ASTM F1801-20
(Practice)Standard Practice for Corrosion Fatigue Testing of Metallic Implant Materials
Standard Practice for Corrosion Fatigue Testing of Metallic Implant Materials
SIGNIFICANCE AND USE
4.1 Implants, particularly orthopedic devices, are usually exposed to dynamic forces. Thus, implant materials must have high fatigue resistance in the physiological environment.
4.1.1 This practice provides a procedure for fatigue testing in a simulated physiological environment. Axial tension-tension fatigue tests in an environmental test chamber are recommended as a standard procedure. The axial fatigue loading shall comply with Practices E466 and E467.
4.1.1.1 Bending and rotating bending beam fatigue tests or torsion tests may be performed in a similar environmental cell.
4.1.2 This practice is intended to assess the fatigue and corrosion fatigue properties of materials that are employed or projected to be employed for implants. This practice is suitable for studying the effects of different material treatments and surface conditions on the fatigue behavior of implant materials. The loading mode of the actual implants may be different from that of this practice. Determining the fatigue behavior of implants and implant components may require separate tests that consider the specific design and loading mode.
4.1.3 As a substitute for body fluid, 0.9 % saline solution is recommended as a standard environment. One of the various Ringer's solutions or another substitute for body fluid may also be suitable for particular tests. However, these various solutions may not give equal fatigue endurance results. The chloride ions are the most critical constituent in these solutions for initiating corrosion fatigue.
4.1.4 Because implants are manufactured from highly corrosion-resistant materials, no visible corrosion may be detectable when inspected by means of optical microscopy or scanning electron microscopy. Only a decrease of fatigue strength in the high cycle range may be noticeable. Therefore, S-N curves covering a broad fatigue loading range should be generated in the test solution and in air. Comparison of fatigue curves generated in air and saline sol...
SCOPE
1.1 This practice covers the procedure for performing corrosion fatigue tests to obtain S-N (3.2.1) fatigue curves or statistically derived fatigue strength values, or both, for metallic implant materials. This practice describes the testing of axially loaded fatigue specimens subjected to a constant amplitude, periodic forcing function in saline solution at 37°C and in air at room temperature. The environmental test method for implant materials may be adapted to other modes of fatigue loading such as bending or torsion. While this practice is not intended to apply to fatigue tests on implantable components or devices, it does provide guidelines for fatigue tests with standard specimens in an environment related to physiological conditions.
1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2020
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-Jun-2014
- Refers
ASTM F601-13 - Standard Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants - Effective Date
- 01-Dec-2013
- Refers
ASTM F86-12a - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 01-Dec-2012
- Effective Date
- 01-Jun-2012
- Effective Date
- 01-Jun-2012
- Refers
ASTM F86-12 - Standard Practice for Surface Preparation and Marking of Metallic Surgical Implants - Effective Date
- 15-May-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Nov-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Dec-2009
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Nov-2008
Overview
ASTM F1801-20 is a standard practice developed by ASTM International for conducting corrosion fatigue testing on metallic materials used in medical implants. This standard is crucial for evaluating how potential implant materials, especially those used in orthopedic applications, perform under cyclic loading in physiological environments. ASTM F1801-20 provides detailed procedures for simulating body-like conditions to assess the resistance of implant alloys to fatigue and corrosion, ensuring their reliability and longevity in service.
By simulating dynamic forces and environmental conditions that implants experience in the human body, the standard supports the development and validation of materials with high fatigue and corrosion resistance - essential for patient safety and device effectiveness.
Key Topics
Fatigue Testing in Simulated Physiological Conditions
The standard outlines how to conduct axial tension-tension fatigue tests in an environmental chamber using a solution that mimics body fluids, typically 0.9% saline at 37°C.Adaptability to Various Fatigue Modes
While axial loading is the main focus, the standard allows adaptation for bending, rotating bending, or torsion fatigue tests, supporting the evaluation of materials under various loading situations.Material and Surface Condition Assessment
The practice is designed to study the effects of different material processing methods and surface treatments on the fatigue behavior of implant materials.Specimen Preparation and Alignment
Specifies best practices for preparing test specimens, including surface finishing, dimensional requirements, and alignment to ensure reproducible and accurate test results.Test Environments and Solutions
Details the recommended use of 0.9% saline or Ringer’s solutions as physiological fluid substitutes, highlighting the significance of chloride ions in corrosion fatigue initiation.Reporting and Data Presentation
Mandates comprehensive reporting, including test conditions, specimen preparation, material properties, and presentation of results as S-N (stress vs. number of cycles) curves.
