ASTM F1798-21
(Test Method)Standard Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants
Standard Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants
SIGNIFICANCE AND USE
5.1 Spinal implants are generally composed of several components that, when connected together, form a spinal implant construct. Spinal implant constructs are designed to provide some stability to the spine while arthrodesis takes place. This test method outlines standardized evaluations of different interconnection mechanisms to facilitate comparison between different designs. Comparisons must be made cautiously and with careful analysis, taking into account the effects that design differences can have on the loading configurations.
5.2 This test method is used to quantify the static and fatigue properties of different implant interconnection designs. The mechanical tests are conducted in vitro using simplified, unidirectional loads and moments. Fatigue testing in a simulated body fluid or saline may have a fretting, corrosive, or lubricating effect on the interconnection and thereby affect the relative performance of tested devices. Hence, the test environment, whether a simulated body fluid, saline (9 g NaCl per 1000 mL H2O), with a saline drip, or dry, is an important characteristic of the test and must be reported accurately.
5.3 The loading of spinal implant constructs in vivo will, in general, differ from the loading configurations used in this test method. The results obtained here cannot be used directly to predict in vivo performance. However, the results can be used to compare different component designs in terms of relative mechanical parameters.
SCOPE
1.1 This test method covers the measurement of uniaxial static and fatigue strength, and resistance to loosening of the component interconnection mechanisms of spinal arthrodesis implants.
1.2 The purpose of this test method is to provide a means of mechanically characterizing different designs of spinal implant interconnections. Ultimately, the various components and interconnections should be combined for static and fatigue testing of the spinal implant construct. It is not the intention of this test method to address the analysis of spinal implant constructs or subconstructs or to define levels of performance of spinal implants, as insufficient knowledge is available to predict the consequences of the use of particular spinal implant designs.
1.3 This standard defines test methods to measure the strength of spinal implant component interconnections and how to report test results.
1.4 The values stated in SI units are to be regarded as standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2021
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 01-May-2015
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jan-2014
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Dec-2012
- Effective Date
- 15-May-2012
- Effective Date
- 01-Jul-2011
- Effective Date
- 01-Jun-2011
- Effective Date
- 01-Jul-2010
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 15-Sep-2009
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Dec-2008
- Effective Date
- 01-Jan-2007
Overview
ASTM F1798-21, titled Standard Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants, provides a comprehensive framework for the mechanical assessment of interconnection mechanisms within spinal implant systems. Developed by ASTM International, this standard establishes uniform testing procedures for measuring the uniaxial static and fatigue strength, as well as resistance to loosening, of component interconnections. Its primary aim is to facilitate accurate comparison of different spinal implant designs, supporting innovation, safety, and product reliability throughout the orthopedic implant industry.
Key Topics
- Static and Fatigue Testing: The standard outlines test methods to evaluate both static (single-load) and fatigue (repeated cyclic load) properties. These tests simulate simplified unidirectional loading conditions that interconnection mechanisms, such as those found in pedicle screws, hooks, or rods, might experience during clinical use.
- Reporting Requirements: ASTM F1798-21 mandates detailed reporting of test configurations, environmental conditions (air, saline solution, or simulated body fluid), loading rates, and outcomes (including failure modes and any visible corrosion or wear).
- Testing Apparatus and Procedures: Specific apparatus configurations are described for applying loads in axial, anterior-posterior, transverse, and torsional directions. Careful alignment and rigid fixation are emphasized to minimize experimental variability.
- Sample Preparation: All test specimens must be previously unused, with appropriate labeling, and maintained according to recognized laboratory practices to ensure data integrity.
- Key Performance Metrics:
- Gripping capacity
- Yield load/moment and ultimate load/moment
- Loosening torque
- Maximum run-out load/moment after 2.5 million load cycles
Applications
ASTM F1798-21 is widely used in the design, testing, and regulatory submission process for spinal arthrodesis implants. The standard is applicable in several contexts:
- Spinal Implant Development: Assists manufacturers in characterizing and benchmarking the mechanical reliability of new interconnection designs before clinical use.
- Quality Control: Ensures consistency across batches of spinal implant components by defining reproducible testing methods.
- Regulatory Compliance: Provides test data for pre-market submissions to regulatory agencies, supporting claims of device safety and performance.
