ASTM D7096-19
(Test Method)Standard Test Method for Determination of the Boiling Range Distribution of Gasoline by Wide-Bore Capillary Gas Chromatography
Standard Test Method for Determination of the Boiling Range Distribution of Gasoline by Wide-Bore Capillary Gas Chromatography
SIGNIFICANCE AND USE
5.1 The determination of the boiling range distribution of gasoline by gas chromatographic simulated distillation provides an insight into the composition of the components from which the gasoline has been blended. Knowledge of the boiling range distribution of gasoline blending components is useful for the control of refinery processes and for the blending of finished gasoline.
5.2 The determination of the boiling range distribution of light hydrocarbon mixtures by gas chromatographic simulated distillation has better precision than the conventional distillation by Test Method D86. Additionally, this test method provides more accurate and detailed information about the composition of the light ends. The distillation data produced by this test method are similar to that which would be obtained from a cryogenic, true boiling point (15 theoretical plates) distillation.
SCOPE
1.1 This test method covers the determination of the boiling range distribution of gasoline and liquid gasoline blending components. It is applicable to petroleum products and fractions with a final boiling point of 280 °C (536 °F) or lower, as measured by this test method.
1.2 This test method is designed to measure the entire boiling range of gasoline and gasoline components with either high or low vapor pressure and is commonly referred to as Simulated Distillation (SimDis) by gas chromatographers.
1.3 This test method has been validated for gasoline containing ethanol. Gasolines containing other oxygenates are not specifically excluded, but they were not used in the development of this test method.
1.4 This test method can estimate the concentration of n-pentane and lighter saturated hydrocarbons in gasoline.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5.1 Results in degrees Fahrenheit can be obtained by simply substituting Fahrenheit boiling points in the calculation of the boiling point-retention time correlation.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2019
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.04.0H - Chromatographic Distribution Methods
Relations
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Dec-2023
- Effective Date
- 01-Oct-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Dec-2019
- Effective Date
- 01-Dec-2019
- Refers
ASTM D4626-95(2019) - Standard Practice for Calculation of Gas Chromatographic Response Factors - Effective Date
- 01-Dec-2019
- Effective Date
- 01-Sep-2019
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Apr-2018
Overview
ASTM D7096-19 sets forth the standard test method for determining the boiling range distribution of gasoline and gasoline blending components using wide-bore capillary gas chromatography (often termed Simulated Distillation or SimDis). Published by ASTM International, this standard provides a precise and detailed approach for analyzing gasoline fractions up to a final boiling point of 280 °C (536 °F).
The method offers significant advantages over conventional atmospheric distillation (such as ASTM D86), including improved resolution and precise characterization of light hydrocarbon mixtures. This is crucial for process control within refineries and for optimizing gasoline blending, contributing to fuel performance and regulatory compliance.
Key Topics
- Simulated Distillation (SimDis) by Gas Chromatography: Employs a non-polar wide-bore capillary column to separate hydrocarbons in order of boiling point, offering a reliable profile of gasoline composition.
- Boiling Range Distribution Analysis: Enables measurement of initial and final boiling points, as well as the composition of blending components, for both high and low vapor pressure gasoline products.
- Enhanced Precision: Provides greater repeatability and detailed information for light ends (such as n-pentane and lighter saturated hydrocarbons) compared to traditional distillation.
- Ethanol and Oxygenate Detection: Validated for gasolines containing ethanol; other oxygenates are not specifically excluded but require careful response factor calibration due to differences in detector response.
- Calibration and Data Integrity: Utilizes reference calibration mixtures and advanced data acquisition systems to ensure accuracy throughout the analysis.
Applications
ASTM D7096-19 is widely used across the petroleum industry for the following applications:
- Refinery Process Control: Accurate boiling range distribution informs adjustments in distillation and blending operations, leading to consistent, high-quality gasoline production.
- Quality Assurance of Gasoline: Detailed analysis supports adherence to fuel standards and regulatory requirements for gasoline and its blending components.
- Blending Optimization: Insight into individual component distributions allows refineries to tailor blends for performance, emissions, and compliance.
- Research and Development: Useful for evaluating the properties of new fuels, including blends with ethanol or other oxygenates.
- Comparison with Other Methods: Offers improved precision relative to ASTM D86, allowing for better control and understanding of the product's volatility profile.
Related Standards
When implementing ASTM D7096-19, the following ASTM standards and practices are commonly referenced or used in conjunction:
- ASTM D86 - Standard Test Method for Distillation of Petroleum Products and Liquid Fuels at Atmospheric Pressure.
- ASTM D4057 - Practice for Manual Sampling of Petroleum and Petroleum Products.
- ASTM D3700 - Practice for Obtaining LPG Samples Using a Floating Piston Cylinder.
- ASTM D4626 - Practice for Calculation of Gas Chromatographic Response Factors.
- ASTM D4815 - Test Method for Determination of MTBE, ETBE, TAME, DIPE, tertiary-Amyl Alcohol and C1 to C4 Alcohols in Gasoline by Gas Chromatography.
- ASTM D5191 - Test Method for Vapor Pressure of Petroleum Products and Liquid Fuels (Mini Method).
- ASTM E1510 - Practice for Installing Fused Silica Open Tubular Capillary Columns in Gas Chromatographs.
Practical Value
Adopting ASTM D7096-19 delivers:
- Reliable boiling range distribution data for gasoline and blending components
- Improved efficiency in monitoring and controlling refinery operations
- Enhanced ability to meet fuel specification requirements and environmental standards
- Seamless alignment with other ASTM and international test methods for comprehensive petroleum analysis
By offering a standardized, reproducible approach to boiling range determination, ASTM D7096-19 supports the petroleum industry’s commitment to quality, safety, and regulatory compliance.
Buy Documents
ASTM D7096-19 - Standard Test Method for Determination of the Boiling Range Distribution of Gasoline by Wide-Bore Capillary Gas Chromatography
REDLINE ASTM D7096-19 - Standard Test Method for Determination of the Boiling Range Distribution of Gasoline by Wide-Bore Capillary Gas Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D7096-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of the Boiling Range Distribution of Gasoline by Wide-Bore Capillary Gas Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 The determination of the boiling range distribution of gasoline by gas chromatographic simulated distillation provides an insight into the composition of the components from which the gasoline has been blended. Knowledge of the boiling range distribution of gasoline blending components is useful for the control of refinery processes and for the blending of finished gasoline. 5.2 The determination of the boiling range distribution of light hydrocarbon mixtures by gas chromatographic simulated distillation has better precision than the conventional distillation by Test Method D86. Additionally, this test method provides more accurate and detailed information about the composition of the light ends. The distillation data produced by this test method are similar to that which would be obtained from a cryogenic, true boiling point (15 theoretical plates) distillation. SCOPE 1.1 This test method covers the determination of the boiling range distribution of gasoline and liquid gasoline blending components. It is applicable to petroleum products and fractions with a final boiling point of 280 °C (536 °F) or lower, as measured by this test method. 1.2 This test method is designed to measure the entire boiling range of gasoline and gasoline components with either high or low vapor pressure and is commonly referred to as Simulated Distillation (SimDis) by gas chromatographers. 1.3 This test method has been validated for gasoline containing ethanol. Gasolines containing other oxygenates are not specifically excluded, but they were not used in the development of this test method. 1.4 This test method can estimate the concentration of n-pentane and lighter saturated hydrocarbons in gasoline. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5.1 Results in degrees Fahrenheit can be obtained by simply substituting Fahrenheit boiling points in the calculation of the boiling point-retention time correlation. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The determination of the boiling range distribution of gasoline by gas chromatographic simulated distillation provides an insight into the composition of the components from which the gasoline has been blended. Knowledge of the boiling range distribution of gasoline blending components is useful for the control of refinery processes and for the blending of finished gasoline. 5.2 The determination of the boiling range distribution of light hydrocarbon mixtures by gas chromatographic simulated distillation has better precision than the conventional distillation by Test Method D86. Additionally, this test method provides more accurate and detailed information about the composition of the light ends. The distillation data produced by this test method are similar to that which would be obtained from a cryogenic, true boiling point (15 theoretical plates) distillation. SCOPE 1.1 This test method covers the determination of the boiling range distribution of gasoline and liquid gasoline blending components. It is applicable to petroleum products and fractions with a final boiling point of 280 °C (536 °F) or lower, as measured by this test method. 1.2 This test method is designed to measure the entire boiling range of gasoline and gasoline components with either high or low vapor pressure and is commonly referred to as Simulated Distillation (SimDis) by gas chromatographers. 1.3 This test method has been validated for gasoline containing ethanol. Gasolines containing other oxygenates are not specifically excluded, but they were not used in the development of this test method. 1.4 This test method can estimate the concentration of n-pentane and lighter saturated hydrocarbons in gasoline. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5.1 Results in degrees Fahrenheit can be obtained by simply substituting Fahrenheit boiling points in the calculation of the boiling point-retention time correlation. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7096-19 is classified under the following ICS (International Classification for Standards) categories: 75.160.20 - Liquid fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7096-19 has the following relationships with other standards: It is inter standard links to ASTM D6300-24, ASTM D4814-24, ASTM D6300-23a, ASTM D86-23a, ASTM D4814-23a, ASTM D86-23ae1, ASTM D4626-23, ASTM D4814-20, ASTM D4814-19a, ASTM D6300-19a, ASTM D4626-95(2019), ASTM E594-96(2019), ASTM D5191-18a, ASTM D4814-18c, ASTM D4814-18a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7096-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7096 − 19
Standard Test Method for
Determination of the Boiling Range Distribution of Gasoline
by Wide-Bore Capillary Gas Chromatography
This standard is issued under the fixed designation D7096; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 Thistestmethodcoversthedeterminationoftheboiling
D86Test Method for Distillation of Petroleum Products and
range distribution of gasoline and liquid gasoline blending
Liquid Fuels at Atmospheric Pressure
components. It is applicable to petroleum products and frac-
D2421Practice for Interconversion of Analysis of C and
tions with a final boiling point of 280°C (536°F) or lower, as
Lighter Hydrocarbons to Gas-Volume, Liquid-Volume, or
measured by this test method.