Applications
The primary application of ASTM F1801-20 is in the biomedical engineering and medical device sectors, particularly for:
Screening and Qualification of Implant Alloys
Used by manufacturers and researchers to evaluate new or existing metallic biomaterials (e.g., stainless steel, titanium alloys) for fatigue and corrosion resistance before clinical application.Material and Surface Treatment Comparison
Enables comparative analysis of the effects of alloy composition or surface modifications (such as coatings and passivation) on a material’s performance in simulated body environments.Regulatory Compliance and Device Approval
Facilitates compliance with international regulatory requirements, supporting submissions for device approval by generating robust data on material durability and safety.Inter-Laboratory Comparison and Research Standardization
Provides a unified methodology for laboratories to produce comparable results, supporting both industrial and academic research in implant materials.
Related Standards
For comprehensive testing and analysis, ASTM F1801-20 references and complements several other ASTM standards, including:
- ASTM E466 - Practice for Conducting Force Controlled Constant Amplitude Axial Fatigue Tests of Metallic Materials
- ASTM E467 - Practice for Verification of Constant Amplitude Dynamic Forces in an Axial Fatigue Testing System
- ASTM E468 - Practice for Presentation of Constant Amplitude Fatigue Test Results for Metallic Materials
- ASTM E4 - Practices for Force Verification of Testing Machines
- ASTM F86 - Practice for Surface Preparation and Marking of Metallic Surgical Implants
- ANSI B46.1 - Surface Texture Standard
By following ASTM F1801-20 in conjunction with these references, organizations ensure industry best practices in corrosion fatigue testing, enhancing the safety, performance, and reliability of metallic implant materials.
Keywords: ASTM F1801-20, corrosion fatigue, metallic implant materials, fatigue testing, physiological environment, S-N curves, orthopedic implants, biomedical materials, saline solution, standard practice.
Buy Documents
ASTM F1801-20 - Standard Practice for Corrosion Fatigue Testing of Metallic Implant Materials
REDLINE ASTM F1801-20 - Standard Practice for Corrosion Fatigue Testing of Metallic Implant Materials
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1801-20 is a standard published by ASTM International. Its full title is "Standard Practice for Corrosion Fatigue Testing of Metallic Implant Materials". This standard covers: SIGNIFICANCE AND USE 4.1 Implants, particularly orthopedic devices, are usually exposed to dynamic forces. Thus, implant materials must have high fatigue resistance in the physiological environment. 4.1.1 This practice provides a procedure for fatigue testing in a simulated physiological environment. Axial tension-tension fatigue tests in an environmental test chamber are recommended as a standard procedure. The axial fatigue loading shall comply with Practices E466 and E467. 4.1.1.1 Bending and rotating bending beam fatigue tests or torsion tests may be performed in a similar environmental cell. 4.1.2 This practice is intended to assess the fatigue and corrosion fatigue properties of materials that are employed or projected to be employed for implants. This practice is suitable for studying the effects of different material treatments and surface conditions on the fatigue behavior of implant materials. The loading mode of the actual implants may be different from that of this practice. Determining the fatigue behavior of implants and implant components may require separate tests that consider the specific design and loading mode. 4.1.3 As a substitute for body fluid, 0.9 % saline solution is recommended as a standard environment. One of the various Ringer's solutions or another substitute for body fluid may also be suitable for particular tests. However, these various solutions may not give equal fatigue endurance results. The chloride ions are the most critical constituent in these solutions for initiating corrosion fatigue. 4.1.4 Because implants are manufactured from highly corrosion-resistant materials, no visible corrosion may be detectable when inspected by means of optical microscopy or scanning electron microscopy. Only a decrease of fatigue strength in the high cycle range may be noticeable. Therefore, S-N curves covering a broad fatigue loading range should be generated in the test solution and in air. Comparison of fatigue curves generated in air and saline sol... SCOPE 1.1 This practice covers the procedure for performing corrosion fatigue tests to obtain S-N (3.2.1) fatigue curves or statistically derived fatigue strength values, or both, for metallic implant materials. This practice describes the testing of axially loaded fatigue specimens subjected to a constant amplitude, periodic forcing function in saline solution at 37°C and in air at room temperature. The environmental test method for implant materials may be adapted to other modes of fatigue loading such as bending or torsion. While this practice is not intended to apply to fatigue tests on implantable components or devices, it does provide guidelines for fatigue tests with standard specimens in an environment related to physiological conditions. 