- Comparative Analysis: Enables researchers and companies to objectively compare the mechanical properties of competing spinal instrumentation systems, thus influencing product selection and clinical decision-making.
- Risk Management: Identifies potential weaknesses in implant interconnection stability, helping to mitigate risks of post-surgical failure.
Related Standards
Several related ASTM standards support or complement ASTM F1798-21 in the field of medical device testing:
- ASTM E4: Practices for Force Calibration and Verification of Testing Machines
- ASTM F383: (Withdrawn) Practice for Static Bend and Torsion Testing of Intramedullary Rods
- ASTM F1717: Test Methods for Spinal Implant Constructs in a Vertebrectomy Model
- ASTM F1582: Terminology Relating to Spinal Implants
These standards collectively provide a robust methodology for assessing the structural integrity and durability of spinal implants used in arthrodesis procedures.
By establishing clear, repeatable, and scientifically valid test methods, ASTM F1798-21 plays a critical role in advancing the safety and effectiveness of spinal arthrodesis implants. It ensures that interconnection mechanisms are thoroughly evaluated for mechanical performance, supporting both manufacturers and healthcare providers in delivering reliable solutions for spinal fusion surgeries.
Buy Documents
ASTM F1798-21 - Standard Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants
REDLINE ASTM F1798-21 - Standard Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F1798-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Evaluating the Static and Fatigue Properties of Interconnection Mechanisms and Subassemblies Used in Spinal Arthrodesis Implants". This standard covers: SIGNIFICANCE AND USE 5.1 Spinal implants are generally composed of several components that, when connected together, form a spinal implant construct. Spinal implant constructs are designed to provide some stability to the spine while arthrodesis takes place. This test method outlines standardized evaluations of different interconnection mechanisms to facilitate comparison between different designs. Comparisons must be made cautiously and with careful analysis, taking into account the effects that design differences can have on the loading configurations. 5.2 This test method is used to quantify the static and fatigue properties of different implant interconnection designs. The mechanical tests are conducted in vitro using simplified, unidirectional loads and moments. Fatigue testing in a simulated body fluid or saline may have a fretting, corrosive, or lubricating effect on the interconnection and thereby affect the relative performance of tested devices. Hence, the test environment, whether a simulated body fluid, saline (9 g NaCl per 1000 mL H2O), with a saline drip, or dry, is an important characteristic of the test and must be reported accurately. 5.3 The loading of spinal implant constructs in vivo will, in general, differ from the loading configurations used in this test method. The results obtained here cannot be used directly to predict in vivo performance. However, the results can be used to compare different component designs in terms of relative mechanical parameters. SCOPE 1.1 This test method covers the measurement of uniaxial static and fatigue strength, and resistance to loosening of the component interconnection mechanisms of spinal arthrodesis implants. 1.2 The purpose of this test method is to provide a means of mechanically characterizing different designs of spinal implant interconnections. Ultimately, the various components and interconnections should be combined for static and fatigue testing of the spinal implant construct. It is not the intention of this test method to address the analysis of spinal implant constructs or subconstructs or to define levels of performance of spinal implants, as insufficient knowledge is available to predict the consequences of the use of particular spinal implant designs. 1.3 This standard defines test methods to measure the strength of spinal implant component interconnections and how to report test results. 1.4 The values stated in SI units are to be regarded as standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Spinal implants are generally composed of several components that, when connected together, form a spinal implant construct. Spinal implant constructs are designed to provide some stability to the spine while arthrodesis takes place. This test method outlines standardized evaluations of different interconnection mechanisms to facilitate comparison between different designs. Comparisons must be made cautiously and with careful analysis, taking into account the effects that design differences can have on the loading configurations. 