
- ISO 4287, ISO 4288 - Surface Texture Measurement
Practical Value
By adopting ASTM F3446-20, organizations can ensure their hip implant systems are rigorously evaluated for frictional performance under standardized test conditions. The methodology offers practical insights into device longevity, safety, and comparative performance, supporting product improvement and regulatory compliance in the field of orthopedic medical devices. The standard is particularly valuable for R&D laboratories, regulatory bodies, and manufacturers engaged in the development and validation of total hip replacements.
Buy Documents
ASTM F3446-20 - Standard Test Method for Determination of Frictional Torque and Friction Factor for Hip Implants Using an Anatomical Motion Hip Simulator
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3446-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Frictional Torque and Friction Factor for Hip Implants Using an Anatomical Motion Hip Simulator". This standard covers: SIGNIFICANCE AND USE 5.1 This test procedure provides a method of evaluating the frictional torque and friction factor of artificial hip joint bearings under the stated in-vitro test conditions. 5.2 Friction is not simply a materials property. The specimen system and the effects on its friction are multi-factorial, including the materials and processing of the components, the design and assembly of the components, the test parameters, and environmental factors (lubricant, temperature, etc.). 5.3 The procedure may be used as a standardized method of measuring friction for a particular system, or as a method of investigating the effects of specific test parameters such as hip sizes, designs, radial clearance, different lubricants, clamping (nonuniform sphericity), misalignments during installation, etc. 5.4 The procedure may be used to study the variation of friction with time as the specimens wear, which is particularly useful for samples that undergo a transition from “run-in” to “steady-state” wear behavior. Since the motion and load waveforms are identical to those specified in ISO 14242-1:2014, standardized friction and wear measurements may be combined and viewed in the correct perspective where they affect each other. 5.5 Frictional torque, and in particular the maximum value, are useful to assess the torques that may compromise fixation, or cause disassociation of modular components in acetabular cup or liner/shell assemblies through a lever-out or torsion-out mechanism. 5.6 Friction factor is a useful parameter for comparison of materials and designs, and provides insights into the lubrication regime operating in the implant system. Friction factor measurement may also be able to detect acetabular liner deformation (clamping referred to earlier). SCOPE 1.1 This test procedure provides a method of evaluating the frictional torque and friction factor of artificial hip joint bearings used in Total Hip Replacement systems. The method presented here was based on a published study, first as a conference paper in 2008 (1)2 and then as a peer-reviewed journal paper (2). The method is compatible with and is capable of being carried out during actual wear testing of total hip replacement implants on wear simulators equipped with multiple degrees of freedom force and moment sensors. 1.2 Although the methodology described does not replicate all physiological loading conditions, it is a means of in-vitro comparison of the frictional torque and friction factor of artificial hip joint bearings used in Total Hip Replacement systems under the stated test conditions. 1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test procedure provides a method of evaluating the frictional torque and friction factor of artificial hip joint bearings under the stated in-vitro test conditions. 5.2 Friction is not simply a materials property. The specimen system and the effects on its friction are multi-factorial, including the materials and processing of the components, the design and assembly of the components, the test parameters, and environmental factors (lubricant, temperature, etc.). 5.3 The procedure may be used as a standardized method of measuring friction for a particular system, or as a method of investigating the effects of specific test parameters such as hip sizes, designs, radial clearance, different lubricants, clamping (nonuniform sphericity), misalignments during installation, etc. 5.4 The procedure may be used to study the variation of friction with time as the specimens wear, which is particularly useful for samples that undergo a transition from “run-in” to “steady-state” wear behavior. Since the motion and load waveforms are identical to those specified in ISO 14242-1:2014, standardized friction and wear measurements may be combined and viewed in the correct perspective where they affect each other. 5.5 Frictional torque, and in particular the maximum value, are useful to assess the torques that may compromise fixation, or cause disassociation of modular components in acetabular cup or liner/shell assemblies through a lever-out or torsion-out mechanism. 