ASTM F1829-23
(Test Method)Standard Test Method for Static Evaluation of Anatomic Glenoid Locking Mechanism in Shear
Standard Test Method for Static Evaluation of Anatomic Glenoid Locking Mechanism in Shear
SIGNIFICANCE AND USE
4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the performance of metal or composite-backed anatomic glenoid prostheses’ locking mechanisms to resist static shear loading.
4.2 The glenoid component is used in shoulder replacements and should conform to the criteria specified in Specification F1378.
4.3 The loading of metal or composite-backed anatomic glenoid prostheses in vivo will, in general, differ from the loading defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method is designed to allow for comparisons between different metal or composite-backed anatomic glenoid locking mechanism designs, when tested under the same testing conditions.
4.4 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the materials being tested and their potential application.
4.5 In order for the test data on metal or composite-backed anatomic glenoid components to be comparable, reproducible, and capable of being correlated among laboratories, it is essential that uniform procedures be established.
SCOPE
1.1 This test method covers a method for determining the static shear disassembly force of modular anatomic glenoid components used in anatomic total shoulder arthroplasty prostheses.
1.2 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison of modular anatomic glenoid component designs and the strength of the retention mechanism between the articular insert and glenoid backing under the stated test conditions.
1.3 This test method covers modular glenoid components comprised of a separate articular insert and backing. The insert and backing can be fabricated from any combination of the following materials: metal alloys, polymeric materials, composite materials.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 28-Feb-2023
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 15-Dec-2018
- Effective Date
- 15-Dec-2018
- Effective Date
- 01-Dec-2017
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jan-2012
- Effective Date
- 15-Dec-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Jan-2007
- Effective Date
- 01-Oct-2005
- Effective Date
- 01-Mar-2004
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Nov-2002
Overview
ASTM F1829-23: Standard Test Method for Static Evaluation of Anatomic Glenoid Locking Mechanism in Shear is a critical standard developed by ASTM International. This document establishes a uniform test method for determining the static shear disassembly force of modular anatomic glenoid components used in total shoulder arthroplasty prostheses. The standard serves as a tool for comparing the effects of materials, design, and manufacturing processes on the performance of the locking mechanisms in metal or composite-backed anatomic glenoid prostheses. These implant systems are essential components in orthopedic shoulder replacements and must conform to high standards to ensure durability and safety.
Key Topics
Scope and Purpose
- Defines procedures for evaluating the static shear strength of modular glenoid prosthesis locking mechanisms.
- Designed for in vitro comparisons, the method provides a repeatable way to assess the retention strength between the articular insert and the glenoid backing.
- Applies to glenoid components made from metal alloys, polymeric, and composite materials.
Test Methodology
- Specifies the use of either mechanical or hydraulic load frames, ensuring accurate force application parallel to the intended implant axis.
- Testing is performed both in the inferior-to-superior and anterior-to-posterior directions, reflecting clinically relevant orientations.
- Requires testing specimens representative of final manufactured quality, with new inserts used for each test.
Reporting and Data Integrity
- Mandates comprehensive documentation, including material details, displacement rates, maximum loads, failure modes, and load-displacement curves.
- Insists on uniform procedures to promote consistency, reproducibility, and comparability across different laboratories.
Significance and Limitations
- While the method does not replicate all physiological forces experienced in vivo, it provides valuable comparative data for design evaluation.
- Users must exercise judgment in determining appropriateness for specific materials and applications.
- Results should not be interpreted as direct clinical performance indicators.
Applications
Orthopedic Device Development
- Enables device manufacturers and researchers to evaluate and optimize the locking mechanisms of anatomic glenoid components, contributing to increased implant longevity and reduced risk of device failure.
- Assists in the selection of materials and design features that enhance the mechanical stability of glenoid prostheses.
Quality Assurance and Product Comparisons
- Utilized during product development and quality control to benchmark new designs against established criteria.
- Supports regulatory compliance and facilitates submission processes by demonstrating adherence to standardized test methods.
Interlaboratory Studies
- Provides a basis for repeatable, comparable assessments between laboratories and organizations, ensuring objective evaluation of shoulder prosthesis components.
Preclinical Research
- Serves as a foundation for further in vitro testing, including dynamic fatigue and worst-case scenario studies, which are integral to comprehensive preclinical evaluation.
Related Standards
- ASTM F1378: Specification for Shoulder Prostheses - outlines criteria for glenoid components used in shoulder replacements.
- ASTM F2028: Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation - complements static shear testing with dynamic performance evaluation.
- ASTM E4: Practices for Force Calibration and Verification of Testing Machines - ensures equipment accuracy essential for reliable test results.