5.2 This test method is used to quantify the static and fatigue properties of different implant interconnection designs. The mechanical tests are conducted in vitro using simplified, unidirectional loads and moments. Fatigue testing in a simulated body fluid or saline may have a fretting, corrosive, or lubricating effect on the interconnection and thereby affect the relative performance of tested devices. Hence, the test environment, whether a simulated body fluid, saline (9 g NaCl per 1000 mL H2O), with a saline drip, or dry, is an important characteristic of the test and must be reported accurately. 5.3 The loading of spinal implant constructs in vivo will, in general, differ from the loading configurations used in this test method. The results obtained here cannot be used directly to predict in vivo performance. However, the results can be used to compare different component designs in terms of relative mechanical parameters. SCOPE 1.1 This test method covers the measurement of uniaxial static and fatigue strength, and resistance to loosening of the component interconnection mechanisms of spinal arthrodesis implants. 1.2 The purpose of this test method is to provide a means of mechanically characterizing different designs of spinal implant interconnections. Ultimately, the various components and interconnections should be combined for static and fatigue testing of the spinal implant construct. It is not the intention of this test method to address the analysis of spinal implant constructs or subconstructs or to define levels of performance of spinal implants, as insufficient knowledge is available to predict the consequences of the use of particular spinal implant designs. 1.3 This standard defines test methods to measure the strength of spinal implant component interconnections and how to report test results. 1.4 The values stated in SI units are to be regarded as standard, with the exception of angular measurements, which may be reported in terms of either degrees or radians. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F1798-21 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F1798-21 has the following relationships with other standards: It is inter standard links to ASTM F1717-15, ASTM E4-14, ASTM F1717-14, ASTM F1717-13, ASTM F1717-12a, ASTM F1717-12, ASTM F1717-11a, ASTM F1717-11, ASTM F1717-10, ASTM E4-10, ASTM E4-09a, ASTM F1717-09, ASTM E4-09, ASTM E4-08, ASTM E4-07. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F1798-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F1798 − 21
Standard Test Method for
Evaluating the Static and Fatigue Properties of
Interconnection Mechanisms and Subassemblies Used in
Spinal Arthrodesis Implants
This standard is issued under the fixed designation F1798; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method covers the measurement of uniaxial
E4Practices for Force Calibration and Verification of Test-
static and fatigue strength, and resistance to loosening of the
ing Machines
component interconnection mechanisms of spinal arthrodesis
F383Practice for Static Bend and Torsion Testing of In-
implants.
tramedullary Rods (Withdrawn 1996)
1.2 Thepurposeofthistestmethodistoprovideameansof
F1717Test Methods for Spinal Implant Constructs in a
mechanicallycharacterizingdifferentdesignsofspinalimplant
Vertebrectomy Model
interconnections. Ultimately, the various components and in-
terconnections should be combined for static and fatigue
3. Terminology
testing of the spinal implant construct. It is not the intention of
3.1 Definitions of Terms Specific to This Standard:
this test method to address the analysis of spinal implant
3.1.1 active length of longitudinal element—the span be-
constructs or subconstructs or to define levels of performance
tween rigid supports (for example, 50 mm is the active length
of spinal implants, as insufficient knowledge is available to
in Fig. 1, Fig. 2, Fig. 3(a), Fig. 3(b), and Fig. 4).
predicttheconsequencesoftheuseofparticularspinalimplant
designs. 3.1.2 global coordinate system—spinal column motion has
six degrees of freedom, having translational motion along, and
1.3 This standard defines test methods to measure the
rotational motion about three axes. The axes are labeled
strength of spinal implant component interconnections and
anterior-posterior orA-P (X), medial-lateral or transverse (Y),
how to report test results.
and caudal-cranial or axial (Z).This coordinate system is right
1.4 The values stated in SI units are to be regarded as
handed with +X in the anterior direction, +Y towards the left
standard, with the exception of angular measurements, which
side of the body, and +Z in the cranial direction. Positive
may be reported in terms of either degrees or radians.
rotations are defined by the right hand rule (see Fig. 5(a)).
1.5 This standard does not purport to address all of the
3.1.3 gripping capacity—the maximum applied load or
safety concerns, if any, associated with its use. It is the
moment across an interconnection mechanism within the first
responsibility of the user of this standard to establish appro-
1.5mmofpermanentdisplacementor5°ofpermanentrotation
priate safety, health, and environmental practices and deter- between the connected components.
mine the applicability of regulatory limitations prior to use.
