ASTM F2580-18
(Practice)Standard Practice for Evaluation of Modular Connection of Proximally Fixed Femoral Hip Prosthesis
Standard Practice for Evaluation of Modular Connection of Proximally Fixed Femoral Hip Prosthesis
SIGNIFICANCE AND USE
4.1 This practice can be used to describe the effects of materials, manufacturing, and design variables on the fatigue performance of metallic femoral hip prostheses subject to cyclic loading for large numbers of cycles.
4.2 The loading of femoral hip designs in vivo will, in general, differ from the loading defined in this practice. The results obtained here cannot be used to directly predict in vivo performance. However, this practice is designed to allow for comparisons between the fatigue performance of different metallic femoral hip designs when tested under similar conditions.
4.3 In order for fatigue data on femoral hip prostheses to be comparable, reproducible, and capable of being correlated among laboratories, it is essential that uniform procedures be established.
SCOPE
1.1 This practice covers a procedure for the fatigue testing of metallic femoral hip prostheses used in hip joint replacements. This practice covers the procedures for the performance of fatigue tests on metallic femoral hip stems using a cyclic, constant-amplitude force. It applies to hip prostheses that utilize proximal metaphyseal fixation and are of a modular construct, and it is intended to evaluate the fatigue performance of the modular connections in the metaphyseal filling (that is, proximal body) region of the stem.
1.2 This practice is intended to provide useful, consistent, and reproducible information about the fatigue performance of metallic hip prostheses while held in a proximally fixated manner, with the distal end not held by a potting medium.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.22 - Arthroplasty
Relations
- Effective Date
- 15-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2020
- Effective Date
- 15-Dec-2012
- Effective Date
- 15-Nov-2012
- Effective Date
- 01-Sep-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 15-May-2012
- Effective Date
- 15-Mar-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Jun-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Jan-2010
- Effective Date
- 01-Jul-2009
Overview
ASTM F2580-18: Standard Practice for Evaluation of Modular Connection of Proximally Fixed Femoral Hip Prosthesis is an internationally recognized guideline developed by ASTM International. This standard specifies procedures for fatigue testing of metallic femoral hip prostheses, particularly those used in hip joint replacements with proximal metaphyseal fixation and modular construction. By establishing uniform testing and reporting protocols, ASTM F2580-18 enables consistent evaluation and comparison of fatigue performance across various femoral hip prosthesis designs. This contributes to high standards in orthopedic medical device development, supporting improved reliability and clinical outcomes.
Key Topics
- Fatigue Testing Procedure: The standard outlines steps for subjecting metallic femoral hip stems to cyclic, constant-amplitude force, focusing on the modular connections within the proximal body of the stem.
- Specimen Selection Criteria: Only finished components with the same geometry as the final product are used, selecting the worst-case size and configuration to ensure thorough testing.
- Test Setup and Apparatus:
- Specific fixture alignment and potting requirements are defined to replicate physiological loading conditions without distal fixation.
- The use of validated fatigue test machines and monitoring systems ensures accurate force application throughout testing.
- Reporting Requirements: To ensure transparency and reproducibility, comprehensive reporting of test parameters, implant details, potting procedures, assembly methods, and failure modes is required.
- Significance of Uniform Procedures: Establishes comparability and reproducibility of fatigue data across different laboratories, manufacturers, and research facilities.
Applications
ASTM F2580-18 is practically applied in several key areas within the medical device and orthopedic implant sectors:
- Design and Development of Hip Prostheses: Manufacturers use the standard to benchmark new modular femoral hip designs, ensuring that products meet industry fatigue performance expectations before market release.
- Quality Assurance and Regulatory Compliance: The guideline is instrumental in demonstrating product durability and safety to regulatory bodies, supporting both initial approvals and ongoing product surveillance.
- Research and Comparative Studies: Academic and clinical researchers rely on this practice to generate reproducible fatigue data, enabling independent comparisons between metallic hip implant designs or manufacturing methods.