Mass Basis
1.2 This test method is designed to measure the entire
D3700Practice for Obtaining LPG Samples Using a Float-
boiling range of gasoline and gasoline components with either
ing Piston Cylinder
high or low vapor pressure and is commonly referred to as
D4057Practice for Manual Sampling of Petroleum and
Simulated Distillation (SimDis) by gas chromatographers.
Petroleum Products
1.3 This test method has been validated for gasoline con-
D4307Practice for Preparation of Liquid Blends for Use as
taining ethanol. Gasolines containing other oxygenates are not
Analytical Standards
specifically excluded, but they were not used in the develop-
D4626Practice for Calculation of Gas Chromatographic
ment of this test method.
Response Factors
D4814Specification for Automotive Spark-Ignition Engine
1.4 This test method can estimate the concentration of
Fuel
n-pentane and lighter saturated hydrocarbons in gasoline.
D4815Test Method for Determination of MTBE, ETBE,
1.5 The values stated in SI units are to be regarded as
TAME, DIPE, tertiary-Amyl Alcohol and C to C Alco-
1 4
standard. No other units of measurement are included in this
hols in Gasoline by Gas Chromatography
standard.
D5191Test Method for Vapor Pressure of Petroleum Prod-
1.5.1 Results in degrees Fahrenheit can be obtained by
ucts and Liquid Fuels (Mini Method)
simply substituting Fahrenheit boiling points in the calculation
D5599Test Method for Determination of Oxygenates in
of the boiling point-retention time correlation.
Gasoline by Gas Chromatography and Oxygen Selective
Flame Ionization Detection
1.6 This standard does not purport to address all of the
D6300Practice for Determination of Precision and Bias
safety concerns, if any, associated with its use. It is the
Data for Use in Test Methods for Petroleum Products and
responsibility of the user of this standard to establish appro-
Lubricants
priate safety, health, and environmental practices and deter-
E594Practice for Testing Flame Ionization Detectors Used
mine the applicability of regulatory limitations prior to use.
in Gas or Supercritical Fluid Chromatography
1.7 This international standard was developed in accor-
E1510Practice for Installing Fused Silica Open Tubular
dance with internationally recognized principles on standard-
Capillary Columns in Gas Chromatographs
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
3. Terminology
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee. 3.1 Definitions:
3.1.1 area slice, n—area under a chromatogram within a
specified retention time interval.
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.04.0H on Chromatographic Distribution Methods. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved May 1, 2019. Published June 2019. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2005. Last previous edition approved in 2016 as D7096–16. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D7096-19. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7096 − 19
3.1.2 final boiling point (FBP), n—the point at which a substantially different from the response of hydrocarbons,
cumulative volume count equal to 99.5% of the total volume responsefactorsareusedtocorrecttheareaslicefortheelution
counts under the chromatogram is obtained. interval of oxygenates.
3.1.3 initial boiling point (IBP), n—the point at which a
6.2 Concentrations of n-pentane and lighter saturated com-
cumulative volume count equal to 0.5% of the total volume
pounds may be estimated from the analysis. However, early
counts under the chromatogram is obtained.
eluting olefins present in the gasoline samples may coelute
with these compounds.
3.1.4 relative volume response factor (RVRF), n—the vol-
umeresponsefactor(see3.1.8)ofacomponent irelativetothe
6.3 For samples containing ethanol, this test method will
volume response factor of n-heptane.
determinethehydrocarbondistribution.Itwillnotsimulatethe
3.1.5 slice time, n—the retention time at the end of a given
azeotrope observed during physical distillation.
area slice.
7. Apparatus
3.1.6 slice width, n—the fixed duration (1 s, or less) of the
retention time intervals into which the chromatogram is di-
7.1 Gas Chromatograph—Any gas chromatograph (GC)
vided. It is determined from the reciprocal of the frequency
designed for use with wide-bore (0.53 mm inside diameter)
used in the acquisition of data.
capillarycolumns,thatmeetstheperformancecriteriaspecified
in Section 11, and has the following features may be used.
3.1.7 volume count, n—the product of a slice area (or an
Typical operating conditions are shown in Table 1.
area under a peak) and a volume response factor.
7.1.1 Column Oven Temperature Programming—The gas
3.1.8 volume response factor, n—a constant of proportion-
chromatograph shall be capable of linear temperature-
ality that relates the area under a chromatogram to liquid
programmed operation from −40°C to 280°C at rates up to
volume.
25°C⁄min.
4. Summary of Test Method
7.1.2 Injection Port—The injection port shall be capable of
operation at temperatures required to completely volatize and
4.1 The sample is vaporized and transported by carrier gas
transfer the sample to the GC column. Non-splitting or
into a non-polar, wide-bore capillary gas chromatographic
split/splitless vaporizing sample ports optimized for use with
column. The column temperature is raised at a reproducible,
wide-bore capillary columns are acceptable. If using a split
linearratesoastoelutethehydrocarboncomponentsinboiling
inletport,itshouldbedesignedtoprovidealinearsamplesplit
point order for measurement by a flame ionization detector.
injection.
Conditions are selected such that n-pentane and lighter satu-
7.1.3 Flame Ionization Detector—The detector shall be
rated hydrocarbons in the calibration mixture are resolved
optimized for the use of wide-bore capillary gas chromato-
discretely. Linear correlation between hydrocarbon boiling
graphic columns and shall conform to the specifications as
point and retention time is established using a known mixture
described in Practice E594.
of hydrocarbons covering the boiling range expected in the
7.1.4 Carrier Gas Controls—The associated carrier gas
sample. Area slices are converted to volume using theoretical
controlsshallbeofsufficientprecisiontoproducereproducible
hydrocarbon volume response factors. Oxygenated samples
column flows in order to maintain analytical integrity.
require experimental determination of oxygenate response
factors.