1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Implants, particularly orthopedic devices, are usually exposed to dynamic forces. Thus, implant materials must have high fatigue resistance in the physiological environment. 4.1.1 This practice provides a procedure for fatigue testing in a simulated physiological environment. Axial tension-tension fatigue tests in an environmental test chamber are recommended as a standard procedure. The axial fatigue loading shall comply with Practices E466 and E467. 4.1.1.1 Bending and rotating bending beam fatigue tests or torsion tests may be performed in a similar environmental cell. 4.1.2 This practice is intended to assess the fatigue and corrosion fatigue properties of materials that are employed or projected to be employed for implants. This practice is suitable for studying the effects of different material treatments and surface conditions on the fatigue behavior of implant materials. The loading mode of the actual implants may be different from that of this practice. Determining the fatigue behavior of implants and implant components may require separate tests that consider the specific design and loading mode. 4.1.3 As a substitute for body fluid, 0.9 % saline solution is recommended as a standard environment. One of the various Ringer's solutions or another substitute for body fluid may also be suitable for particular tests. However, these various solutions may not give equal fatigue endurance results. The chloride ions are the most critical constituent in these solutions for initiating corrosion fatigue. 4.1.4 Because implants are manufactured from highly corrosion-resistant materials, no visible corrosion may be detectable when inspected by means of optical microscopy or scanning electron microscopy. Only a decrease of fatigue strength in the high cycle range may be noticeable. Therefore, S-N curves covering a broad fatigue loading range should be generated in the test solution and in air. Comparison of fatigue curves generated in air and saline sol... SCOPE 1.1 This practice covers the procedure for performing corrosion fatigue tests to obtain S-N (3.2.1) fatigue curves or statistically derived fatigue strength values, or both, for metallic implant materials. This practice describes the testing of axially loaded fatigue specimens subjected to a constant amplitude, periodic forcing function in saline solution at 37°C and in air at room temperature. The environmental test method for implant materials may be adapted to other modes of fatigue loading such as bending or torsion. While this practice is not intended to apply to fatigue tests on implantable components or devices, it does provide guidelines for fatigue tests with standard specimens in an environment related to physiological conditions. 1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1801-20 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1801-20 has the following relationships with other standards: It is inter standard links to ASTM E4-14, ASTM F601-13, ASTM F86-12a, ASTM E1012-12, ASTM E1012-12e1, ASTM F86-12, ASTM E467-08e1, ASTM E468-11, ASTM E739-10, ASTM E4-10, ASTM F86-04(2009), ASTM E4-09a, ASTM E4-09, ASTM E4-08, ASTM E467-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1801-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F1801 − 20
Standard Practice for
Corrosion Fatigue Testing of Metallic Implant Materials
This standard is issued under the fixed designation F1801; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This practice covers the procedure for performing cor- 2.1 ASTM Standards:
rosion fatigue tests to obtain S-N (3.2.1) fatigue curves or E4Practices for Force Verification of Testing Machines
statistically derived fatigue strength values, or both, for metal- E466Practice for Conducting Force Controlled Constant
lic implant materials. This practice describes the testing of Amplitude Axial Fatigue Tests of Metallic Materials
E467Practice for Verification of Constant Amplitude Dy-
axially loaded fatigue specimens subjected to a constant
namic Forces in an Axial Fatigue Testing System
amplitude, periodic forcing function in saline solution at 37°C
E468Practice for Presentation of Constant Amplitude Fa-
and in air at room temperature.The environmental test method
tigue Test Results for Metallic Materials
forimplantmaterialsmaybeadaptedtoothermodesoffatigue
E739PracticeforStatisticalAnalysisofLinearorLinearized
loading such as bending or torsion. While this practice is not
Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
intendedtoapplytofatiguetestsonimplantablecomponentsor
E1012Practice for Verification of Testing Frame and Speci-
devices, it does provide guidelines for fatigue tests with
men Alignment Under Tensile and Compressive Axial
standard specimens in an environment related to physiological
Force Application
conditions.