5.6 Friction factor is a useful parameter for comparison of materials and designs, and provides insights into the lubrication regime operating in the implant system. Friction factor measurement may also be able to detect acetabular liner deformation (clamping referred to earlier). SCOPE 1.1 This test procedure provides a method of evaluating the frictional torque and friction factor of artificial hip joint bearings used in Total Hip Replacement systems. The method presented here was based on a published study, first as a conference paper in 2008 (1)2 and then as a peer-reviewed journal paper (2). The method is compatible with and is capable of being carried out during actual wear testing of total hip replacement implants on wear simulators equipped with multiple degrees of freedom force and moment sensors. 1.2 Although the methodology described does not replicate all physiological loading conditions, it is a means of in-vitro comparison of the frictional torque and friction factor of artificial hip joint bearings used in Total Hip Replacement systems under the stated test conditions. 1.3 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F3446-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3446-20 has the following relationships with other standards: It is inter standard links to ASTM F2025-06(2018), ASTM F732-17, ASTM G40-15, ASTM G40-13, ASTM F86-12a, ASTM F2025-06(2012), ASTM F86-12, ASTM G40-12, ASTM F732-00(2011), ASTM G40-10b, ASTM G40-10a, ASTM G40-10, ASTM F86-04(2009), ASTM G40-09, ASTM F2025-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3446-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3446 − 20
Standard Test Method for
Determination of Frictional Torque and Friction Factor for
Hip Implants Using an Anatomical Motion Hip Simulator
This standard is issued under the fixed designation F3446; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test procedure provides a method of evaluating the
F86 Practice for Surface Preparation and Marking of Metal-
frictional torque and friction factor of artificial hip joint
lic Surgical Implants
bearings used in Total Hip Replacement systems. The method
F732 Test Method for Wear Testing of Polymeric Materials
presented here was based on a published study, first as a
Used in Total Joint Prostheses
conference paper in 2008 (1) and then as a peer-reviewed
F2025 Practice for Gravimetric Measurement of Polymeric
journal paper (2). The method is compatible with and is
Components for Wear Assessment
capable of being carried out during actual wear testing of total
G40 Terminology Relating to Wear and Erosion
hip replacement implants on wear simulators equipped with
2.2 ISO Standards:
multiple degrees of freedom force and moment sensors.
ISO 14242-1 Implants for Surgery—Wear ofTotal Hip-Joint
1.2 Although the methodology described does not replicate
Prostheses, Part 1: Loading and Displacement Parameters
all physiological loading conditions, it is a means of in-vitro
for Wear-Testing Machines and Corresponding Environ-
comparison of the frictional torque and friction factor of
mental Conditions for Test
artificial hip joint bearings used in Total Hip Replacement
ISO 14242-2 Implants for Surgery—Wear ofTotal Hip-Joint
systems under the stated test conditions.
Prostheses, Part 2: Methods of Measurement
ISO 4287 Surface Texture: Profile Method—Terms, Defini-
1.3 Units—The values stated in SI units are to be regarded
tions and Surface Texture Parameters
as standard. No other units of measurement are included in this
ISO 4288 Surface Texture: Profile Method—Rules and Pro-
standard.
cedures for the Assessment of Surface Texture
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
3. Terminology
responsibility of the user of this standard to establish appro-
3.1 Definitions:
priate safety, health, and environmental practices and deter-
3.1.1 friction, n—the resisting force tangential to the com-
mine the applicability of regulatory limitations prior to use.
mon boundary between two bodies when, under the action of
1.5 This international standard was developed in accor-
an external force, one body moves or tends to move relative to
dance with internationally recognized principles on standard-
the surface of the other.
ization established in the Decision on Principles for the
3.1.2 friction coeffıcient, n—usually, friction force divided
Development of International Standards, Guides and Recom-
by the applied compressive load.
mendations issued by the World Trade Organization Technical
3.1.3 friction factor, n—in the spherical portions of articu-
Barriers to Trade (TBT) Committee.
lar surfaces, this factor is defined here for use as an effective
frictional coefficient, equal to the overall total tangential
This test method is under the jurisdiction ofASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee For referenced ASTM standards, visit the ASTM website, www.astm.org, or
F04.22 on Arthroplasty. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved Nov. 15, 2020. Published January 2021. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
F3446-20. the ASTM website.