3.1.4 local coordinate system—thespine’sglobalcoordinate
1.6 This international standard was developed in accor-
system shall be applied locally at the position of the intercon-
dance with internationally recognized principles on standard-
nection. The local direction, z, shall be centered through the
ization established in the Decision on Principles for the
longitudinal element of the x-y plane. The local direction, x,
Development of International Standards, Guides and Recom-
shall be defined as parallel to the axis of a screw or back of a
mendations issued by the World Trade Organization Technical
hook. The local transverse axis, y, shall be parallel to a
Barriers to Trade (TBT) Committee.
transverse element (see Fig. 5(b) and Fig. 5(c)).
1 2
ThistestmethodisunderthejurisdictionofASTMCommitteeF04onMedical For referenced ASTM standards, visit the ASTM website, www.astm.org, or
andSurgicalMaterialsandDevicesandisthedirectresponsibilityofSubcommittee contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
F04.25 on Spinal Devices. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Oct. 1, 2021. Published October 2021. Originally the ASTM website.
approved in 1997. Last previous edition approved in 2013 as F1798–13. DOI: The last approved version of this historical standard is referenced on
10.1520/F1798-21. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1798 − 21
3.1.8 spinal arthrodesis implant—an implant that provides
temporary correction and stability to vertebrae while bony
fusion occurs.
3.1.9 subassembly failure—permanent deformation result-
ing from fracture, plastic deformation, loosening, or slippage
that renders the subassembly ineffective or unable to ad-
equately resist load.
3.1.10 subassembly permanent deformation—the displace-
ment (mm) or angular displacement (degree of the subassem-
bly relative to the unloaded condition) remaining after the
applied load moment or torque has been removed.
3.1.11 tightening torque—thespecifiedtorquethatisapplied
to the various threaded fasteners of the spinal arthrodesis
implant during assembly.
3.1.12 ultimate load/moment of the subassembly—
maximumloadormomentappliedtoasubassembly(seePoint
Ein Fig. 6).
3.1.13 yield load/moment of the subassembly—the load or
FIG. 1 A-P Test Apparatus for Subassembly
moment required to produce a permanent deformation equal to
0.020 times the active length of the longitudinal element (see
Point D in Fig. 6).
4. Summary of Test Method
4.1 Vertebral attachment components (for example, hook,
screws, bands) and transverse elements must be attached to
longitudinalelements(forexample,rods,plates)toformspinal
implant subassemblies.
4.2 The interconnections are tested only in the relevant
directions of loading by applying loads at specific locations
relative to the local coordinate system.
4.3 The interconnections and subassemblies are tested stati-
callyinaload-to-failuremodeandalsocanbetestedcyclically
to estimate the maximum run-out value at 2.5 × 10 cycles.
5. Significance and Use
5.1 Spinal implants are generally composed of several
components that, when connected together, form a spinal
implant construct. Spinal implant constructs are designed to
provide some stability to the spine while arthrodesis takes
FIG. 2 Transverse Test Apparatus for Subassembly
place. This test method outlines standardized evaluations of
different interconnection mechanisms to facilitate comparison
between different designs. Comparisons must be made cau-
tiouslyandwithcarefulanalysis,takingintoaccounttheeffects
3.1.5 loosening torque—the torque required to disconnect
that design differences can have on the loading configurations.
thevariousthreadedfastenersofthespinalarthrodesisimplant.
5.2 This test method is used to quantify the static and
3.1.6 maximum run-out load/moment—the maximum load
fatigue properties of different implant interconnection designs.
or moment that can be applied to a subassembly where all the
The mechanical tests are conducted in vitro using simplified,
tested constructs have withstood 2.5 × 106 cycles without a
unidirectional loads and moments. Fatigue testing in a simu-
failure.
lated body fluid or saline may have a fretting, corrosive, or
3.1.7 relevant directions of loading—those directions of
lubricating effect on the interconnection and thereby affect the
loading in which a particular component interconnection is
relative performance of tested devices. Hence, the test
designed to provide resistance to loading. For example, a
environment, whether a simulated body fluid, saline (9g NaCl
particular spinal hook may be designed to withstand a positive
per 1000 mL H O), with a saline drip, or dry, is an important
axial load, A-P load, and flexion-extension moment, but not a
characteristic of the test and must be reported accurately.