By enabling structured and reproducible static shear testing of modular glenoid locking mechanisms, ASTM F1829-23 contributes to the safe and effective development of orthopedic implants for total shoulder arthroplasty, supporting long-term clinical outcomes and regulatory standards in the medical device industry.
Buy Documents
ASTM F1829-23 - Standard Test Method for Static Evaluation of Anatomic Glenoid Locking Mechanism in Shear
REDLINE ASTM F1829-23 - Standard Test Method for Static Evaluation of Anatomic Glenoid Locking Mechanism in Shear
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1829-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Static Evaluation of Anatomic Glenoid Locking Mechanism in Shear". This standard covers: SIGNIFICANCE AND USE 4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the performance of metal or composite-backed anatomic glenoid prostheses’ locking mechanisms to resist static shear loading. 4.2 The glenoid component is used in shoulder replacements and should conform to the criteria specified in Specification F1378. 4.3 The loading of metal or composite-backed anatomic glenoid prostheses in vivo will, in general, differ from the loading defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method is designed to allow for comparisons between different metal or composite-backed anatomic glenoid locking mechanism designs, when tested under the same testing conditions. 4.4 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the materials being tested and their potential application. 4.5 In order for the test data on metal or composite-backed anatomic glenoid components to be comparable, reproducible, and capable of being correlated among laboratories, it is essential that uniform procedures be established. SCOPE 1.1 This test method covers a method for determining the static shear disassembly force of modular anatomic glenoid components used in anatomic total shoulder arthroplasty prostheses. 1.2 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison of modular anatomic glenoid component designs and the strength of the retention mechanism between the articular insert and glenoid backing under the stated test conditions. 1.3 This test method covers modular glenoid components comprised of a separate articular insert and backing. The insert and backing can be fabricated from any combination of the following materials: metal alloys, polymeric materials, composite materials. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the performance of metal or composite-backed anatomic glenoid prostheses’ locking mechanisms to resist static shear loading. 4.2 The glenoid component is used in shoulder replacements and should conform to the criteria specified in Specification F1378. 4.3 The loading of metal or composite-backed anatomic glenoid prostheses in vivo will, in general, differ from the loading defined in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method is designed to allow for comparisons between different metal or composite-backed anatomic glenoid locking mechanism designs, when tested under the same testing conditions. 4.4 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the appropriateness of the method in view of the materials being tested and their potential application. 4.5 In order for the test data on metal or composite-backed anatomic glenoid components to be comparable, reproducible, and capable of being correlated among laboratories, it is essential that uniform procedures be established. SCOPE 1.1 This test method covers a method for determining the static shear disassembly force of modular anatomic glenoid components used in anatomic total shoulder arthroplasty prostheses. 1.2 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison of modular anatomic glenoid component designs and the strength of the retention mechanism between the articular insert and glenoid backing under the stated test conditions. 1.3 This test method covers modular glenoid components comprised of a separate articular insert and backing. The insert and backing can be fabricated from any combination of the following materials: metal alloys, polymeric materials, composite materials. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1829-23 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1829-23 has the following relationships with other standards: It is inter standard links to ASTM F1378-18e1, ASTM F1378-18, ASTM F1378-17, ASTM E4-14, ASTM F1378-12, ASTM F1378-05(2010), ASTM E4-10, ASTM E4-09a, ASTM E4-09, ASTM E4-08, ASTM E4-07, ASTM F1378-05, ASTM F1378-04, ASTM E4-03, ASTM E4-02. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1829-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F1829 − 23
Standard Test Method for
Static Evaluation of Anatomic Glenoid Locking Mechanism
in Shear
This standard is issued under the fixed designation F1829; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This test method covers a method for determining the 2.1 ASTM Standards:
static shear disassembly force of modular anatomic glenoid E4 Practices for Force Calibration and Verification of Test-
components used in anatomic total shoulder arthroplasty pros- ing Machines
theses. F1378 Specification for Shoulder Prostheses
1.2 Although the methodology described does not replicate
3. Terminology
all physiological force conditions, it is a means of in vitro
3.1 Anatomic Total Shoulder Replacement (TSR) Defini-
comparison of modular anatomic glenoid component designs
tions:
and the strength of the retention mechanism between the
3.1.1 anatomic total shoulder arthroplasty system,
articular insert and glenoid backing under the stated test
n—shoulder implant system that has a concave glenoid com-
conditions.
ponent and a convex humeral component design.
1.3 This test method covers modular glenoid components
3.1.2 anatomic glenoid component, n—the concave pros-
comprised of a separate articular insert and backing. The insert
thetic portion that replaces, in part or in total, the glenoid fossa
and backing can be fabricated from any combination of the
of the scapula and articulates with the natural humeral head or
following materials: metal alloys, polymeric materials, com-
a prosthetic replacement.
posite materials.