5. Significance and Use
TABLE 1 Typical Operating Conditions for Wide Bore
5.1 The determination of the boiling range distribution of Column Inlets
gasoline by gas chromatographic simulated distillation pro-
Column length (m) 30 15
Column I.D. (mm) 0.53 0.53
vides an insight into the composition of the components from
Stationary phase 100 % poly- 100 % poly-
whichthegasolinehasbeenblended.Knowledgeoftheboiling
dimethylsiloxane dimethylsiloxane
range distribution of gasoline blending components is useful
Film thickness (µm) 5 5
Carrier gas helium helium
for the control of refinery processes and for the blending of
Carrier flow (mL/min) 20 15
finished gasoline.
Auxiliary flow (mL/min) 10 10
Column initial temperature 40 40
5.2 The determination of the boiling range distribution of
(°C)
light hydrocarbon mixtures by gas chromatographic simulated
Initial time (min) 1 1
distillation has better precision than the conventional distilla-
Program rate (°C/min) 25 20
Final temperature (°C) 265 230
tion by Test Method D86. Additionally, this test method
Final hold (min) 4.00 2.50
provides more accurate and detailed information about the
Injection inlet purged-packed purged-packed
compositionofthelightends.Thedistillationdataproducedby
Sample introduction auto syringe auto syringe
injection injection
this test method are similar to that which would be obtained
Injector temperature (°C) 250 250
from a cryogenic, true boiling point (15 theoretical plates)
Detector temperature (°C) 280 300
distillation. Hydrogen flow (mL/min) 45 30
Air flow (mL/min) 450 300
6. Interferences Sample size (µL) 0.1 – 0.2 0.2
Area slice width (s) 0.5 – 0.2 0.5 – 0.2
6.1 Ethanolorotheroxygenatesmaycoelutewithhydrocar-
Datarate(Hz) 2–5 2–5
bonspresentinthesample.Sincetheresponseofoxygenatesis
D7096 − 19
TABLE 2 Typical Operating Conditions for Capillary Column Inlet
quency of 2Hz to 5 Hz. The software should also be able to
Column length (m) 30 store the data for future recall, inspection, and analysis. The
Column I.D. (mm) 0.53
dataacquisitionsoftwareshouldbecapableofpresentingareal
Stationary phase 100 %
time plot. It may also be capable of controlling the operating
polydimethylsiloxane
Film thickness 5 µm variables of the gas chromatograph. Specialized software is
Carrier gas helium (ramped flow)
necessary to obtain the boiling point distribution.
Carrier flow (mL/min) 5 mL ⁄min (0.5 min) to
20 mL ⁄min @
7.5 Bulk Sample Containers, floating piston cylinders (see
60 mL ⁄min
9.1.1); epoxy phenolic-lined metal cans; glass bottles with
Column initial temperature (°C) 40
Initial time (min) 1
polytetrafluoroethylene-lined screw caps.
Program rate (°C/min) 25
Final temperature (°C) 245
8. Reagents and Materials
Final hold (min) 4
Injection port split
8.1 Calibration Mixture—Asyntheticmixtureofpureliquid
Sample introduction automatic syringe
injection
hydrocarbons with boiling points that encompass the boiling
Injector temperature (°C) 250
range of the sample shall be used for retention time determi-
Detector temperature (°C) 250
nation and response factor validation. Response factors for
Hydrogen flow (mL/min) 30
Air flow (mL/min) 300
propane, isobutane, and n-butane are extrapolated from the
Sample size (µL) 1 uL
relative molar response of the n-paraffins. An example of a
Split ratio 1:50
relative response factor mixture with suggested nominal com-
Data rate 5 Hz
position is given in Table 3. This mixture shall be accurately
prepared on a mass basis using Practice D4307 or equivalent.
8.1.1 Asinglecalibrationstandardmaybeusedforretention
7.1.5 Baseline Correction—The gas chromatograph (or an-
time-boilingpointdeterminationandresponsefactorvalidation
other component of the gas chromatographic system) shall be
provided isopentane and heavier components are known quan-
capable of subtracting the area slice of a blank run from the
titatively. Gaseous components propane, isobutane, and
corresponding area slice of a sample run. This can be done
n-butane are added in small quantities (< 0.2 volume % each).
internally on some gas chromatographs (baseline compensa-
These small quantities do not significantly change the concen-
tion)orexternallybysubtractingastored,digitizedsignalfrom
trations of the remaining hydrocarbons. This standard would
a blank run.
also be used for measuring performance criteria in Section 11.
7.2 Sample Introduction—Sample introduction may be by It may be practical to generate this standard by bubbling a
meansofaconstantvolumeliquidsamplevalveorbyinjection
small amount of gaseous propane, isobutane, and n-butane
with a micro syringe through a septum. An automatic sample (Warning—Extremely flammable gases.) into a quantitative
introduction device is essential to the reproducibility of the
mixture of isopentane and heavier components.
analysis. Manual injections are not recommended. Poor injec-
8.1.2 A combination of two calibration standards may also
tiontechniquecanresultinpoorresolution.Ifcolumnoverload
be used.Aquantitative standard, containing known concentra-
occurs, peak skewing may result, leading to variation in
tions of isopentane and heavier compounds, is used to deter-
retention times.
mine response factors. A qualitative standard, containing a
7.2.1 Samples with a vapor pressure (VP) of less than
wideboilingrangeofcompoundsincludingpropane, n-butane,
16psiaasmeasuredbyTestMethodD5191,orequivalent,may
and isobutane is used for measuring the retention time-boiling
be introduced into the gas chromatograph by syringe injection
point relationship and establishing the performance criteria
into a heated, vaporizing inlet. Samples with vapor pressures
outlined in Section 11.
between 12psia and 16 psia should be kept chilled (refriger-
8.2 Calibration Mixture with Oxygenates—When samples
atedorinacooledsampletray)andmayrequireinjectionwith
to be measured contain oxygenates, the calibration mixture
a cooled syringe. Samples with a vapor pressure above 16psia
(see 8.1) shall also contain the oxygenates. Therefore, the
should be introduced by way of a constant volume liquid
identity of the oxygenate(s) shall be known prior to analysis of
sampling valve. Refer to 9.1 for sampling practices.
the sample. Oxygenate content may be determined by Test
7.3 Column—Any wide bore (0.53 mm inside diameter)
Method D4815, Test Method D5599, or equivalent.
open tubular (capillary) column, coated with a non-polar
Oxygenates,suchasethanol,shouldbeaddedtothecalibration
(100% polydimethylsiloxane) phase that meets the perfor-
mixtureatanapproximateconcentrationasthatinthesamples.
mance criteria of Section 11 may be used. Columns of 15m to
This mixture is used to define the retention time boundary and
30m lengths with a stationary phase film thickness of 5.0µm
relative volume response of the oxygenate to be applied to this
have been successfully used. With either of these columns,
region. For gasoline containing other oxygenates, determine if
initial cryogenic temperatures are not necessary.
the oxygenate coelutes with any of the hydrocarbons listed in
7.4 Data Acquisition System—A computer provided with a Table 3. If a coelution occurs, the coeluting hydrocarbon
monitor, printer, and data acquisition software is necessary to should not be included in the blend. Typical compositions of
carry out this analysis. The computer should have sufficient oxygenated blends are given in Table 4. Typical relative
hardware capacity and random access memory in order to run volume response factors, molecular weights, and densities for
the data acquisition program while acquiring data at a fre- various oxygenated compounds are provided in Table 5.