E1150Definitions of Terms Relating to Fatigue (Withdrawn
1.2 The values stated in either SI units or inch-pound units
1996)
are to be regarded separately as standard. The values stated in
F86Practice for Surface Preparation and Marking of Metal-
each system may not be exact equivalents; therefore, each
lic Surgical Implants
system shall be used independently of the other. Combining
F601Practice for Fluorescent Penetrant Inspection of Me-
values from the two systems may result in non-conformance
tallic Surgical Implants
with the standard.
G15Terminology Relating to Corrosion and CorrosionTest-
ing (Withdrawn 2010)
1.3 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
2.2 ANSI Standard:
responsibility of the user of this standard to establish appro-
ANSI B46.1Surface Texture
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
3. Terminology
1.4 This international standard was developed in accor-
3.1 Definitions:
dance with internationally recognized principles on standard-
3.1.1 Theterminologyusedinconjunctionwiththispractice
ization established in the Decision on Principles for the
complies with E1150 and Terminology G15.
Development of International Standards, Guides and Recom-
3.2 Definitions of Terms Specific to This Standard:
mendations issued by the World Trade Organization Technical
3.2.1 S-N curves—S-N curves (also known as Wöhler-
Barriers to Trade (TBT) Committee.
curves)showthecorrelationbetweentheappliedstress(S)and
the counted number (N) of cycles to failure.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
ThispracticeisunderthejurisdictionofASTMCommitteeF04onMedicaland Standards volume information, refer to the standard’s Document Summary page on
Surgical Materials and Devices and is the direct responsibility of Subcommittee the ASTM website.
F04.15 on Material Test Methods. The last approved version of this historical standard is referenced on
Current edition approved Oct. 1, 2020. Published October 2020. Originally www.astm.org.
approved in 1997. Last previous edition approved in 2014 as F1801–97(2014). Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
DOI: 10.1520/F1801-20. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1801 − 20
4. Significance and Use 5.2.1 Tension-tensionfatiguetestsmaybeperformedonone
of the following types of axial fatigue testing machines:
4.1 Implants, particularly orthopedic devices, are usually
5.2.1.1 Mechanical,
exposed to dynamic forces. Thus, implant materials must have
5.2.1.2 Electromechanical or magnetically driven, and
high fatigue resistance in the physiological environment.
5.2.1.3 Hydraulic or electrohydraulic.
4.1.1 This practice provides a procedure for fatigue testing
5.2.2 The machine shall have a load-monitoring system,
in a simulated physiological environment. Axial tension-
such as a transducer mounted in series with the specimen. The
tension fatigue tests in an environmental test chamber are
test loads shall be monitored continuously in the early stage of
recommended as a standard procedure. The axial fatigue
the test and periodically thereafter, to ensure that the desired
loading shall comply with Practices E466 and E467.
load is maintained. The magnitude of the varying loads,
4.1.1.1 Bending and rotating bending beam fatigue tests or
measured dynamically as described in Practice E467, shall be
torsion tests may be performed in a similar environmental cell.
maintained within an accuracy of less than or equal to 2% of
4.1.2 This practice is intended to assess the fatigue and
the extreme loads applied during testing.
corrosion fatigue properties of materials that are employed or
5.3 Non Axial Fatigue Testing—Corrosion fatigue tests un-
projectedtobeemployedforimplants.Thispracticeissuitable
derloadingconditionsdifferentfromaxialtension-tensionmay
for studying the effects of different material treatments and
be requested. In such cases established experimental arrange-
surfaceconditionsonthefatiguebehaviorofimplantmaterials.
ments for bending, rotating bending beam, or torsional testing
Theloadingmodeoftheactualimplantsmaybedifferentfrom
may replace the axial tension-tension mode.An environmental
that of this practice. Determining the fatigue behavior of
test chamber is attached to the equipment and the environmen-
implants and implant components may require separate tests
tal tests are carried out under conditions as described in this
that consider the specific design and loading mode.
standard. Except for the mechanical testing arrangements the
4.1.3 As a substitute for body fluid, 0.9% saline solution is
conditions of this standard practice apply where possible.
recommended as a standard environment. One of the various
Reporting should follow Section 9 and should include all
Ringer’ssolutionsoranothersubstituteforbodyfluidmayalso
details where the testing deviates from the standard procedure.
be suitable for particular tests. However, these various solu-
tions may not give equal fatigue endurance results. The
5.4 Environmental Chamber:
chlorideionsarethemostcriticalconstituentinthesesolutions
5.4.1 For corrosion fatigue testing, the machine shall be
for initiating corrosion fatigue.