3.1.3 glenoid backing, n—the metallic or composite mate-
1.4 The values stated in SI units are to be regarded as
rial prosthetic portion of a multi-piece anatomic glenoid
standard. No other units of measurement are included in this
component that attaches to the scapula.
standard.
3.1.4 glenoid liner, n—the polymeric prosthetic portion of a
1.5 This standard does not purport to address all of the
multiple-piece anatomic glenoid component that articulates
safety concerns, if any, associated with its use. It is the
with the humeral head.
responsibility of the user of this standard to establish appro-
3.2 Additional Definitions:
priate safety, health, and environmental practices and deter-
3.2.1 collar, n—flange at the junction of the humeral neck
mine the applicability of regulatory limitations prior to use.
and stem.
1.6 This international standard was developed in accor-
dance with internationally recognized principles on standard-
3.2.2 keel, (or pegs), n—single or multiple projections that
ization established in the Decision on Principles for the
provide resistance to translation or rotation of the glenoid
Development of International Standards, Guides and Recom-
component, or both, by mating with cavities created in the
mendations issued by the World Trade Organization Technical
glenoid fossa.
Barriers to Trade (TBT) Committee.
3.2.3 neck, n—segment connecting the head and the stem.
3.2.4 glenoid plane, n—in symmetrical anatomic glenoids,
the glenoid plane is defined by joining the two articular edges;
This test method is under the jurisdiction of ASTM Committee F04 on Medical
and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved March 1, 2023. Published March 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1997. Last previous edition approved in 2017 as F1829 – 17. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/F1829-23. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1829 − 23
in planar and asymmetric anatomic glenoids, it is defined by between different metal or composite-backed anatomic glenoid
the back (medial) surface. locking mechanism designs, when tested under the same
testing conditions.
3.2.4.1 Discussion—Although the glenoid fossa is not truly
4.4 This test method may not be appropriate for all types of
a planar structure, the terms plane of the glenoid and glenoid
implant applications. The user is cautioned to consider the
plane have both been used in the scientific literature to describe
appropriateness of the method in view of the materials being
the anatomic orientation of the glenoid.
tested and their potential application.
3.2.5 humeral head, n—the bearing member that articulates
with the glenoid.
4.5 In order for the test data on metal or composite-backed
anatomic glenoid components to be comparable, reproducible,
3.2.6 humeral component, n—the prosthetic portion that
and capable of being correlated among laboratories, it is
replaces, in part or in total, the proximal humerus or humeral
essential that uniform procedures be established.
head and articulates with the natural glenoid fossa or a
prosthetic replacement.
5. Apparatus
3.2.7 humeral stem, n—segment intended for insertion
5.1 The test fixture shall be constructed so that the line of
within the humeral medullary canal.
load application is parallel to the intended axis of the implant
3.3 Definitions of Terms Specific to This Standard:
(that is, inferior to superior or anterior to posterior).
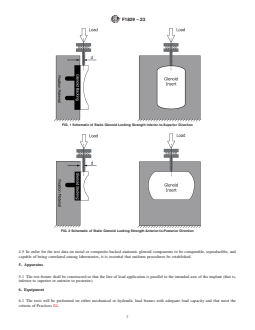
3.3.1 “d”, n—offset distance from the edge of the glenoid
backing locking mechanism to the centerline of the point of
6. Equipment
force application on the articular insert as shown in Figs. 1 and
6.1 The tests will be performed on either mechanical or
2.
hydraulic load frames with adequate load capacity and that
meet the criteria of Practices E4.
4. Significance and Use
4.1 This test method can be used to describe the effects of
7. Sampling
materials, manufacturing, and design variables on the perfor-
7.1 A minimum of five samples with the load oriented in the
mance of metal or composite-backed anatomic glenoid pros-
inferior-to-superior direction shall be tested per device.
theses’ locking mechanisms to resist static shear loading.
7.2 A minimum of five samples with the load oriented in the
4.2 The glenoid component is used in shoulder replace-
anterior-to-posterior direction shall be tested per device.
ments and should conform to the criteria specified in Specifi-
cation F1378.
8. Sample and Test Specimen
4.3 The loading of metal or composite-backed anatomic
8.1 All articular insert test components shall be representa-
glenoid prostheses in vivo will, in general, differ from the
tive of final manufac
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1829 − 17 F1829 − 23
Standard Test Method for
Static Evaluation of Anatomic Glenoid Locking Mechanism
in Shear
This standard is issued under the fixed designation F1829; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers a method for determining the static shear disassembly force of modular anatomic glenoid components
used in anatomic total shoulder arthroplasty prostheses.