- Risk Management and Material Selection: Insights from testing inform materials engineering, surgical technique development, and the identification of design-related failure modes that can influence long-term clinical performance.
- Global Trade and Standardization: The standard supports harmonization of testing protocols internationally, facilitating compliance with global regulations and trade requirements.
Related Standards
For a comprehensive understanding and implementation, ASTM F2580-18 should be considered alongside the following related standards:
- ASTM E467: Practice for Verification of Constant Amplitude Dynamic Forces in an Axial Fatigue Testing System.
- ASTM E468: Practice for Presentation of Constant Amplitude Fatigue Test Results for Metallic Materials.
- ISO 7206-4: Implants for surgery - Partial and total hip joint prostheses – Part 4: Determination of endurance properties and performance of stemmed femoral components.
These standards complement ASTM F2580-18 by providing additional methodologies for test verification, data presentation, terminology, and alignment with international best practices in orthopedic device fatigue testing.
Keywords: ASTM F2580-18, modular femoral hip prosthesis, fatigue testing, proximal fixation, orthopedic implant standards, hip replacement, metallic hip stems, medical device evaluation, fatigue performance, modular connection, arthroplasty devices.
Buy Documents
ASTM F2580-18 - Standard Practice for Evaluation of Modular Connection of Proximally Fixed Femoral Hip Prosthesis
REDLINE ASTM F2580-18 - Standard Practice for Evaluation of Modular Connection of Proximally Fixed Femoral Hip Prosthesis
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F2580-18 is a standard published by ASTM International. Its full title is "Standard Practice for Evaluation of Modular Connection of Proximally Fixed Femoral Hip Prosthesis". This standard covers: SIGNIFICANCE AND USE 4.1 This practice can be used to describe the effects of materials, manufacturing, and design variables on the fatigue performance of metallic femoral hip prostheses subject to cyclic loading for large numbers of cycles. 4.2 The loading of femoral hip designs in vivo will, in general, differ from the loading defined in this practice. The results obtained here cannot be used to directly predict in vivo performance. However, this practice is designed to allow for comparisons between the fatigue performance of different metallic femoral hip designs when tested under similar conditions. 4.3 In order for fatigue data on femoral hip prostheses to be comparable, reproducible, and capable of being correlated among laboratories, it is essential that uniform procedures be established. SCOPE 1.1 This practice covers a procedure for the fatigue testing of metallic femoral hip prostheses used in hip joint replacements. This practice covers the procedures for the performance of fatigue tests on metallic femoral hip stems using a cyclic, constant-amplitude force. It applies to hip prostheses that utilize proximal metaphyseal fixation and are of a modular construct, and it is intended to evaluate the fatigue performance of the modular connections in the metaphyseal filling (that is, proximal body) region of the stem. 1.2 This practice is intended to provide useful, consistent, and reproducible information about the fatigue performance of metallic hip prostheses while held in a proximally fixated manner, with the distal end not held by a potting medium. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This practice can be used to describe the effects of materials, manufacturing, and design variables on the fatigue performance of metallic femoral hip prostheses subject to cyclic loading for large numbers of cycles. 4.2 The loading of femoral hip designs in vivo will, in general, differ from the loading defined in this practice. The results obtained here cannot be used to directly predict in vivo performance. However, this practice is designed to allow for comparisons between the fatigue performance of different metallic femoral hip designs when tested under similar conditions. 