D7096 − 19
TABLE 3 Typical Calibration Mixture Composition and Properties of Hydrocarbons
A
A
BP Relative Density
B C
Nominal Approx. FID
Compound 15.6/15.6 °C
Vol% RVRF
Mass%
°C °F
(60/60°F)
D
Propane –42.1 –43.8 0.5070 – – 1.394
D
Isobutane –11.8 10.8 0.5629 – – 1.241
D
n-Butane –0.51 31.1 0.5840 – – 1.196
Isopentane 27.8 82.1 0.6247 2.5 3.1 1.111
n-Pentane 36.1 96.9 0.6311 3.0 3.7 1.099
2-Methylpentane 60.3 140.5 0.6578 4.0 4.7 1.050
n-Hexane 68.7 155.7 0.6638 3.0 3.5 1.040
2,4-Dimethylpentane 80.5 176.9 0.6764 5.5 6.3 1.017
n-Heptane 98.4 209.2 0.6882 7.5 8.4 1.000
Toluene 110.6 231.1 0.8743 15.5 13.7 0.724
n-Octane 125.7 258.2 0.7070 7.0 7.6 0.971
p-Xylene 138.4 281.1 0.8666 16.0 14.2 0.736
n-Propylbenzene 159.2 318.6 0.8683 6.5 5.8 0.739
n-Decane 174.1 345.5 0.7342 4.5 4.7 0.932
n-Butylbenzene 183.3 361.9 0.8660 6.0 5.3 0.745
n-Dodecane 216.3 421.4 0.7527 3.5 3.6 0.907
n-Tridecane 235.5 455.8 0.7617 4.5 4.6 0.895
n-Tetradecane 253.6 488.4 0.7633 3.0 3.0 0.893
n-Pentadecane 270.7 519.2 0.7722 5.0 5.0 0.882
n-Hexadecane 286.9 548.3 0.7772 3.0 3.0 0.876
A
Normal boiling points and relative densities (15.6/15.6 °C) obtained from Physical Constants of Hydrocarbon and Non-Hydrocarbon Compounds,ASTM Data Series DS
4B, 1988. The Fahrenheit values have been rounded to the nearest 0.1 °F. The Centigrade column has been converted from the °F values prior to rounding as listed in
ASTM Data Series DS 4B.
B
Volume percent is calculated as mass percent divided by specific gravity, then normalized to 100 volume percent.
C
FID volume response factors, as specified for use with this test method, are calculated from theoretical mass response factors and are relative to n-heptane (RVRF =
1).
D
Necessary if sample is expected to contain components boiling lower than isopentane. These gases are added non-quantitatively to the liquid calibration mixture.
TABLE 4 Typical Composition of Relative Response Mixtures Containing Oxygenates
A
Component Mass % Vol % Component Mass % Vol %*
Ethanol 9.35 9.09
Isopentane 2.27 2.80 Isopentane 2.37 2.93
n-Pentane 2.72 3.33 n-Pentane 2.84 3.47
2-Methylpentane 3.63 4.25 MTBE 9.17 9.50
n-Hexane 2.72 3.16 n-Hexane 2.84 3.30
2,4-Dimethylpentane 4.99 5.68 2,4-Dimethylpentane 5.20 5.94
n-Heptane 6.80 7.63 n-Heptane 7.10 7.97
Toluene 14.05 12.44 Toluene 14.67 13.00
n-Octane 6.35 6.93 n-Octane 6.62 7.24
p-Xylene 14.50 12.93 p-Xylene 15.14 13.51
n-Propylbenzene 5.89 5.25 n-Propylbenzene 6.15 5.48
n-Decane 4.08 4.29 n-Decane 4.26 4.48
Butylbenzene 5.44 4.86 Butylbenzene 5.68 5.07
n-Dodecane 3.17 3.25 n-Dodecane 3.31 3.40
n-Tridecane 4.08 4.14 n-Tridecane 4.26 4.33
n-Tetradecane 2.72 2.74 n-Tetradecane 2.84 2.86
n-Pentadecane 4.53 4.53 n-Pentadecane 4.73 4.73
n-Hexadecane 2.72 2.70 n-Hexadecane 2.84 2.82
A
Volume percent is calculated from the weight percent using specific gravity.
8.3 Carrier Gas—Helium, 99.999 mol% pure. (Warning— 9. Sampling
Compressed gas under high pressure.)
9.1 Sampling from Bulk Storage—Hydrocarbonliquidswith
8.4 Detector Gasses:
vapor pressures of 16 psia or less may be sampled either into
8.4.1 Fuel—Hydrogen, 99.999 mol% pure. (Warning—
a floating piston cylinder or into an open container.
Extremely flammable gas under pressure.)
9.1.1 Piston Cylinder Sampling—Refer to Practice D3700
8.4.2 Oxidant—Air, 99.999% free of hydrocarbons and
for instructions on transferring a representative sample of a
water. (Warning—Compressed gas under high pressure. Sup-
hydrocarbon fluid from a source into a floating piston cylinder.
ports combustion.)
9.1.2 Open Container Sampling—Refer to Practice D4057
8.5 Reference Gasoline—A gasoline sample that has been for instructions on manual sampling from bulk storage into
analyzed by laboratories participating in a test method coop- open containers. Seal containers immediately after sampling
erativestudy.(Warning—Extremelyflammableliquid.Vapors and preserve the samples by storing at 0°C to 4°C and
are harmful if inhaled.) maintaining that temperature until prior to analysis.
D7096 − 19
TABLE 5 Typical Relative Response Factors by Weight and
9.3 Calibration Mixture—Thecalibrationmixtureshouldbe
Volume, Molecular Weights (MW), and Densities for
storedintherefrigerator(0°Cto4°C)untilreadyforuse.The
A
Oxygenated Compounds
calibration mixture shall be warmed to room temperature
Relative
A before sub-sampling (or analysis) to ensure that all
Density
15.6/ B C
components, particularly the C to C paraffins, are com-
Compound MW RWRF RVRF
12 16
15.6 °C
pletely dissolved.
(60/
60 °F)
Methanol 32.0 0.7963 3.008 2.600
10. Preparation of Apparatus
Ethanol 46.1 0.7939 2.188 1.90
tert-Butanol 74.1 0.7922 1.298 1.128
10.1 Chromatographic Operating Conditions—Place in ser-
Methyl-tert-butyl ether (MTBE) 88.2 0.7460 1.528 1.410
vice in accordance with the manufacturer’s instructions. Typi-
Ethyl-tert-butyl ether (ETBE) 102.2 0.7452 1.385 1.279
cal operating conditions are shown in Tables 1 and 2. Other
tert-Amyl methyl ether (TAME) 102.2 0.7758 1.339 1.188
A conditionsmaybeusedprovidedtheymeetthecriteriaoutlined
Relative densities from Test Method D4814.
B
Weight response factors, relative to n-heptane and to be determined
in Section 11. Ensure that all components in the calibration
experimentally.
mixture elute completely before the maximum oven tempera-
C
Volume response factors, relative to n-heptane and to be determined experi-
ture is reached.
mentally. RVRFs from the precision study ranged from 1.86-1.92 for ethanol.
10.2 Column Preparation—Follow Practice E1510 for rec-
ommended installation and conditioning procedures.
9.2 Aliquoting Samples for Test:
11. System Performance
9.2.1 Sampling from an Open Container—Cooled samples
11.1 Conformance with the performance criteria shall be
are transferred to a pre-cooled septum vial and sealed imme-
establisheduponinitialset-upofthistestmethodandwhenever
diately. Obtain the test specimen for analysis directly from the
any changes are made to the apparatus or the operating
sealed septum vial for automatic injection.
conditions.To check system performance, analyze in duplicate
9.2.2 Sampling from a Floating Piston Cylinder—Samples
the calibration mixture (see 8.1 or 8.2), following the proce-
containedinfloatingpistoncylindersaretransferreddirectlyto
dure described in Section 13. Using these results, confirm that
a liquid sampling valve in the gas chromatograph by means of
the following criteria have been met.
the ballast pressure in the cylinders. Before injection, verify
that ballast pressure is sufficiently high to completely liquefy 11.2 Resolution—The system shall be able to identify the
the sample. beginning and end of the elution of n-pentane and lighter
FIG. 1 Parameters for Resolution Calculation
D7096 − 19
saturated hydrocarbons from the column.The resolution (R)of 11.5 Minimum Propane Retention—Selection of column
dodecane and tridecane shall be between 6 and 10 when length and instrument operating conditions shall be such as to
calculated according to Eq 1 (also see Fig. 1).
provide a minimum retention time for propane of at least 10 s
(0.167 min).
2d
R 5 (1)
1.699 W 1W
~ !