1.2 Although the methodology described does not replicate all physiological force conditions, it is a means of in vitro comparison
of modular anatomic glenoid component designs and the strength of the retention mechanism between the articular insert and
glenoid backing under the stated test conditions.
1.3 This test method covers modular glenoid components comprised of a separate articular insert and backing. The insert and
backing can be fabricated from any combination of the following materials: metal alloys, polymeric materials, composite materials.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Calibration and Verification of Testing Machines
F1378 Specification for Shoulder Prostheses
F2028 Test Methods for Dynamic Evaluation of Glenoid Loosening or Disassociation
3. Terminology
3.1 Anatomic Total Shoulder Replacement (TSR) DefinitionsDefinitions:
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty.
Current edition approved Dec. 1, 2017March 1, 2023. Published January 2018March 2023. Originally approved in 1997. Last previous edition approved in 20162017 as
F1829 – 16.F1829 – 17. DOI: 10.1520/F1829-17.10.1520/F1829-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1829 − 23
3.1.1 anatomic total shoulder arthroplasty system, n—shoulder implant system that has a concave glenoid component and a
convex humeral component design.
3.1.2 anatomic glenoid component, n—the concave prosthetic portion that replaces, in part or in total, the glenoid fossa of the
scapula and articulates with the natural humeral head or a prosthetic replacement.
3.1.3 glenoid backing, n—the metallic or composite material prosthetic portion of a multi-piece anatomic glenoid component that
attaches to the scapula.
3.1.4 glenoid liner, n—the polymeric prosthetic portion of a multiple piece multiple-piece anatomic glenoid component that
articulates with the humeral head.
3.2 Additional DefinitionsDefinitions:
3.2.1 collar, n—flange at the junction of the humeral neck and stem.
3.2.2 keel, (or pegs), n—single or multiple projections that provide resistance to translation or rotation of the glenoid component,
or both, by mating with cavities created in the glenoid fossa.
3.2.3 neck, n—segment connecting the head and the stem.
3.2.4 glenoid plane, n—in symmetrical anatomic glenoids, the glenoid plane is defined by joining the two articular edges; in planar
and asymmetric anatomic glenoids, it is defined by the back (medial) surface.
3.2.4.1 Discussion—
Although the glenoid fossa is not truly a planar structure, the terms plane of the glenoid and glenoid plane have both been used
in the scientific literature to describe the anatomic orientation of the glenoid.
3.2.5 humeral head, n—the bearing member that articulates with the glenoid.
3.2.6 humeral component, n—the prosthetic portion that replaces, in part or in total, the proximal humerus or humeral head and
articulates with the natural glenoid fossa or a prosthetic replacement.
3.2.7 humeral stem, n—segment intended for insertion within the humeral medullary canal.
3.3 Definitions of Terms Specific to This Standard:
3.3.1 “d”, n—offset distance from the edge of the glenoid backing locking mechanism to the centerline of the point of force
application on the articular insert as shown in Fig. 1Figs. 1 and 2 and . Fig. 2.
4. Significance and Use
4.1 This test method can be used to describe the effects of materials, manufacturing, and design variables on the performance of
metal or composite-backed anatomic glenoid prostheses’ locking mechanisms to resist static shear loading.
4.2 The glenoid component is used in shoulder replacements and should conform to the criteria specified in Specification F1378.
4.3 The loading of metal or composite-backed anatomic glenoid prostheses in vivo will, in general, differ from the loading defined
in this test method. The results obtained here cannot be used to directly predict in vivo performance. However, this test method
is designed to allow for comparisons between different metal or composite-backed anatomic glenoid locking mechanism designs,
when tested under similar circumstances.the same testing conditions.
4.4 This test method may not be appropriate for all types of implant applications. The user is cautioned to consider the
appropriateness of the method in view of the materials being tested and their potential application.
F1829 − 23
FIG. 1 Schematic of Static Glenoid Locking Strength Inferior-to-Superior Direction
FIG. 2 Schematic of Static Glenoid Locking Strength Anterior-to-Posterior Direction
4.5 In order for the test data on metal or composite-backed anatomic glenoid components to be comparable, reproducible, and
capable of being correlated among laboratories, it is essential that uniform procedures be established.
5. Apparatus
5.1 The test fixture shall be constructed so that the line of load application is parallel to the intended axis of the implant (that is,
inferior to superior or anterior to posterior).
6. Equipment
6.1 The tests will be performed on either mechanical or hydraulic load frames with adequate load capacity and that meet the
criteri
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...