4.3 In order for fatigue data on femoral hip prostheses to be comparable, reproducible, and capable of being correlated among laboratories, it is essential that uniform procedures be established. SCOPE 1.1 This practice covers a procedure for the fatigue testing of metallic femoral hip prostheses used in hip joint replacements. This practice covers the procedures for the performance of fatigue tests on metallic femoral hip stems using a cyclic, constant-amplitude force. It applies to hip prostheses that utilize proximal metaphyseal fixation and are of a modular construct, and it is intended to evaluate the fatigue performance of the modular connections in the metaphyseal filling (that is, proximal body) region of the stem. 1.2 This practice is intended to provide useful, consistent, and reproducible information about the fatigue performance of metallic hip prostheses while held in a proximally fixated manner, with the distal end not held by a potting medium. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2580-18 is classified under the following ICS (International Classification for Standards) categories: 11.040.40 - Implants for surgery, prosthetics and orthotics. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2580-18 has the following relationships with other standards: It is inter standard links to ASTM E1823-24a, ASTM E1823-24, ASTM E1823-20, ASTM E1823-12e, ASTM E1823-12d, ASTM E1823-12c, ASTM E1823-12b, ASTM E1823-12a, ASTM E1823-12, ASTM E467-08e1, ASTM E468-11, ASTM E1823-11, ASTM E1823-10a, ASTM E1823-10, ASTM E1823-09b. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2580-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2580 − 18
Standard Practice for
Evaluation of Modular Connection of Proximally Fixed
Femoral Hip Prosthesis
This standard is issued under the fixed designation F2580; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E467 Practice for Verification of Constant Amplitude Dy-
namic Forces in an Axial Fatigue Testing System
1.1 This practice covers a procedure for the fatigue testing
E468 Practice for Presentation of Constant Amplitude Fa-
of metallic femoral hip prostheses used in hip joint replace-
tigue Test Results for Metallic Materials
ments. This practice covers the procedures for the performance
E1823 Terminology Relating to Fatigue and Fracture Testing
of fatigue tests on metallic femoral hip stems using a cyclic,
2.2 ISO Standards:
constant-amplitude force. It applies to hip prostheses that
ISO 7206–4 Implants for surgery -- Partial and total hip joint
utilize proximal metaphyseal fixation and are of a modular
prostheses -- Part 4: Determination of endurance proper-
construct, and it is intended to evaluate the fatigue performance
ties and performance of stemmed femoral components
of the modular connections in the metaphyseal filling (that is,
proximal body) region of the stem.
3. Terminology
1.2 This practice is intended to provide useful, consistent,
3.1 Definitions:
and reproducible information about the fatigue performance of
3.1.1 R value, n—The R value is the ratio of the minimum
metallic hip prostheses while held in a proximally fixated
load to the maximum load.
manner, with the distal end not held by a potting medium.
minimum load
1.3 The values stated in SI units are to be regarded as
R 5
maximum load
standard. No other units of measurement are included in this
3.2 Definitions of Terms Specific to This Standard:
standard.
3.2.1 extraction, n—removal of the femoral hip implant
1.4 This standard does not purport to address all of the
from the femur during surgery.
safety concerns, if any, associated with its use. It is the
3.2.2 extractor hole, n—a hole in the proximal body of the
responsibility of the user of this standard to establish appro-
stem in which an apparatus is placed to remove the implant
priate safety, health, and environmental practices and deter-
from the femur.
mine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accor-
3.2.3 femoral head, n—convex spherical bearing member
dance with internationally recognized principles on standard-
for articulation with the natural acetabulum or prosthetic
ization established in the Decision on Principles for the
acetabulum.
Development of International Standards, Guides and Recom-
3.2.4 femoral head offset, n—the perpendicular distance
mendations issued by the World Trade Organization Technical
from the centerline of the implant stem to the center of the
Barriers to Trade (TBT) Committee.
femoral head.
3.2.5 frontal plane, n—the plane that lies in the medial-
2. Referenced Documents
lateral direction of the implant. Adduction occurs in this plane.
2.1 ASTM Standards:
3.2.6 implant centerline, n—the axis that runs vertically
from the proximal body of the implant down the center of the
stem to the distal end.