1 2
11.6 Response Factor Validation—Refer to Practice D4626
for calculation of gas chromatographic response factors. To
where:
validate the experimental response factors, it is necessary to
d = time between the peak maxima of dodecane and
know the concentrations of the response factor standard
tridecane, s,
components in both volume and mole percents. If conversion
W = peak width at half height of dodecane, s, and
W = peak width at half height of tridecane, s.
from one basis to another is required, a review of Practice
D2421 is recommended. Appendix X4 provides sample calcu-
11.3 Peak Skewing—Peak skewing can result in retention
lations for response factor validation.
time variance. Check skewness by calculating the ratio of the
segments A/B as shown in Eq 2, on peaks in the calibration 11.6.1 Volume response factors for each hydrocarbon com-
mixture. The ratio should be between 0.8 to 1.3. A graphical ponent in the calibration mixture (not including the gaseous
example of skew is given in Fig. 2.
components) are calculated according to Eq 3. The values
obtained shall agree within 610% of the theoretical volume
A
S 5 (2)
response factors listed in Table 3.
B
RVRF 5 ~V 3A !/~V 3A ! (3)
i i C7 C7 i
where:
A = segment of the peak width (at 5% of peak height)
where:
before the peak apex, and
RVRF = volume response factor of component i, relative to
i
B = segment of the peak width (at 5% of peak height) after
the volume response factor of n-heptane,
the peak apex.
A = area of n-heptane peak,
C7
V = volume percent n-heptane,
11.4 Retention Time Repeatability—For consecutive analy- C7
A = area of component i, and
i
ses of the retention time mixture, the maximum difference in
V = volume percent component i.
i
retention time for any component shall be 3 s (0.05 min), or
less.
FIG. 2 Peak Skewness
D7096 − 19
This same equation is used for the determination of the
response factors of the oxygenate components that may be
present in the gasoline.
11.6.2 The relative volume response factors of the gases are
obtained by first determining the relative molar response
factors of the C –C n-paraffins as calculated by Eq 4.
5 16
RMR 5 ~A 3M !/~A 3M ! (4)
i i C7 C7 i
where:
RMR = molar response factor of component i, relative to
i
molar response factor of n-heptane,
A = area of n-heptane peak,
C7
M = molar percent n-heptane,
C7
A = area of component i, and
i
M = molar percent component i.
i
11.6.3 The relative molar response factor (RMR) is a linear
FIG. 3 Boiling Point Calibration Curve
function of the molecular weight for the n-paraffins. Thus, the
RMR is plotted versus the molecular weight. The data for the
i
linear plot is subjected to a least squares fit. The plot should
12.1.2 Relative Volume Response Factors Calibration—
have a minimum least square fit (r ) of 0.99. By extrapolation,
Tabulate, for all components in the calibration mix, the
the RMR for propane and n-butane are calculated from the
retention time, area, and volume percent for each component.
i
resulting equation. Since the molecular weight of isobutane is
Utilize Eq 3 to calculate the relative volume response factor of
the same as that of n-butane, both compounds have the same
each hydrocarbon heavier than n-butane. Calculate the molar
RMR; however, since their densities are not the same, their
percent composition of each n-paraffin in the mix. Plot the
relative volume response factors will be different. Because of
molar percent of each component versus the molecular weight,
the low boiling point of isopentane and the difficulty in
as described in 11.6. Fig. 4 shows a relative molar response
handling it on a balance, this compound is sometimes consid-
plot. Using Eq 4 and 5, calculate the relative volume response
ered a gaseous component.
factors for propane, isobutane, and n-butane. Tabulate the
11.6.4 Convert the relative molar response factors of the
relative volume response factors and compare them to the
gases to relative volume response factors utilizing the follow-
theoretical volume response factors listed in Table 3.If
ing equation (Eq 5):
agreement between experimental and theoretical response
factors is within 10%, the theoretical values should be used to
RVR 5 MW 3RMR 3D / MW 3RMR 3D (5)
~ ! ~ !
i i C7 C7 C7 i i
determine the distillation results.
where:
NOTE 1—If the concentrations of propane, n-butane, and isobutane in
RVR = relative volume response factor for the gas i,
i
thecalibrationmixtureareknown,differencesnotedbetweentheobserved
MW = the molecular weight of ith gas,
i
and calculated molar response factors (MRF) indicate loss of light
MW = the molecular weight n-heptane,
C7 components.Ifafreshcalibrationmixtureisused,thesedifferencescanbe
D = the density of the ith gas, and
indicative of sampling problems. Deviation of the molar response factors
i
of the heavier components from the linear relationship could indicate
D = the density of n-heptane.
C7
problems in volatilizing the sample. Possible reasons include injection
RMR and RMR are the relative molar response factors for
i C7
port temperature being too low, insufficient carrier gas flow, or lack of
ith gas and for n-heptane, respectively, as determined by Eq 4.
homogeneity during sampling. Fig. 4 illustrates these effects.
12. Calibration and Standardization
12.1 Non-oxygenated Gasoline—Prior to the analysis of
samples, the analyzer should be calibrated to establish the
boiling point versus retention time relationship. Calibration is
carried out by analyzing the retention time and qualitative
calibration mix(es) (see 8.1) using the procedure outlined in
Section 13. Results from the calibration analyses are used to
determine the following:
12.1.1 Boiling Point—Retention Time Correlation—
Tabulate the retention time of each peak maximum and
atmosphericboilingpointindegreesCelsius(orFahrenheit)of
each component in the calibrat
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7096 − 16 D7096 − 19
Standard Test Method for
Determination of the Boiling Range Distribution of Gasoline
by Wide-Bore Capillary Gas Chromatography
This standard is issued under the fixed designation D7096; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of the boiling range distribution of gasoline and liquid gasoline blending
components. It is applicable to petroleum products and fractions with a final boiling point of 280 °C (536 °F) or lower, as measured
by this test method.
1.2 This test method is designed to measure the entire boiling range of gasoline and gasoline components with either high or
low vapor pressure and is commonly referred to as Simulated Distillation (SimDis) by gas chromatographers.
1.3 This test method has been validated for gasoline containing ethanol. Gasolines containing other oxygenates are not
specifically excluded, but they were not used in the development of this test method.
1.4 This test method can estimate the concentration of n-pentane and lighter saturated hydrocarbons in gasoline.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5.1 Results in degrees Fahrenheit can be obtained by simply substituting Fahrenheit boiling points in the calculation of the
boiling point-retention time correlation.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D86 Test Method for Distillation of Petroleum Products and Liquid Fuels at Atmospheric Pressure
D2421 Practice for Interconversion of Analysis of C and Lighter Hydrocarbons to Gas-Volume, Liquid-Volume, or Mass Basis
D3700 Practice for Obtaining LPG Samples Using a Floating Piston Cylinder
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4307 Practice for Preparation of Liquid Blends for Use as Analytical Standards
D4626 Practice for Calculation of Gas Chromatographic Response Factors
D4814 Specification for Automotive Spark-Ignition Engine Fuel
D4815 Test Method for Determination of MTBE, ETBE, TAME, DIPE, tertiary-Amyl Alcohol and C to C Alcohols in
1 4
Gasoline by Gas Chromatography
D5191 Test Method for Vapor Pressure of Petroleum Products and Liquid Fuels (Mini Method)
D5599 Test Method for Determination of Oxygenates in Gasoline by Gas Chromatography and Oxygen Selective Flame
Ionization Detection
D6300 Practice for Determination of Precision and Bias Data for Use in Test Methods for Petroleum Products and Lubricants
E594 Practice for Testing Flame Ionization Detectors Used in Gas or Supercritical Fluid Chromatography
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.04.0H on Chromatographic Distribution Methods.
Current edition approved Jan. 1, 2016May 1, 2019. Published February 2016June 2019. Originally approved in 2005. Last previous edition approved in 20102016 as
D7096 – 10.D7096 – 16. DOI: 10.1520/D7096-16.10.1520/D7096-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7096 − 19
E1510 Practice for Installing Fused Silica Open Tubular Capillary Columns in Gas Chromatographs
3. Terminology
3.1 Definitions:
3.1.1 area slice, n—area under a chromatogram within a specified retention time interval.
3.1.2 final boiling point (FBP), n—the point at which a cumulative volume count equal to 99.5 % of the total volume counts
under the chromatogram is obtained.