negative axial load or axial torsion. Hence, positive axial load,
A-P load, and flexion-extension moment are the relevant 5.3 The loading of spinal implant constructs in vivo will, in
directions of loading. general, differ from the loading configurations used in this test
F1798 − 21
FIG. 3 Flexion-Extension Moment Test Apparatus for Subassembly
F1798 − 21
FIG. 4 Transverse Moment Test Apparatus for Subassembly
method. The results obtained here cannot be used directly to least5mmshallextendbeyondtheinterconnectionlinkageand
predict in vivo performance. However, the results can be used remain unfixed. Axial loads are applied to the interconnection
to compare different component designs in terms of relative mechanism along the axis of the longitudinal element via a
mechanical parameters. sleeve(collar)whichfreelysurroundsthelongitudinalelement.
Thesleeve(collar)shouldevenlydistributetheloadaroundthe
6. Apparatus
interconnection. An alternate method, depicted in Fig. 7(b),
applies the load to the longitudinal element and pushes it
6.1 Machines used for the test shall conform to the require-
through the interconnection clamp.
ments of Practices E4.
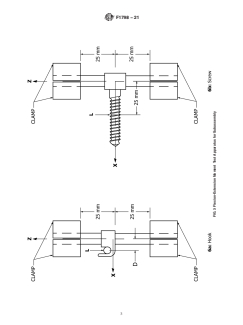
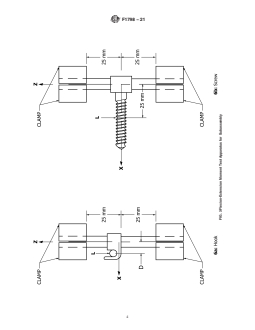
6.2 The apparatus for axial (z) gripping capacity measure- 6.3 TheapparatusforA-P(x)mechanicalpropertymeasure-
mentsofaninterconnectionmechanismisdepictedinFig.7(a). ments of a subassembly is depicted in Fig. 1. Both ends of the
One end of the longitudinal element shall be clamped rigidly, longitudinal element shall be clamped rigidly, with the inter-
leaving5mmexposedbetweentheinterconnectionmechanism connection centered on a 50-mm section of the longitudinal
and the test machine base.Asection of longitudinal element at element. The local origin of the interconnection mechanism
F1798 − 21
FIG. 5 Coordinate System
F1798 − 21
FIG. 6 Load/Displacement Curve
shall be centered between the mounts. Loads are applied to the longitudinal element. The local origin of the interconnection
interconnection (perpendicular to the longitudinal element) via mechanism shall be centered between the mounts. Loads are
a clamp on the hook, screw, or band. The load should be applied to the interconnection (perpendicular to the longitudi-
centered through the local x-coordinate axis. nal element) via a clamp on the transverse connector.The load
should be centered through the local y-coordinate axis.
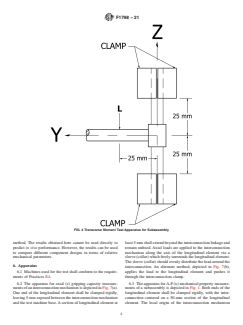
6.4 The apparatus for transverse (y) mechanical property
measurements of a subassembly is depicted in Fig. 2. Both 6.5 The apparatus for flexion-extension moment (My) me-
ends of the longitudinal element shall be clamped rigidly, with chanical property measurements of a subassembly is depicted
the interconnection centered on a 50-mm section of the in Fig. 3. Both ends of the longitudinal element shall be
F1798 − 21
FIG. 7 Axial Gripping Capacity Test Apparatus
clampedrigidly,withtheinterconnectioncenteredona50-mm 7. Sampling
section of the longitudinal element. The local origin of the
7.1 Thesamplestestedshallbepreviouslyunusedparts,and
interconnection mechanism shall be centered between the
shall not be re-tested.
mounts. Loads are applied to the interconnection (parallel to
7.2 The test constructs shall be labeled and maintained
the longitudinal element). For spinal hooks, the load shall be
according to good laboratory practice.
applied via a cylinder set in the hook notch (see Fig. 3(a)). For
other elements (screws) the load shall be applied 25 mm from
7.3 Static tests of mechanical properties should have a
the local z-axis (see Fig. 3(b)). minimum sample size of five.