This practice is under the jurisdiction of ASTM Committee F04 on Medical and
3.2.7 pivot axis, n—the center of rotation of the pivot fixture
Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.22 on Arthroplasty. (and prosthesis potted within it) within the test fixture setup; its
Current edition approved July 1, 2018. Published September 2018. Originally
location is determined by the intersection of the neck and stem
approved in 2007. Last previous edition approved in 2013 as F2580 – 13. DOI:
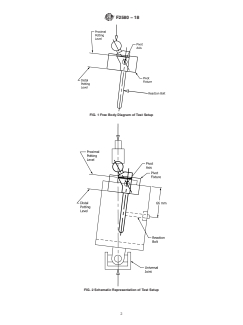
centerlines of the prothesis (Figs. 1 and 2).
10.1520/F2580-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2580 − 18
FIG. 1 Free Body Diagram of Test Setup
FIG. 2 Schematic Representation of Test Setup
F2580 − 18
3.2.8 pivot fixture, n—the fixture in which the specimen is of the reaction bolt should be adjustable to accommodate stems
potted, and is attached to the main test fixture; characterized by of different lengths and design features.
two pins on the side that serve as the pivot axis.
6.5 The fixtures and aligning materials used should be of a
3.2.9 rotational plane, n—the plane that lies perpendicular
design that positions the implant, when potted, so that: the
to the stem axis of the implant.
point defined by the intersection of the neck and stem center-
lines is coincident with the pivot axis (Fig. 1), the stem is fixed
3.2.10 sagittal plane, n—the plane that lies perpendicular to
vertically in both medial/lateral and anterior/posterior
the frontal plane; flexion occurs in this plane.
directions, the stem is aligned facing forward in the rotational
plane (that is, the frontal plane is normal to the pivot axis of the
4. Significance and Use
fixture (Fig. 3)), and that any mating surfaces between modular
4.1 This practice can be used to describe the effects of
components of the specimen do not come in contact with the
materials, manufacturing, and design variables on the fatigue
potting medium.
performance of metallic femoral hip prostheses subject to
cyclic loading for large numbers of cycles.
7. Equipment Characteristics
4.2 The loading of femoral hip designs in vivo will, in
7.1 Perform the tests on a fatigue test machine with ad-
general, differ from the loading defined in this practice. The
equate load capacity.
results obtained here cannot be used to directly predict in vivo
7.2 Analyze the action of the machine to ensure that the
performance. However, this practice is designed to allow for
desired form and periodic force amplitude is maintained for the
comparisons between the fatigue performance of different
duration of the test (see Practice E467 or use a validated
metallic femoral hip designs when tested under similar condi-
strain-gauged part).
tions.
7.3 The test machine shall have a load monitoring system
4.3 In order for fatigue data on femoral hip prostheses to be
such as a transducer mounted in line with the specimen.
comparable, reproducible, and capable of being correlated
Monitor the test loads continuously in the early stages of the
among laboratories, it is essential that uniform procedures be
test and periodically thereafter to ensure the desired load cycle
established.
is maintained. Maintain the varying load as determined by
suitable dynamic verification at all times to within 62 % of the
5. Specimen Selection
largest compressive force being used.
5.1 The test component selected shall have the same geom-
etry as the final product, and shall be in finished condition. The
8. Procedure
test component shall be of the worst-case size and configura-
8.1 This procedure details a potting method centered about
tion (that is, the component that produces the highest stresses)
potting the proximal body portion of the implant first, and
of the implant family to be tested.
assembling the remainder of the implant after potting. Other
5.2 The femoral head component selected for load applica-
methods of potting the specimen exist, including methods for
tion shall be of the same design and material as a current
implants that are not of a modular design, and may be used in
product in use, but may be previously tested.
place of this, providing that the general terms and limitations
5.3 The femoral head selected shall offer the greatest load
offset from the hip centerline, to represent a worst-case
bending scenario during testing.