3.1.3 initial boiling point (IBP), n—the point at which a cumulative volume count equal to 0.5 % of the total volume counts
under the chromatogram is obtained.
3.1.4 relative volume response factor (RVRF), n—the volume response factor (see 3.1.8) of a component i relative to the volume
response factor of n-heptane.
3.1.5 slice time, n—the retention time at the end of a given area slice.
3.1.6 slice width, n—the fixed duration (1 s, or less) of the retention time intervals into which the chromatogram is divided. It
is determined from the reciprocal of the frequency used in the acquisition of data.
3.1.7 volume count, n—the product of a slice area (or an area under a peak) and a volume response factor.
3.1.8 volume response factor, n—a constant of proportionality that relates the area under a chromatogram to liquid volume.
4. Summary of Test Method
4.1 The sample is vaporized and transported by carrier gas into a non-polar, wide-bore capillary gas chromatographic column.
The column temperature is raised at a reproducible, linear rate so as to elute the hydrocarbon components in boiling point order
for measurement by a flame ionization detector. Conditions are selected such that n-pentane and lighter saturated hydrocarbons in
the calibration mixture are resolved discretely. Linear correlation between hydrocarbon boiling point and retention time is
established using a known mixture of hydrocarbons covering the boiling range expected in the sample. Area slices are converted
to volume using theoretical hydrocarbon volume response factors. Oxygenated samples require experimental determination of
oxygenate response factors.
5. Significance and Use
5.1 The determination of the boiling range distribution of gasoline by gas chromatographic simulated distillation provides an
insight into the composition of the components from which the gasoline has been blended. Knowledge of the boiling range
distribution of gasoline blending components is useful for the control of refinery processes and for the blending of finished
gasoline.
5.2 The determination of the boiling range distribution of light hydrocarbon mixtures by gas chromatographic simulated
distillation has better precision than the conventional distillation by Test Method D86. Additionally, this test method provides more
accurate and detailed information about the composition of the light ends. The distillation data produced by this test method are
similar to that which would be obtained from a cryogenic, true boiling point (15 theoretical plates) distillation.
6. Interferences
6.1 Ethanol or other oxygenates may coelute with hydrocarbons present in the sample. Since the response of oxygenates is
substantially different from the response of hydrocarbons, response factors are used to correct the area slice for the elution interval
of oxygenates.
6.2 Concentrations of n-pentane and lighter saturated compounds may be estimated from the analysis. However, early eluting
olefins present in the gasoline samples may coelute with these compounds.
6.3 For samples containing ethanol, this test method will determine the hydrocarbon distribution. It will not simulate the
azeotrope observed during physical distillation.
7. Apparatus
7.1 Gas Chromatograph—Any gas chromatograph (GC) designed for use with wide-bore (0.53 mm inside diameter) capillary
columns, that meets the performance criteria specified in Section 11, and has the following features may be used. Typical operating
conditions are shown in Table 1.
7.1.1 Column Oven Temperature Programming—The gas chromatograph shall be capable of linear temperature-programmed
operation from −40 °C to 280 °C at rates up to 25 °C ⁄min.
7.1.2 Injection Port—The injection port shall be capable of operation at temperatures required to completely volatize and
transfer the sample to the GC column. Non-splitting or split/splitless vaporizing sample ports optimized for use with wide-bore
capillary columns are acceptable. If using a split inlet port, it should be designed to provide a linear sample split injection.
7.1.3 Flame Ionization Detector—The detector shall be optimized for the use of wide-bore capillary gas chromatographic
columns and shall conform to the specifications as described in Practice E594.
D7096 − 19
TABLE 1 Typical Operating Conditions for Wide Bore
Column Inlets
Column length (m) 30 15
Column I.D. (mm) 0.53 0.53
Stationary phase 100 % poly- 100 % poly-
dimethylsiloxane dimethylsiloxane
Film thickness (μm) 5 5
Carrier gas helium helium
Carrier flow (mL/min) 20 15
Auxiliary flow (mL/min) 10 10
Column initial temperature 40 40
(°C)
Initial time (min) 1 1
Program rate (°C/min) 25 20
Final temperature (°C) 265 230
Final hold (min) 4.00 2.50
Injection inlet purged-packed purged-packed
Sample introduction auto syringe auto syringe
injection injection
Injector temperature (°C) 250 250
Detector temperature (°C) 280 300
Hydrogen flow (mL/min) 45 30
Air flow (mL/min) 450 300
Sample size (μL) 0.1 – 0.2 0.2
Area slice width (s) 0.5 – 0.2 0.5 – 0.2
Data rate (Hz) 2 – 5 2 – 5
TABLE 2 Typical Operating Conditions for Capillary Column Inlet
Column length (m) 30
Column I.D. (mm) 0.53
Stationary phase 100 %
polydimethylsiloxane
Film thickness 5 μm
Carrier gas helium (ramped flow)
Carrier flow (mL/min) 5 mL ⁄min (0.5 min) to
20 mL ⁄min @
60 mL ⁄min
Column initial temperature (°C) 40
Initial time (min) 1
Program rate (°C/min) 25
Final temperature (°C) 245
Final hold (min) 4
Injection port split
Sample introduction automatic syringe
injection
Injector temperature (°C) 250
Detector temperature (°C) 250
Hydrogen flow (mL/min) 30
Air flow (mL/min) 300
Sample size (μL) 1 uL
Split ratio 1:50
Data rate 5 Hz
7.1.4 Carrier Gas Controls—The associated carrier gas controls shall be of sufficient precision to produce reproducible column
flows in order to maintain analytical integrity.
7.1.5 Baseline Correction—The gas chromatograph (or another component of the gas chromatographic system) shall be capable
of subtracting the area slice of a blank run from the corresponding area slice of a sample run. This can be done internally on some
gas chromatographs (baseline compensation) or externally by subtracting a stored, digitized signal from a blank run.
7.2 Sample Introduction—Sample introduction may be by means of a constant volume liquid sample valve or by injection with
a micro syringe through a septum. An automatic sample introduction device is essential to the reproducibility of the analysis.
Manual injections are not recommended. Poor injection technique can result in poor resolution. If column overload occurs, peak
skewing may result, leading to variation in retention times.
7.2.1 Samples with a vapor pressure (VP) of less than 16 psia as measured by Test Method D5191, or equivalent, may be
introduced into the gas chromatograph by syringe injection into a heated, vaporizing inlet. Samples with vapor pressures between
12 psia and 16 psia should be kept chilled (refrigerated or in a cooled sample tray) and may require injection with a cooled syringe.
Samples with a vapor pressure above 16 psia should be introduced by way of a constant volume liquid sampling valve. Refer to
9.1 for sampling practices.
D7096 − 19
7.3 Column—Any wide bore (0.53 mm inside diameter) open tubular (capillary) column, coated with a non-polar (100 %
polydimethylsiloxane) phase that meets the performance criteria of Section 11.311 may be used. Columns of 15 metre15 m to
30 metre30 m lengths with a stationary phase film thickness of 5.0 μm have been successfully used. With either of these columns,
initial cryogenic temperatures are not necessary.
7.4 Data Acquisition System—A computer provided with a monitor, printer, and data acquisition software is necessary to carry
out this analysis. The computer should have sufficient hardware capacity and random access memory in order to run the data
acquisition program while acquiring data at a frequency of 2 Hz to 5 Hz. The software should also be able to store the data for
future recall, inspection, and analysis. The data acquisition software should be capable of presenting a real time plot. It may also
be capable of controlling the operating variables of the gas chromatograph. Specialized software is necessary to obtain the boiling
point distribution.
7.5 Bulk Sample Containers, floating piston cylinders (see 9.1.1); epoxy phenolic-lined metal cans; glass bottles with
polytetrafluoroethylene-lined screw caps.
8. Reagents and Materials
8.1 Calibration Mixture—A synthetic mixture of pure liquid hydrocarbons with boiling points that encompass the boiling range
of the sample shall be used for retention time determination and response factor validation. Response factors for propane,
isobutane, and n-butane are extrapolated from the relative molar response of the n-paraffins. An example of a relative response
factor mixture with suggested nominal composition is given in Table 3. This mixture shall be accurately prepared on a mass basis
using Practice D4307 or equivalent.