fittedwithanenvironmentaltestcellsurroundingthespecimen
4.1.4 Because implants are manufactured from highly
gauge section as shown in Fig. 1. A heated solution reservoir,
corrosion-resistant materials, no visible corrosion may be
a solution pump, and connecting lines for circulating the test
detectable when inspected by means of optical microscopy or
solution to the specimen surface are required. The solution
scanning electron microscopy. Only a decrease of fatigue
should be pumped from the reservoir through the system at a
strength in the high cycle range may be noticeable. Therefore,
rate that will maintain the temperature at 37 6 1°C in the test
S-N curves covering a broad fatigue loading range should be
cell, but with flow rates low enough to avoid flow-dependent
generated in the test solution and in air. Comparison of fatigue
phenomenalikeerosion-corrosion.Thereservoirshouldhavea
curvesgeneratedinairandsalinesolutionmaybetheonlyway
minimum capacity of 1000 mL per square centimeter of
to assess the effect of the saline environment.
specimensurfaceexposedtotheelectrolyte.Thereservoirshall
4.1.5 Where the fatigue behavior of a material system is
be vented to the atmosphere. If the solution volume decreases,
already established, it may suffice to test modifications of the
the reservoir shall be replenished with distilled water to
materialpropertiesorsurfaceconditioninonlyaselectedstress
maintain the saline concentration, or the solution should be
range.
exchanged. During long testing periods exchange of the
4.1.6 The recommended loading frequency of one hertz solution is recommended.Atypical environmental test cell for
corresponds to the frequency of weight bearing during walk- axial fatigue testing is shown in Fig. 1.
ing. For screening tests, higher test frequencies may be used; 5.4.2 The test equipment should be manufactured of mate-
but it must be realized that higher frequencies may affect the
rials or should be protected in such a manner that corrosion is
results. avoided. In particular galvanic corrosion in conjunction with
4.1.7 Summary of Standard Conditions—For inter- the test specimen and loosening of the specimen grips due to
laboratory comparisons the following conditions are consid- corrosion must be avoided.
ered as the standard test. Axial tension-tension tests with
cylindrical specimens in 0.9% saline solution at 37°C and air
6. Test Solution
at room temperature under a loading frequency of 1 Hz.
6.1 To prepare the saline solution, dissolve9gof reagent-
grade sodium chloride in distilled water and make up to 1000
5. Testing Equipment
mL.Ifasubstitutefor0.9%salinesolutionisused,suchasone
5.1 The mechanics of the testing machine should be ana- of the various Ringer’s solutions, note that in the report.
lyzed to ensure that the machine is capable of maintaining the
desired form and magnitude of loading for the duration of the 7. Test Specimen
test (see Practices E4).
7.1 Specimen Design:
5.2 Axial Fatigue Testing: 7.1.1 Axial Fatigue Testing:
F1801 − 20
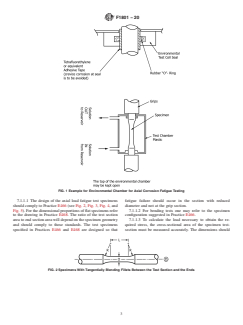
FIG. 1 Example for Environmental Chamber for Axial Corrosion Fatigue Testing
7.1.1.1 The design of the axial load fatigue test specimens fatigue failure should occur in the section with reduced
should comply to Practice E466 (see Fig. 2, Fig. 3, Fig. 4, and diameter and not at the grip section.
Fig.5).Forthedimensionalproportionsofflatspecimensrefer 7.1.1.2 For bending tests one may refer to the specimen
to the drawing in Practice E468. The ratio of the test section configuration suggested in Practice E466.
area to end section area will depend on the specimen geometry 7.1.1.3 To calculate the load necessary to obtain the re-
and should comply to those standards. The test specimens quired stress, the cross-sectional area of the specimen test-
specified in Practices E466 and E468 are designed so that section must be measured accurately. The dimensions should
FIG. 2 Specimens With Tangentially Blending Fillets Between the Test Section and the Ends
F1801 − 20
FIG. 3 Specimens With a Continuous Radius Between Ends
FIG. 4 Specimens With Tangentially Blending Fillets Between the Uniform Test Section an
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1801 − 97 (Reapproved 2014) F1801 − 20
Standard Practice for
Corrosion Fatigue Testing of Metallic Implant Materials
This standard is issued under the fixed designation F1801; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers the procedure for performing corrosion fatigue tests to obtain S-N (3.2.1) fatigue curves or statistically
derived fatigue strength values, or both, for metallic implant materials. This practice describes the testing of axially loaded fatigue
specimens subjected to a constant amplitude, periodic forcing function in saline solution at 37°C and in air at room temperature.