2 4
The boldface numbers in parentheses refer to the list of references at the end of Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
this standard. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3446 − 20
frictionalforcesonthehipfemoralheadcomponentdividedby sizes, designs, radial clearance, different lubricants, clamping
the overall compressive load on that hip component. (nonuniform sphericity), misalignments during installation,
etc.
3.1.4 frictional torque, n—in the case of spherical portions
of articular surfaces, such as those of a THR, the actual 5.4 The procedure may be used to study the variation of
frictional force at any instant varies at different locations of the friction with time as the specimens wear, which is particularly
surfaces.Theoverallfrictioncanbeconvenientlycharacterized useful for samples that undergo a transition from “run-in” to
as a frictional torque, which represents the overall tangential “steady-state” wear behavior. Since the motion and load
forces on all of the femoral head component surface multiplied waveforms are identical to those specified in ISO 14242-
by a nominal radius of that spherical articulating surface. 1:2014, standardized friction and wear measurements may be
combined and viewed in the correct perspective where they
3.1.5 overall tangential force, n—total of all tangential
affect each other.
forces on the hip femoral head component surface equal to the
overall frictional torque divided by the nominal radius of the 5.5 Frictional torque, and in particular the maximum value,
articular surfaces of the THR system. are useful to assess the torques that may compromise fixation,
or cause disassociation of modular components in acetabular
3.1.6 THR system, n—total hip replacement implant system,
cup or liner/shell assemblies through a lever-out or torsion-out
typically comprising a femoral stem, femoral head, and either
mechanism.
a bearing liner and an acetabular shell, or a one-piece acetabu-
lar cup. 5.6 Friction factor is a useful parameter for comparison of
materials and designs, and provides insights into the lubrica-
3.2 For definitions relating to erosion and wear, refer to
tion regime operating in the implant system. Friction factor
Terminology G40.
measurement may also be able to detect acetabular liner
deformation (clamping referred to earlier).
4. Principle and Summary of the Test Method
6. Interferences
4.1 The femoral and acetabular components of a test speci-
men are placed in position in their normal configuration
6.1 Incorrect fixturing and alignment of the test samples,
simulating a THR system used in vivo.
such as the center of the femoral head not being concentric
with the centers of angular rotations of the test machine, will
4.2 The test apparatus, of which a schematic example is
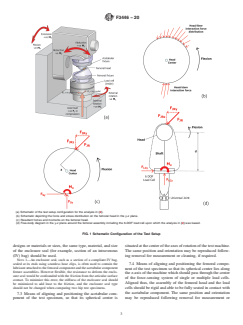
not affect the analysis or results when using the simulator and
shown in Fig. 1(a), transmits a specified time-varying com-
load cell configuration shown in Fig. 1(a) (full analysis was
pressiveforcebetweenthecomponents,togetherwithspecified
published in (2)). However, if the femoral head is not centered
relative angular displacements to simulate a physiological
with the load cell system axis to within 0.25 mm, it may result
activity such as walking as simulated in ISO 14242-1.
in exaggerated or attenuated friction values and errors in
4.3 All forces and moments (see Fig. 1) on the specimens
calculating the frictional torque reaching over 0.75 Nm. The
are measured by one (six degrees of freedom) or multiple load
effects of such misalignment were also analyzed (2) and some
cellscapableofmeasuring:(1)axialforcealongthefemoral(z)
estimates as the above were provided for its effects and simple
axis, F;(2) moment about the z-axis, M;(3 and 4) forces
z z
ways to check and correct for it were also described. One way
along orthogonal axes, F and F;(5 and 6) moments about
x y
to reduce (for example, halve) such errors is to center the
orthogonal axes, M and M . The nature of these forces and
x y
femoral head more closely to the load cell center, to within
moments and how they interact are described in (2).