6.6 The apparatus for transverse moment (Mx) mechanical
7.4 Fatiguetestsfordeterminingthemaximumrun-outload
property measurements of a subassembly is depicted in Fig. 4. or moment of a subassembly at 2.5 × 10 cycles shall utilize a
As in the previous test, 6.5, both ends of the longitudinal
rundown, half-interval approach with one specimen per run
element shall be clamped rigidly, with the interconnection down interval or half-interval and three consecutive specimens
centered on a 50-mm section of the longitudinal element. The
showing run-out to 2.5 × 10 cycles. Alternative methods for
localoriginoftheinterconnectionmechanismshallbecentered determining the starting point of the fatigue curve are the
between the mounts. Loads are applied to the interconnection
run-up method or choosing 75% of the ultimate static load or
(parallel to the longitudinal element), 25 mm from the z-axis. moment.
6.7 The apparatus for axial torque (Mz) gripping capacity
8. Procedure for Measuring Static Mechanical Properties
measurements of an interconnection mechanism is depicted in
Fig. 8(a) and is similar to that described in 6.2, with the 8.1 Measure the tightening torques for any set screws or
exception that the axial torque is applied via notches in the nuts which are incorporated into the interconnection linkage.
sleeve that surrounds the longitudinal element. An alternative
8.2 Apply all tightening, crimping, or locking mechanisms
method is to hold the interconnection rigidly and apply the
as specified by the manufacturer.
torsional f
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F1798 − 13 F1798 − 21
Standard Test Method for
Evaluating the Static and Fatigue Properties of
Interconnection Mechanisms and Subassemblies Used in
Spinal Arthrodesis Implants
This standard is issued under the fixed designation F1798; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the measurement of uniaxial static and fatigue strength, and resistance to loosening of the component
interconnection mechanisms of spinal arthrodesis implants.
1.2 The purpose of this test method is to provide a means of mechanically characterizing different designs of spinal implant
interconnections. Ultimately, the various components and interconnections should be combined for static and fatigue testing of the
spinal implant construct. It is not the intention of this test method to address the analysis of spinal implant constructs or
subconstructs or to define levels of performance of spinal implants, as insufficient knowledge is available to predict the
consequences of the use of particular spinal implant designs.
1.3 This test method sets out definitions for use in measuring standard defines test methods to measure the strength of component
interconnections of spinal implants, possible test methods themselves, and the reporting ofspinal implant component intercon-
nections and how to report test results.
1.4 The values stated in SI units are to be regarded as standard, with the exception of angular measurements, which may be
reported in terms of either degrees or radians.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E4 Practices for Force Calibration and Verification of Testing Machines
F383 Practice for Static Bend and Torsion Testing of Intramedullary Rods (Withdrawn 1996)
This test method is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices. and is the direct responsibility of Subcommittee
F04.25 on Spinal Devices.
Current edition approved Dec. 1, 2013Oct. 1, 2021. Published February 2014October 2021. Originally approved in 1997. Last previous edition approved in 20082013 as
F1798 – 97F1798 – 13.(2008). DOI: 10.1520/F1798-13. 10.1520/F1798-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F1798 − 21
F1582F1717 Terminology Relating to Spinal ImplantsTest Methods for Spinal Implant Constructs in a Vertebrectomy Model
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 active length of longitudinal element—the span between rigid supports (for example, 50 mm is the active length in Fig. 1,
Fig. 2, Fig. 3((a),a), Fig. 3((b),b), and Fig. 4.).
3.1.2 global coordinate system—spinal column motion has six degrees of freedom, having translational motion along, and
rotational motion about three axes. The axes are labeled anterior-posterior or a-pA-P (X), medial-lateral or transverse (Y), and
caudal-cranial or axial (Z). This coordinate system is right handed with +X in the anterior direction, +Y towards the left side of
the body, and +Z in the cranial direction. Positive rotations are defined by the right hand rule (see Fig. 5((a)).a)).
3.1.3 gripping capacity—the maximum applied load or moment across an interconnection mechanism within the first 1.5 mm of
permanent displacement or 5° of permanent rotation between the connected components.
3.1.4 local coordinate system—the spine’s global coordinate system shall be applied locally at the position of the interconnection.