8.1.1 A single calibration standard may be used for retention time-boiling point determination and response factor validation
provided isopentane and heavier components are known quantitatively. Gaseous components propane, isobutane, and n-butane are
added in small quantities (< 0.2 volume % each). These small quantities do not significantly change the concentrations of the
remaining hydrocarbons. This standard would also be used for measuring performance criteria in Section 11. It may be practical
to generate this standard by bubbling a small amount of gaseous propane, isobutane, and n-butane (Warning—
WarningExtremely—Extremely flammable gases.) into a quantitative mixture of isopentane and heavier components.
8.1.2 A combination of two calibration standards may also be used. A quantitative standard, containing known concentrations
of isopentane and heavier compounds, is used to determine response factors. A qualitative standard, containing a wide boiling range
of compounds including propane, n-butane, and isobutane is used for measuring the retention time-boiling point relationship and
establishing the performance criteria outlined in Section 11.
8.2 Calibration Mixture with Oxygenates—When samples to be measured contain oxygenates, the calibration mixture (see 8.1)
shall also contain the oxygenates. Therefore, the identity of the oxygenate(s) shall be known prior to analysis of the sample.
TABLE 3 Typical Calibration Mixture Composition and Properties of Hydrocarbons
A
A
Relative Density
BP B C
Nominal Approx. FID
Compound 15.6/15.6 °C
Vol% RVRF
Mass%
°C °F
(60/60°F)
D
Propane –42.1 –43.8 0.5070 – – 1.394
D
Isobutane –11.8 10.8 0.5629 – – 1.241
D
n-Butane –0.51 31.1 0.5840 – – 1.196
Isopentane 27.8 82.1 0.6247 2.5 3.1 1.111
n-Pentane 36.1 96.9 0.6311 3.0 3.7 1.099
2-Methylpentane 60.3 140.5 0.6578 4.0 4.7 1.050
n-Hexane 68.7 155.7 0.6638 3.0 3.5 1.040
2,4-Dimethylpentane 80.5 176.9 0.6764 5.5 6.3 1.017
n-Heptane 98.4 209.2 0.6882 7.5 8.4 1.000
Toluene 110.6 231.1 0.8743 15.5 13.7 0.724
n-Octane 125.7 258.2 0.7070 7.0 7.6 0.971
p-Xylene 138.4 281.1 0.8666 16.0 14.2 0.736
n-Propylbenzene 159.2 318.6 0.8683 6.5 5.8 0.739
n-Decane 174.1 345.5 0.7342 4.5 4.7 0.932
n-Butylbenzene 183.3 361.9 0.8660 6.0 5.3 0.745
n-Dodecane 216.3 421.4 0.7527 3.5 3.6 0.907
n-Tridecane 235.5 455.8 0.7617 4.5 4.6 0.895
n-Tetradecane 253.6 488.4 0.7633 3.0 3.0 0.893
n-Pentadecane 270.7 519.2 0.7722 5.0 5.0 0.882
n-Hexadecane 286.9 548.3 0.7772 3.0 3.0 0.876
A
Normal boiling points and relative densities (15.6/15.6 °C) obtained from Physical Constants of Hydrocarbon and Non-Hydrocarbon Compounds, ASTM Data Series DS
4B, 1988. The Fahrenheit values have been rounded to the nearest 0.1 °F. The Centigrade column has been converted from the °F values prior to rounding as listed in
ASTM Data Series DS 4B.
B
Volume percent is calculated as mass percent divided by specific gravity, then normalized to 100 volume percent.
C
FID volume response factors, as specified for use with this test method, are calculated from theoretical mass response factors and are relative to n-heptane (RVRF =
1).
D
Necessary if sample is expected to contain components boiling lower than isopentane. These gases are added non-quantitatively to the liquid calibration mixture.
D7096 − 19
TABLE 4 Typical Composition of Relative Response Mixtures Containing Oxygenates
A
Component Mass % Vol % Component Mass % Vol %*
Ethanol 9.35 9.09
Isopentane 2.27 2.80 Isopentane 2.37 2.93
n-Pentane 2.72 3.33 n-Pentane 2.84 3.47
2-Methylpentane 3.63 4.25 MTBE 9.17 9.50
n-Hexane 2.72 3.16 n-Hexane 2.84 3.30
2,4-Dimethylpentane 4.99 5.68 2,4-Dimethylpentane 5.20 5.94
n-Heptane 6.80 7.63 n-Heptane 7.10 7.97
Toluene 14.05 12.44 Toluene 14.67 13.00
n-Octane 6.35 6.93 n-Octane 6.62 7.24
p-Xylene 14.50 12.93 p-Xylene 15.14 13.51
n-Propylbenzene 5.89 5.25 n-Propylbenzene 6.15 5.48
n-Decane 4.08 4.29 n-Decane 4.26 4.48
Butylbenzene 5.44 4.86 Butylbenzene 5.68 5.07
n-Dodecane 3.17 3.25 n-Dodecane 3.31 3.40
n-Tridecane 4.08 4.14 n-Tridecane 4.26 4.33
n-Tetradecane 2.72 2.74 n-Tetradecane 2.84 2.86
n-Pentadecane 4.53 4.53 n-Pentadecane 4.73 4.73
n-Hexadecane 2.72 2.70 n-Hexadecane 2.84 2.82
A
Volume percent is calculated from the weight percent using specific gravity.
Oxygenate content may be determined by Test Method D4815, Test Method D5599, or equivalent. Oxygenates, such as ethanol,
should be added to the calibration mixture at an approximate concentration as that in the samples. This mixture is used to define
the retention time boundary and relative volume response of the oxygenate to be applied to this region. For gasoline containing
other oxygenates, determine if the oxygenate coelutes with any of the hydrocarbons listed in Table 3. If a coelution occurs, the
coeluting hydrocarbon should not be included in the blend. Typical compositions of oxygenated blends are given in Table 4.
Typical relative volume response factors, molecular weights, and densities for various oxygenated compounds are provided in
Table 5.
8.3 Carrier Gas—Helium, 99.999 mol% pure. (Warning—WarningCompressed—Compressed gas under high pressure.)
8.4 Detector Gasses:
8.4.1 Fuel—Hydrogen, 99.999 mol% pure. (Warning—WarningExtremely—Extremely flammable gas under pressure.)
8.4.2 Oxidant—Air, 99.999 % free of hydrocarbons and water. (Warning—WarningCompressed—Compressed gas under high
pressure. Supports combustion.)
8.5 Reference Gasoline—A gasoline sample that has been analyzed by laboratories participating in a test method cooperative
study. (Warning—WarningExtremely—Extremely flammable liquid. Vapors are harmful if inhaled.)
9. Sampling
9.1 Sampling from Bulk Storage—Hydrocarbon liquids with vapor pressures of 16 psia or less may be sampled either into a
floating piston cylinder or into an open container.
9.1.1 Piston Cylinder Sampling—Refer to Practice D3700 for instructions on transferring a representative sample of a
hydrocarbon fluid from a source into a floating piston cylinder.
TABLE 5 Typical Relative Response Factors by Weight and
Volume, Molecular Weights (MW), and Densities for
A
Oxygenated Compounds
Relative
A
Density
15.6/ B C
Compound MW RWRF RVRF
15.6 °C
(60/
60 °F)
Methanol 32.0 0.7963 3.008 2.600
Ethanol 46.1 0.7939 2.188 1.90
tert-Butanol 74.1 0.7922 1.298 1.128
Methyl-tert-butyl ether (MTBE) 88.2 0.7460 1.528 1.410
Ethyl-tert-butyl ether (ETBE) 102.2 0.7452 1.385 1.279
tert-Amyl methyl ether (TAME) 102.2 0.7758 1.339 1.188
A
Relative densities from Test Method D4814.
B
Weight response factors, relative to n-heptane and to be determined
experimentally.
C
Volume response factors, relative to n-heptane and to be determined experi-
mentally. RVRFs from the precision study ranged from 1.86-1.92 for ethanol.