The environmental test method for implant materials may be adapted to other modes of fatigue loading such as bending or torsion.
While this practice is not intended to apply to fatigue tests on implantable components or devices, it does provide guidelines for
fatigue tests with standard specimens in an environment related to physiological conditions.
1.2 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each
system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the
two systems may result in non-conformance with the standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Verification of Testing Machines
E466 Practice for Conducting Force Controlled Constant Amplitude Axial Fatigue Tests of Metallic Materials
E467 Practice for Verification of Constant Amplitude Dynamic Forces in an Axial Fatigue Testing System
E468 Practice for Presentation of Constant Amplitude Fatigue Test Results for Metallic Materials
E739 Practice for Statistical Analysis of Linear or Linearized Stress-Life (S-N) and Strain-Life (ε-N) Fatigue Data
E1012 Practice for Verification of Testing Frame and Specimen Alignment Under Tensile and Compressive Axial Force
Application
E1150 Definitions of Terms Relating to Fatigue (Withdrawn 1996)
F86 Practice for Surface Preparation and Marking of Metallic Surgical Implants
F601 Practice for Fluorescent Penetrant Inspection of Metallic Surgical Implants
This practice is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.15
on Material Test Methods.
Current edition approved Oct. 1, 2014Oct. 1, 2020. Published November 2014October 2020. Originally approved in 1997. Last previous edition approved in 20092014
ε1
as F1801 – 97 (2014).(2009) . DOI: 10.1520/F1801-97R14.10.1520/F1801-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1801 − 20
G15 Terminology Relating to Corrosion and Corrosion Testing (Withdrawn 2010)
2.2 ANSI Standard:
ANSI B46.1 Surface Texture
3. Terminology
3.1 Definitions:
3.1.1 The terminology used in conjunction with this practice complies to Terminology with E1150 and Terminology G15.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 S-N curves—S-N curves (also known as Wöhler-curves) show the correlation between the applied stress (S) and the counted
number (N) of cycles to failure.
4. Significance and Use
4.1 Implants, particularly orthopedic devices, are usually exposed to dynamic forces. Thus, implant materials must have high
fatigue resistance in the physiological environment.
4.1.1 This practice provides a procedure for fatigue testing in a simulated physiological environment. Axial tension-tension fatigue
tests in an environmental test chamber are recommended as a standard procedure. The axial fatigue loading shall comply with
PracticePractices E466 and Practice E467.
4.1.1.1 Bending and rotating bending beam fatigue tests or torsion tests may be performed in a similar environmental cell.
4.1.2 This practice is intended to assess the fatigue and corrosion fatigue properties of materials that are employed or projected
to be employed for implants. This practice is suitable for studying the effects of different material treatments and surface conditions
on the fatigue behavior of implant materials. The loading mode of the actual implants may be different from that of this practice.
Determining the fatigue behavior of implants and implant components may require separate tests that consider the specific design
and loading mode.
4.1.3 As a substitute for body fluid, 0.9 % saline solution is recommended as a standard environment. One of the various Ringer’s
solutions or another substitute for body fluid may also be suitable for particular tests. However, these various solutions may not
give equal fatigue endurance results. The chloride ions are the most critical constituent in these solutions for initiating corrosion
fatigue.
4.1.4 Because implants are manufactured from highly corrosion-resistant materials, no visible corrosion may be detectable by
optical or electron-optical (SEM) means.when inspected by means of optical microscopy or scanning electron microscopy. Only
a decrease of fatigue strength in the high cyclic life cycle range may be noticeable. Therefore, S-N curves covering a broad fatigue
loading range should be generated in 0.9 % saline solution (Ringer’s solutions) and the test solution and in air. Comparison of
fatigue curves generated in air and saline solution may be the only way to assess the effect of the saline environment.
4.1.5 Where the fatigue behavior of a material system is already established, it may suffice to test modifications of the material
properties or surface condition in only a selected stress range.
4.1.6 The recommended loading frequency of one hertz corresponds to the frequency of weight-bearing weight bearing during
walking. For screening tests, higher test frequencies may be used; but it must be realized that higher frequencies may affect the
results.