6. Apparatus
6.1 The hip implant may be tested in different orientations
to better reproduce specific testing conditions that are being
evaluated. For example: An anatomical orientation of 9°
flexion, and 10° adduction (per ISO 7206-4), or vertically in
both planes. The criteria used to determine the orientation
should be reported.
6.2 Care shall be taken to ensure that the fixation of the
implant does not produce abnormal stress concentrations that
could change the failure mode of the part.
6.3 A fixed-bearing load applicator shall be used to keep the
specimen aligned in the chosen orientation during testing, as
well as a fixture that allows the stem to bend during testing,
such as a u-joint.
NOTE 1—Once assembled, the pivot axis will be coincident with the
6.4 The fixture used to hold the implant during testing
point on the implant defined by the intersection of the neck and stem
should have a reaction bolt that will oppose the loading on the
centerlines.
femoral head, keeping the implant in equilibrium. The position FIG. 3 Proximal Sleeve Component Potted in Pivot Fixture
F2580 − 18
are still
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2580 − 13 F2580 − 18
Standard Practice for
Evaluation of Modular Connection of Proximally Fixed
Femoral Hip Prosthesis
This standard is issued under the fixed designation F2580; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers a procedure for the fatigue testing of metallic femoral hip prostheses used in hip joint replacements.
This practice covers the procedures for the performance of fatigue tests on metallic femoral hip stems using a cyclic,
constant-amplitude force. It applies to hip prostheses that utilize proximal metaphyseal fixation and are of a modular construct, and
it is intended to evaluate the fatigue performance of the modular connections in the metaphyseal filling (that is, proximal body)
region of the stem.
1.2 This practice is intended to provide useful, consistent, and reproducible information about the fatigue performance of
metallic hip prostheses while held in a proximally fixated manner, with the distal end not held by a potting medium.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E467 Practice for Verification of Constant Amplitude Dynamic Forces in an Axial Fatigue Testing System
E468 Practice for Presentation of Constant Amplitude Fatigue Test Results for Metallic Materials
E1150E1823 Definitions of Terms Terminology Relating to Fatigue and Fracture Testing (Withdrawn 1996)
2.2 ISO Standards:
ISO 7206–4 Determination of Endurance Properties of Stemmed Femoral Components with Application of TorsionImplants for
surgery -- Partial and total hip joint prostheses -- Part 4: Determination of endurance properties and performance of stemmed
femoral components
3. Terminology
3.1 Definitions:
3.1.1 R value, n—The R value is the ratio of the minimum load to the maximum load.
minimum load
R 5
maximum load
3.2 Definitions of Terms Specific to This Standard:
3.2.1 extraction—extraction, n—removal of the femoral hip implant from the femur during surgery.
This practice is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.22
on Arthroplasty.
Current edition approved Feb. 1, 2013July 1, 2018. Published February 2013September 2018. Originally approved in 2007. Last previous edition approved in 20092013
as F2580 – 09.F2580 – 13. DOI: 10.1520/F2580-13.10.1520/F2580-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2580 − 18
3.2.2 extractor hole—hole, n—a hole in the proximal body of the stem in which an apparatus is placed to remove the implant
from the femur.
3.2.3 femoral head—head, n—convex spherical bearing member for articulation with the natural acetabulum or prosthetic
acetabulum.
3.2.4 femoral head offset—offset, n—the perpendicular distance from the centerline of the implant stem to the center of the
femoral head.
3.2.5 frontal plane—plane, n—the plane that lies in the medial-lateral direction of the implant. Adduction occurs in this plane.
3.2.6 implant centerline—centerline, n—the axis that runs vertically from the proximal body of the implant,implant down the
center of the stem to the distal end.
3.2.7 pivot axis—axis, n—the center of rotation of the pivot fixture (and prosthesis potted within it) within the test fixture setup;
its location is determined by the intersection of the neck and stem centerlines of the prothesis (Figs. 1 and 2).