D7096 − 19
9.1.2 Open Container Sampling—Refer to Practice D4057 for instructions on manual sampling from bulk storage into open
containers. Seal containers immediately after sampling and preserve the samples by storing at 0 °C to 4 °C and maintaining that
temperature until prior to analysis.
9.2 Aliquoting Samples for Test:
9.2.1 Sampling from an Open Container—Cooled samples are transferred to a pre-cooled septum vial and sealed immediately.
Obtain the test specimen for analysis directly from the sealed septum vial for automatic injection.
9.2.2 Sampling from a Floating Piston Cylinder—Samples contained in floating piston cylinders are transferred directly to a
liquid sampling valve in the gas chromatograph by means of the ballast pressure in the cylinders. Before injection, verify that
ballast pressure is sufficiently high to completely liquefy the sample.
9.3 Calibration Mixture—The calibration mixture should be stored in the refrigerator (0 °C to 4 °C) until ready for use. The
calibration mixture shall be warmed to room temperature before sub-sampling (or analysis) to ensure that all components,
particularly the C to C paraffins, are completely dissolved.
12 16
10. Preparation of Apparatus
10.1 Chromatographic Operating Conditions—Place in service in accordance with the manufacturer’s instructions. Typical
operating conditions are shown in Tables 1 and 2. Other conditions may be used provided they meet the criteria outlined in Section
11. Ensure that all components in the calibration mixture elute completely before the maximum oven temperature is reached.
10.2 Column Preparation—Follow Practice E1510 for recommended installation and conditioning procedures.
11. System Performance
11.1 Conformance with the performance criteria shall be established upon initial set-up of this test method and whenever any
changes are made to the apparatus or the operating conditions. To check system performance, analyze in duplicate the calibration
mixture (see 8.1 or 8.2), following the procedure described in Section 13. Using these results, confirm that the following criteria
have been met.
11.2 Resolution—The system shall be able to identify the beginning and end of the elution of n-pentane and lighter saturated
hydrocarbons from the column. The resolution (R) of dodecane and tridecane shall be between 6 and 10 when calculated according
to Eq 1 (also see Fig. 1).
FIG. 1 Parameters for Resolution Calculation
D7096 − 19
2d
R 5 (1)
1.699 ~W 1W !
1 2
where:
d = time between the peak maxima of dodecane and tridecane, s,
W = peak width at half height of dodecane, s, and
W = peak width at half height of tridecane, s.
11.3 Column Selectivity—Using a linear least squares fit of the data for only the n-paraffins (C through C ), establish the
5 16
boiling point versus retention time relationship (see 12.1.1). From this relationship, calculate the apparent boiling point of each
of the aromatics in the calibration mixture from their observed retention times. The apparent boiling point of each aromatic shall
not differ from its actual boiling point by more than 2 °C (3 °F).
11.3 Peak Skewing—Peak skewing can result in retention time variance. Check skewness by calculating the ratio of the
segments A/B as shown in Eq 2, on peaks in the calibration mixture. The ratio should be between 0.8 to 1.3. A graphical example
of skew is given in Fig. 2.
A
S 5 (2)
B
where:
A = segment of the peak width (at 5 % of peak height) before the peak apex, and
B = segment of the peak width (at 5 % of peak height) after the peak apex.
11.4 Retention Time Repeatability—For consecutive analyses of the retention time mixture, the maximum difference in retention
time for any component shall be 3 s (0.05 min), or less.
11.5 Minimum Propane Retention—Selection of column length and instrument operating conditions shall be such as to provide
a minimum retention time for propane of at least 10 s (0.167 min).
11.6 Response Factor Validation—Refer to Practice D4626 for calculation of gas chromatographic response factors. To validate
the experimental response factors, it is necessary to know the concentrations of the response factor standard components in both
volume and mole percents. If conversion from one basis to another is required, a review of Practice D2421 is recommended.
Appendix X4 provides sample calculations for response factor validation.
11.6.1 Volume response factors for each hydrocarbon component in the calibration mixture (not including the gaseous
components) are calculated according to Eq 3. The values obtained shall agree within 610 % of the theoretical volume response
factors listed in Table 3.
FIG. 2 Peak Skewness
D7096 − 19
RVRF 5 V 3A / V 3A (3)
~ ! ~ !
i i C7 C7 i
where:
RVRF = volume response factor of component i, relative to the volume response factor of n-heptane,
i
A = area of n-heptane peak,
C7
V = volume percent n-heptane,
C7
A = area of component i, and
i
V = volume percent component i.
i
This same equation is used for the determination of the response factors of the oxygenate components that may be present in
the gasoline.
11.6.2 The relative volume response factors of the gases are obtained by first determining the relative molar response factors
of the C – C n-paraffins as calculated by Eq 4.
5 16
RMR 5 A 3M / A 3M (4)
~ ! ~ !
i i C7 C7 i
where:
RMR = molar response factor of component i, relative to molar response factor of n-heptane,
i
A = area of n-heptane peak,
C7
M = molar percent n-heptane,
C7
A = area of component i, and
i
M = molar percent component i.
i
11.6.3 The relative molar response factor (RMR) is a linear function of the molecular weight for the n-paraffins. Thus, the RMR
i
is plotted versus the molecular weight. The data for the linear plot is subjected to a least squares fit. The plot should have a
minimum least square fit (r ) of 0.99. By extrapolation, the RMR for propane and n-butane are calculated from the resulting
i
equation. Since the molecular weight of isobutane is the same as that of n-butane, both compounds have the same RMR; however,
since their densities are not the same, their relative volume response factors will be different. Because of the low boiling point of
isopentane and the difficulty in handling it on a balance, this compound is sometimes considered a gaseous component.
11.6.4 Convert the relative molar response factors of the gases to relative volume response factors utilizing the following
equation (Eq 5):
RVR 5 MW 3RMR 3D / MW 3RMR 3D (5)
~ ! ~ !
i i C7 C7 C7 i i
where:
RVR = relative volume response factor for the gas i,
i
MW = the molecular weight of ith gas,
i
MW = the molecular weight n-heptane,
C7
D = the density of the ith gas, and
i
D = the density of n-heptane.
C7
RMR and RMR are the relative molar response factors for ith gas and for n-heptane, respectively, as determined by Eq 4.
i C7
12. Calibration and Standardization
12.1 Non-oxygenated Gasoline—Prior to the analysis of samples, the analyzer should be calibrated to establish the boiling point
versus retention time relationship. Calibration is carried out by analyzing the retention time and qualitative calibration mix(es) (see
8.1) using the procedure outlined in Section 13. Results from the calibration analyses are used to determine the following:
12.1.1 Boiling Point—Retention Time Correlation—Tabulate the retention time of each peak maximum and atmospheric boiling
point in degrees Celsius (or Fahrenheit) of each component in the calibration mixture. Plot the retention times of the hydrocarbon
components versus the corresponding atmospheric boiling point temperatures, as shown in Fig. 3. Visually verify that the
calibration curve is essentially a straight line with slight curvature for the lowest boiling components.
12.1.2 Relative Volume Response Factors Calibration—Tabulate, for all components in the calibration mix, the retention time,
area, and volume percent for each component. Utilize Eq 3 to calculate the relative volume response factor of each hydrocarbon
heavier than n-butane. Calculate the molar percent composition of each n-paraffin in the mix. Plot the molar percent of each
component versus the molecular weight, as described in 11.711.6. Fig. 4 shows a relative molar response plot. Using Eq 4 and 5,
calculate the relative volume response factors for propane, isobutane, and n-butane. Tabulate the relative volume response factors
and compare them to the theoretical volume response factors listed in Table 3. If agreement between experimental and theoretical
response factors is within 10 %, the theoretical values should be used to determine the distillation results.
NOTE 1—If the concentrations of propane, n-butane, and isobutane in the calibration mixture are known, differences noted between the observed and
calculated molar response factors (MRF) indicate loss of
...



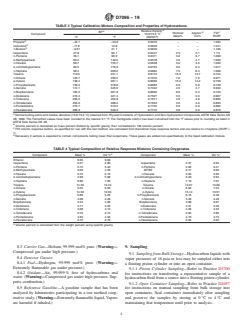




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...