The local direction, z, shall be centered through the longitudinal element of the x-y plane. The local direction, x, shall be defined
as parallel to the axis of a screw or back of a hook. The local transverse axis, y, shall be parallel to a transverse element (See(see
Fig. 5((b)b) and Fig. 5((c)).c)).
3.1.5 loosening torque—the torque required to disconnect the various threaded fasteners that might comprise the implant’s
interconnection mechanism.of the spinal arthrodesis implant.
3.1.6 major directions of loading—directions of the predominant forces and moments (relative to the local axes) to which vertebral
connection elements are subjected, (that is, axial load, Fz; A-P load, Fx; axial torsion, Mz; and flexion-extension moment, My).
3.1.6 maximum run out run-out load/moment—the maximum load or moment that can be applied to a subassembly where all the
tested constructs have withstood 2.5 × 106 cycles without a failure.
3.1.7 relevant directions of loading—those directions of loading in which a particular component interconnection is designed to
provide resistance to loading. For example, a particular spinal hook may be designed to withstand a positive axial load, A-P load,
and flexion-extension moment, but not a negative axial load or axial torsion. Hence, positive axial load, A-P load, and
flexion-extension moment are the relevant directions of loading.
FIG. 1 A-P Test Apparatus for Subassembly
F1798 − 21
FIG. 2 Transverse Test Apparatus for Subassembly
3.1.8 spinal arthrodesis implant—an implant applied to the spine with the intention of providing that provides temporary
correction and stability to vertebrae while bony fusion occurs.
3.1.9 subassembly failure—permanent deformation resulting from fracture, plastic deformation, loosening, or slippage that renders
the subassembly ineffective or unable to adequately resist load.
3.1.10 subassembly permanent deformation—the displacement (mm) or angular displacement (degree of the subassembly relative
to the unloaded condition) remaining after the applied load moment or torque has been removed. Care must be taken to ensure that
the loading fixtures are rigid and do not contribute to the measurement of deflection.
3.1.11 tightening torque—the specified torque that is applied to the various threaded fasteners that might comprise the implant’s
interconnection mechanism.of the spinal arthrodesis implant during assembly.
3.1.12 ultimate load/moment of the subassembly—maximum load or moment applied to a subassembly (see Point E in Fig. 6).
3.1.13 yield load/moment of the subassembly—the load or moment required to produce a permanent deformation equal to 0.020
times the active length of the longitudinal element (see Point D in Fig. 6).
4. Summary of Test MethodsMethod
4.1 Vertebral attachment components (for example, hook, screws, bands) and transverse elements must be attached to longitudinal
elements (for example, rods, plates) to form spinal implant subassemblies.
4.2 The interconnections are tested only in the relevant directions of loading by applying loads at specific locations relative to the
local coordinate system.
4.3 The interconnections and subassemblies are tested statically in a load-to-failure mode and also can be tested cyclically to
estimate the maximum run out run-out value at 2.5 × 10 cycles.
5. Significance and Use
5.1 Spinal implants are generally composed of several components that, when connected together, form a spinal implant construct.
Spinal implant constructs are designed to provide some stability to the spine while arthrodesis takes place. This test method
F1798 − 21
FIG. 3 Flexion-Extension Moment Test Apparatus for Subassembly
F1798 − 21
FIG. 4 Transverse Moment Test Apparatus for Subassembly
outlines standardized evaluations of different interconnection mechanisms to facilitate comparison between different designs.
Comparisons must be made cautiously and with careful analysis, taking into account the effects that design differences can have
on the loading configurations.
5.2 This test method is used to quantify the static and fatigue properties of different implant interconnection designs. The
mechanical tests are conducted in vitro using simplified, unidirectional loads and moments. Fatigue testing in a simulated body
fluid or saline may have a fretting, corrosive, or lubricating effect on the interconnection and thereby affect the relative performance
of tested devices. Hence, the test environment, whether a simulated body fluid, saline (9g(9 g NaCl per 1000 mL H O), with a
saline drip, or dry, is an important characteristic of the test and must be reported accurately.
5.3 The loading of spinal implant constructs in vivo will, in general, differ from the loading configurations used in this test method.
The results obtained here cannot be used directly to predict in vivo performance. However, the results can be used to compare
different component designs in terms of relative mechanical parameters.