0.13 mm. In this example, the 130 µm alignment accuracy is
very practical and even more alignment accuracy for two
4.4 By conducting vertical and horizontal linear force equi-
rigidly fastened mechanical components can be targeted.
librium as well as moment equilibrium on a free-body diagram
(FBD) that includes all significant net forces and moments,
7. Apparatus
calculation of the frictional torque and friction factor is
achieved, as described in detail in (2). 7.1 Testing machine, capable of producing the relative
angular displacements and corresponding forces as shown
5. Significance and Use schematically in Fig. 1(a) and based on the wear test procedure
ISO 14242-1, operating at a frequency of 1.0 6 0.1 Hz. The
5.1 This test procedure provides a method of evaluating the
flexion/extension, abduction/adduction, and internal/external
frictional torque and friction factor of artificial hip joint
rotations may be applied to either the acetabular cup or the
bearings under the stated in-vitro test conditions.
femoral head in order to create the relative rotations shown in
5.2 Friction is not simply a materials property. The speci-
Fig. 1(a).
men system and the effects on its friction are multi-factorial,
7.2 Means of mounting and enclosing the test specimen,
including the materials and processing of the components, the
using a corrosion-resistant material, capable of holding the
design and assembly of the components, the test parameters,
femoral and acetabular components using attachment methods
and environmental factors (lubricant, temperature, etc.).
comparable to the intended anatomical fixation. An enclosure
5.3 The procedure may be used as a standardized method of which is capable of isolating the test specimen to prevent
measuring friction for a particular system, or as a method of third-body contamination from the test machine and the
investigating the effects of specific test parameters such as hip atmosphere shall be provided. When comparing two hip
F3446 − 20
(a) Schematic of the test setup configuration for the analysis in (2).
(b) Schematic depicting the force and stress distribution on the femoral head in the y-z plane.
(c) Resultant forces and moments on the femoral head.
(d) Free-body diagram in the y-z plane around the femoral assembly including the 6-DOF load cell upon which the analysis in (2) was based.
FIG. 1 Schematic Configuration of the Test Setup
designs or materials or sizes, the same type, material, and size situatedatthecenteroftheaxesofrotationofthetestmachine.
of the enclosure seal (for example, section of an intravenous
The same position and orientation may be reproduced follow-
(IV) bag) should be used.
ing removal for measurement or cleaning, if required.
NOTE 1—An enclosure seal, such as a section of a compliant IV bag,
7.4 Means of aligning and positioning the femoral compo-
sealed at its ends using seamless hose clips, is often used to contain the
lubricantattachedtothefemoralcomponentandtheacetabularcomponent
nent of the test specimen so that its spherical center lies along
fixture assemblies. However flexible, the resistance to deform the enclo-
the z-axis of the machine which should pass through the center
sure seal would be confounded with the friction from the articular surface
of the force-sensing system of single or multiple load cells.
contact. To minimize this error, the stiffness of the enclosure seal should
be minimized to add least to the friction, and the enclosure seal type Aligned thus, the assembly of the femoral head and the load
should not be changed when comparing two hip test specimens.
cells should be rigid and able to be fully seated in contact with
the acetabular component. The same position and orientation
7.3 Means of aligning and positioning the acetabular com-
ponent of the test specimen, so that its spherical center is may be reproduced following removal for measurement or
F3446 − 20
cleaning, if required. The importance of this alignment and example, bone cement or a machined replica of the inner
estimates of errors resulting from misalignments in the con- surface of the backing) unless this is impractical due to
figuration shown in Fig. 1(a) were described in a published physical features of the implant system. If the component
study (2). forming the articulating surface is fixed to the backing by a
NOTE 2—Other simulator machine configurations where the load cell is
rim/snap-fit system, the machined replica shall provide the
positioned on the acetabular side will require accurate alignment of the
same fixation conditions. If it is not practical to use the normal
femoral head and need similar assessment.