3.2.8 pivot fixture—fixture, n—the fixture in which the specimen is potted, and is attached to the main test fixture; characterized
by two pins on the side that serve as the pivot axis.
3.2.9 rotational plane—plane, n—the plane that lies perpendicular to the stem axis of the implant.
3.2.10 sagittal plane—plane, n—the plane that lies perpendicular to the Frontalfrontal plane; flexion occurs in this plane.
4. Significance and Use
4.1 This practice can be used to describe the effects of materials, manufacturing, and design variables on the fatigue
performance of metallic femoral hip prostheses subject to cyclic loading for large numbers of cycles.
4.2 The loading of femoral hip designs in vivo will, in general, differ from the loading defined in this practice. The results
obtained here cannot be used to directly predict in vivo performance. However, this practice is designed to allow for comparisons
between the fatigue performance of different metallic femoral hip designs,designs when tested under similar conditions.
4.3 In order for fatigue data on femoral hip prostheses to be comparable, reproducible, and capable of being correlated among
laboratories, it is essential that uniform procedures be established.
5. Specimen Selection
5.1 The test component selected shall have the same geometry as the final product, and shall be in finished condition. The test
component shall be of the worst-case size and configuration (that is, the component that produces the highest stresses) of the
implant family to be tested.
5.2 The femoral head component selected for load application shall be of the same design and material as a current product in
use, but may be previously tested.
5.3 The femoral head selected shall offer the greatest load offset from the hip centerline, to represent a worst-case bending
scenario during testing.
FIG. 1 Free Body Diagram of Test Setup
F2580 − 18
FIG. 2 Schematic Representation of the Test Set-upSetup
6. Apparatus
6.1 The hip implant may be tested in different orientations to better reproduce specific testing conditions that are being
evaluated. For example: An anatomical orientation of 9° flexion, and 10° adduction (per ISO 7206-4), or vertically in both planes.
The criteria used to determine the orientation should be reported.
6.2 Care shall be taken to ensure that the fixation of the implant does not produce abnormal stress concentrations that could
change the failure mode of the part.
6.3 A fixed-bearing load applicator shall be used to keep the specimen aligned in the chosen orientation during testing, as well
as a fixture that allows the stem to bend during testing, such as a u-joint.
6.4 The fixture used to hold the implant during testing should have a reaction bolt that will oppose the loading on the femoral
head, keeping the implant in equilibrium. The position of the reaction bolt should be adjustable to accommodate stems of different
lengths and design features.
6.5 The fixtures and aligning materials used should be of a design that positions the implant, when potted, so that: the point
defined by the intersection of the neck and stem centerlines is coincident with the pivot axis (Fig. 1), the stem is fixed vertically
in both medial/lateral and anterior/posterior directions, the stem is aligned facing forward in the rotational plane (that is, the frontal
plane is normal to the pivot axis of the fixture),fixture (Fig. 3))), and that any mating surfaces between modular components of
the specimen do not come in contact with the potting medium.
7. Equipment Characteristics
7.1 Perform the tests on a fatigue test machine with adequate load capacity.
F2580 − 18
NOTE 1—Once assembled, the pivot axis will be coincident with the point on the implant defined by the intersection of the neck and stem centerlines.
FIG. 3 Proximal Sleeve Component Potted in Pivot Fixture
7.2 Analyze the action of the machine to ensure that the desired form and periodic force amplitude is maintained for the duration
of the test (see Practice E467 or use a validated strain-gauged part).
7.3 The test machine shall have a load monitoring system such as a transducer mounted in line with the specimen. Monitor the
test loads continuously in the early stages of the test and periodically thereafter to ensure the desired load cycle is maintained.
Maintain the varying load as determined by suitable dynamic verification at all times to within 62 % of the largest compressive
force being used.
8. Procedure
8.1 This procedure details a potting method centered about potting the proximal body portion of the implant first, and
assembling the remainder of the implant after potting. Other methods of potting the specimen exist, including methods for implants
that
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...