F1798 − 21
F1798 − 21
FIG. 5 Coordinate System
FIG. 6 Load/Displacement Curve
6. Apparatus
6.1 Machines used for the test shall conform to the requirements of Practices E4.
6.2 The apparatus for axial (z) gripping capacity measurements of an interconnection mechanism is depicted in Fig. 7((a).a). One
F1798 − 21
FIG. 7 Axial Gripping Capacity Test Apparatus
end of the longitudinal element shall be clamped rigidly, leaving 5 mm exposed between the interconnection mechanism and the
test machine base. A section of longitudinal element at least 5 mm shall extend beyond the interconnection linkage and remain
unfixed. Axial loads are applied to the interconnection mechanism along the axis of the longitudinal element via a sleeve (collar)
which freely surrounds the longitudinal element. The sleeve (collar) should evenly distribute the load around the interconnection.
An alternate method, depicted in Fig. 7((b),b), applies the load to the longitudinal element and pushes it through the
interconnection clamp.
6.3 The apparatus for A-P (x) mechanical property measurements of a subassembly is depicted in Fig. 1. Both ends of the
longitudinal element shall be clamped rigidly, with the interconnection centered on a 50-mm section of the longitudinal element.
The local origin of the interconnection mechanism shall be centered between the mounts. Loads are applied to the interconnection
(perpendicular to the longitudinal element) via a clamp on the hook, screw, or band. The load should be centered through the local
x coordinate x-coordinate axis.
6.4 The apparatus for transverse (y) mechanical property measurements of a subassembly is depicted in Fig. 2. Both ends of the
longitudinal element shall be clamped rigidly, with the interconnection centered on a 50-mm section of the longitudinal element.
The local origin of the interconnection mechanism shall be centered between the mounts. Loads are applied to the interconnection
(perpendicular to the longitudinal element) via a clamp on the transverse connector. The load should be centered through the local
y coordinate y-coordinate axis.
6.5 The apparatus for flexion-extension moment (My) mechanical property measurements of a subassembly is depicted in Fig. 3.
Both ends of the longitudinal element shall be clamped rigidly, with the interconnection centered on a 50-mm section of the
longitudinal element. The local origin of the interconnection mechanism shall be centered between the mounts. Loads are applied
to the interconnection (parallel to the longitudinal element). For spinal hooks, the load shall be applied via a cylinder set in the
hook notch (see Fig. 3((a)).a)). For other elements (screws) the load shall be applied 25 mm from the local z axis z-axis (see Fig.
3((b)).b)).
6.6 The apparatus for transverse moment (Mx) mechanical property measurements of a subassembly is depicted in Fig. 4. As in
the previous test, 6.5, both ends of the longitudinal element shall be clamped rigidly, with the interconnection centered on a 50-mm
section of the longitudinal element. The local origin of the interconnection mechanism shall be centered between the mounts. Loads
are applied to the interconnection (parallel to the longitudinal element), 25 mm from the z axis.z-axis.
6.7 The apparatus for axial torque (Mz) gripping capacity measurements of an interconnection mechanism is depicted in Fig.
F1798 − 21
8((a)a) and is similar to that described in 6.2, with the exception that the axial torque is applied via notches in the sleeve that
surrounds the longitudinal element. An alternative method is to hold the interconnection rigidly and apply the torsional force to
the longitudinal element as shown in Fig. 8((b).b). A third alternative is to apply the torque via a force applied to a moment arm
as shown in Fig. 8((c),c), but this alternative may introduce an additional variable of bending of the anchor component. In any case,
care must be taken to evaluate and minimize the effect of the torsional properties of the longitudinal element on the results.
6.8 For each of these apparatuses, care must be taken to ensure that the loading fixtures are rigid and do not contribute to the
measurement of deflection.
7. Sampling
7.1 The samples tested shall be previously unused parts, and shall not be re-tested.
7.2 The test constructs shall be labeled and maintained according to good laboratory practice.
7.3 Static tests of mechanical properties should have a minimum sample size of five.
7.4 Fatigue tests for determining the maximum run out run-out load or moment of a subassembly at 2.5 × 10 cycles shall utilize
a run down, rundown, half-interval approach with one specimen per run down interval or half-interval and three consecutive
specimens
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...