backing or cement fixation due to physical features of the
NOTE 3—Any self-aligning mechanisms utilized in the above (7.3 or
implant system, the support system for the acetabular compo-
7.4) need to be verified to be frictionless unless they are outside the hip
nent should represent normal design features and conditions of
specimen/load-cellassembly,astheymayintroduceerrorsinthefrictional
forces and moments measured. For example, the universal joint used for
use but should preferably allow removal of the component for
self-alignment shown in Fig. 1(d) is below the 6-degree of freedom load
cleaning or measurement of wear without destruction.
cell, and so its friction (if any) does not introduce errors in the
measurements or the analysis (2). Different configurations from that 10.2 Femoral heads should be mounted on test spigots
shown in Fig. 1(a) and (d) should be given similar considerations with
having the same materials, dimensions, and surface finishes as
regard to such potential errors.
the stem tapers they are designed to mate with. The spigot
7.5 Motion and force control system, capable of generating
should mount the femoral head such that its center is located as
and measuring the compressive axial force and relative rota-
prescribed in 7.3 and 7.4.
tions based on ISO 14242-1 and measuring the forces and
torques described in the publication (2) with a 6-degree of
11. Test Preparation
freedom load cell.
11.1 Specimen Preparation:
NOTE 4—Multiple degrees of freedom load cells can exhibit “cross-
11.1.1 The governing rule for specimen preparation is that
talk” where a force or moment in one axis can cause an artifact of extra
thefabricationprocessparallelsthatusedorintendedforusein
measured force or moment in another axis. Optionally, one could consider
estimating such error effects by applying an axial compressive force (for the production of actual prostheses, in order to produce a
example, 3kN) at the neutral alignment of the hip system (flexion-
specimenwithcomparablebulkmaterialpropertiesandsurface
extension angle = 0°, abduction-adduction angle = 0°, and internal-
characteristics (see Practice F86). However, there may be
external angle = 0°) while measuring all the resulting forces (F,F ) and
x y
situations where it is desired to investigate the effects of a
torques (M,M,M ). These (unintended) forces and torques resulting
x y z
specific test parameter, in which case test specimens may be
from “cross-talk” and/or some implant misalignment can be processed to
estimate potential frictional errors.
produced with appropriately modified features. For example,
the effects of head roughening on friction may be studied by
7.6 Lubrication system, capable of maintaining the contact
intentionally producing head specimens with artificially rough-
surfaces immersed in the fluid test medium. The use of sealed
ened articular surfaces.
enclosures may reduce or prevent evaporation.
NOTE 5—Pre-test and post-test measurements of surface roughness can
7.7 Temperature control system, capable of maintaining the
optionally be made for all femoral heads and acetabular cups such as:
temperature of the fluid test medium at 37 6 2 °C.
spherical radius, maximum peak-to-valley deviation from sphericity, and
surface finish (R,R , and R ). Definitions of these parameters are given
8. Fluid Test Medium (Lubricant) a q sk
in ISO 4287 and measurement procedures in ISO 4288.
8.1 The fluid test medium shall be the same as what is
11.1.2 The femoral head and acetabular cup pairs may be
specified in ISO 14242-1. To minimize microbial
chosen at random, or based on other features such as radial
contamination, the fluid test medium shall be stored frozen
clearance. The rationale for the pairings should be included in
until required for testing. An antimicrobial reagent (such as
the test report.
sodium azide) can be added. A complete description of the
11.2 Sterilization:
lubricating solution, dilution, and protein concentration should
11.2.1 Sterilize the specimens in a manner typical of that in
be included in the test report. Warning—Antimicrobial re-
clinical use for such devices, unless it can be proven that this
agents are potentially hazardous.
has no effect on friction properties. Report sterilization pro-
9. Hazards
cessing parameters prior to each test, if known. Sterilization of
all test and control specimens within a specific test group
9.1 Antimicrobial reagents (such as sodium azide) are
potentially hazardous. should be done simultaneously (ideally, in a single
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...