4.1.7 Summary of Standard Conditions—For inter-laboratory comparisons the following conditions are considered as the standard
test. Axial tension-tension tests with cylindrical specimens in 37°C 0.9 % saline solution at 37°C and air at room temperature under
a loading frequency of 1 Hz.
5. Testing Equipment
5.1 The mechanics of the testing machine should be analyzed to ensure that the machine is capable of maintaining the desired form
and magnitude of loading for the duration of the test (see Practices E4).
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
F1801 − 20
5.2 Axial Fatigue Testing:
5.2.1 Tension-tension fatigue tests may be performed on one of the following types of axial fatigue testing machines:
5.2.1.1 Mechanical,
5.2.1.2 Electromechanical or magnetically driven, and
5.2.1.3 Hydraulic or electrohydraulic.
5.2.2 The machine shall have a load-monitoring system, such as a transducer mounted in series with the specimen. The test loads
shall be monitored continuously in the early stage of the test and periodically thereafter, to ensure that the desired load is
maintained. The magnitude of the varying loads, measured dynamically as described in Practice E467, shall be maintained within
an accuracy of less than or equal to 2 % of the extreme loads applied during testing.
5.3 Non Axial Fatigue Testing—Corrosion fatigue tests under loading conditions different from axial tension-tension may be
requested. In such cases established experimental arrangements for bending, rotating bending beam, or torsional testing may
replace the axial tension-tension mode. An environmental test chamber is attached to the equipment and the environmental tests
are carried out under conditions as described in this standard. Except for the mechanical testing arrangements the conditions of
this standard practice apply where possible. Reporting should follow Section 9 and should include all details where the testing
deviates from the standard procedure.
5.4 Environmental Chamber:
5.4.1 For corrosion fatigue testing, the machine shall be fitted with an environmental test cell surrounding the specimen gauge
section as shown in Fig. 1. A heated solution reservoir, a solution pump, and connecting lines for circulating the test solution to
the specimen surface are required. The solution should be pumped from the reservoir through the system at a rate that will maintain
the temperature at 37 6 1°C in the test cell, but with flow rates low enough to avoid flow-dependent phenomena like
erosion-corrosion. The reservoir should have a minimum capacity of 1000 mL per square centimeter of specimen surface exposed
to the electrolyte. The reservoir shall be vented to the atmosphere. If the solution volume decreases, the reservoir shall be
replenished with distilled water to maintain the saline concentration, or the solution should be exchanged. During long testing
periods exchange of the solution is recommended. A typical environmental test cell for axial fatigue testing is shown in Fig. 1.
5.4.2 The test equipment should be manufactured of materials or should be protected in such a manner that corrosion is avoided.
In particular galvanic corrosion in conjunction with the test specimen and loosening of the specimen grips due to corrosion must
be avoided.
6. Test Solution
6.1 To prepare the saline solution, dissolve 9 g of reagent-grade sodium chloride in distilled water and make up to 1000 mL. If
other typical Ringer’s solutions are used, note the solution a substitute for 0.9 % saline solution is used, such as one of the various
Ringer’s solutions, note that in the report.
7. Test Specimen
7.1 Specimen Design:
7.1.1 Axial Fatigue Testing:
7.1.1.1 The design of the axial load fatigue test specimens should comply to Practice E466 (see Fig. 2, Fig. 3, Fig. 4, and Fig.
5). For the dimensional proportions of flat specimens refer to the drawing in Practice E468. The ratio of the test section area to
end section area will depend on the specimen geometry and should comply to those standards. The test specimens specified in
PracticePractices E466 and Practice E468 are designed so that fatigue failure should occur in the section with reduced diameter
and not at the grip section.
7.1.1.2 For bending tests one may refer to the specimen configuration suggested in Practice E466.
F1801 − 20
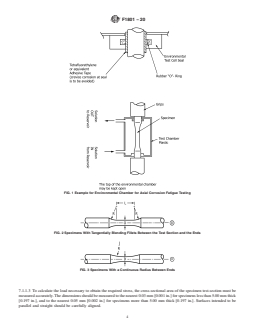
FIG. 1 Example for Environmental Chamber for Axial Corrosion Fatigue Testing
FIG. 2 Specimens With Tangentially Blending Fillets Between the Test Section and the Ends
FIG. 3 Specimens With a Continuous Radius Between Ends
7.1.1.3 To calculate the load necessary to obtain the required stress, the cross-sectional area of the specimen test-section must be
measured accurately. The di
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...